Advanced FeCl3-Catalyzed C-H Coupling for Industrial Alkyl Olefin Production
Advanced FeCl3-Catalyzed C-H Coupling for Industrial Alkyl Olefin Production
The landscape of organic synthesis is constantly evolving towards more sustainable and cost-effective methodologies, particularly in the production of high-value olefinic intermediates essential for the pharmaceutical and agrochemical sectors. A groundbreaking development detailed in Chinese Patent CN112299945A introduces a highly efficient method for synthesizing hydrocarbyl olefins through the direct coupling of double-bond carbon-hydrogen bonds with saturated carbon-hydrogen bonds. This innovative approach utilizes readily available olefins and sulfoxides in a one-pot reaction mediated by inexpensive iron salts and hydrogen peroxide. By replacing traditional precious metal catalysts and complex alkylating agents with earth-abundant iron and common solvents like DMSO, this technology represents a paradigm shift in cost reduction in fine chemical manufacturing. The process not only achieves high yields and excellent selectivity but also operates under mild, atmospheric conditions, making it exceptionally suitable for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alkylation of olefins to generate higher-order substituted structures has relied heavily on classical cross-coupling reactions such as the Heck reaction, which typically necessitates the use of expensive palladium catalysts and pre-functionalized halogenated hydrocarbons. As illustrated in prior art schemes, earlier methods often required complex alkylating agents containing carbon-halogen, carbon-nitrogen, or carbon-oxygen bonds, which are not only costly to synthesize but also generate significant amounts of hazardous halogenated waste. Furthermore, many of these traditional protocols are limited in their substrate scope, frequently failing to accommodate sterically hindered tri-substituted olefins or aliphatic cyclic olefins. The reliance on precious metals like palladium or cobalt introduces severe supply chain vulnerabilities and necessitates rigorous downstream purification to meet stringent residual metal specifications required for high-purity pharmaceutical intermediates. These cumulative factors create substantial barriers to industrial application, driving up production costs and complicating regulatory compliance.
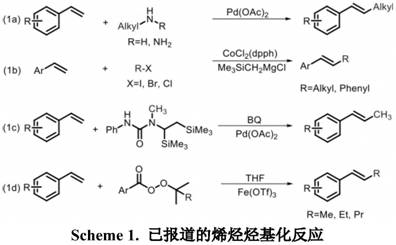
The Novel Approach
In stark contrast to these legacy methods, the disclosed invention leverages a radical-mediated mechanism that bypasses the need for pre-functionalized substrates entirely. By employing sulfoxides, specifically dimethyl sulfoxide (DMSO), as both the solvent and the source of the alkyl group, the process achieves remarkable atom economy and operational simplicity. The reaction proceeds via a direct C-H activation pathway where a hydrogen atom on the olefin carbon is replaced by a hydrocarbon group derived from the sulfoxide, effectively elongating the carbon chain without generating stoichiometric salt byproducts. This methodology is universally applicable to both aryl-terminal and non-aryl terminal olefins, offering a versatile platform for constructing diverse polysubstituted olefin architectures. The use of iron salts as catalysts, coupled with hydrogen peroxide as a green oxidant, ensures that the process remains economically viable and environmentally benign, addressing the critical pain points of cost and sustainability faced by modern chemical manufacturers.
Mechanistic Insights into FeCl3-Catalyzed Radical Coupling
Understanding the underlying mechanistic pathway is crucial for R&D directors aiming to optimize this process for specific API intermediates. Detailed mechanistic studies, including radical trapping experiments with BHT and deuterium labeling, confirm that the reaction proceeds through a free-radical mechanism initiated by the interaction between the iron catalyst and hydrogen peroxide. The proposed catalytic cycle begins with the generation of hydroxyl radicals (•OH) via the Fenton-like reaction between Fe3+/Fe2+ and H2O2. These highly reactive hydroxyl radicals then abstract a hydrogen atom from the sulfoxide solvent (DMSO), generating a methyl radical (•CH3). This methyl radical subsequently adds to the double bond of the olefin substrate, forming a stabilized benzylic or alkyl radical intermediate. This intermediate is then trapped by another hydroxyl radical to form a transient alcohol species, which undergoes thermal trans-elimination dehydration under the reaction heating conditions to yield the final alkylated olefin product. This elegant cascade avoids the formation of stable organometallic intermediates typical of palladium cycles, thereby reducing the risk of catalyst poisoning and side reactions.
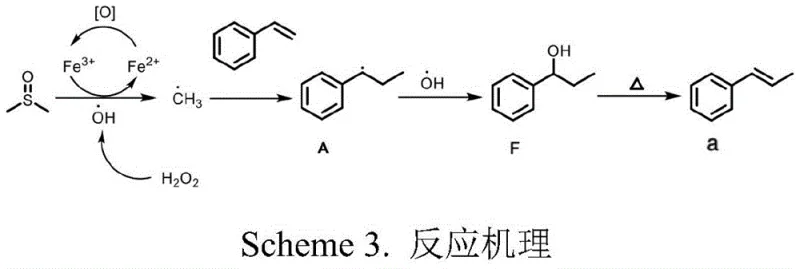
From an impurity control perspective, this radical mechanism offers distinct advantages regarding selectivity and byproduct profiles. Unlike ionic pathways that might suffer from rearrangement issues or regio-selectivity problems, the radical addition to the terminal double bond is highly regioselective, favoring the formation of the more stable radical intermediate. The subsequent dehydration step is driven thermodynamically to form the conjugated olefin system, ensuring high stereo- and regio-selectivity. Experimental data indicates that even with complex substrates containing electron-withdrawing or electron-donating groups, the reaction maintains high fidelity, producing the desired (Z)- or (E)-alkyl olefins with minimal isomerization. For quality assurance teams, this implies a cleaner crude reaction profile, which simplifies the purification workflow and enhances the overall recovery of the target high-purity alkyl olefins, ultimately supporting tighter control over the impurity spectrum in the final drug substance.
How to Synthesize (Z)-2-Methylstyrene Efficiently
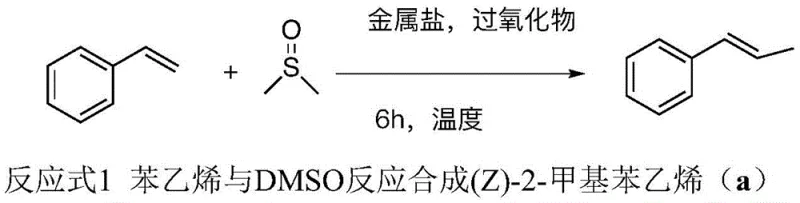
The synthesis of (Z)-2-methylstyrene serves as a benchmark reaction to demonstrate the efficacy of this iron-catalyzed protocol. The standard procedure involves a straightforward one-pot setup where styrene is reacted with DMSO in the presence of FeCl3·6H2O and aqueous hydrogen peroxide. The reaction is heated to 140°C for 6 hours, after which the product is isolated via simple extraction and column chromatography. This protocol has been optimized to balance reaction rate and yield, ensuring that the radical generation is sustained throughout the process without excessive decomposition of the oxidant. For process chemists looking to implement this route, the key parameters to monitor include the stoichiometry of hydrogen peroxide, which should ideally be maintained at 4 to 6 equivalents relative to the olefin, and the reaction temperature, which is critical for the final elimination step. Detailed standardized synthetic steps for this transformation and its variations are provided in the technical guide below.
- Prepare the reaction mixture by combining the olefin substrate, dimethyl sulfoxide (DMSO) as both solvent and alkylating agent, ferric chloride hexahydrate (FeCl3·6H2O) as the catalyst, and hydrogen peroxide (H2O2) as the oxidant in a Schlenk tube.
- Seal the reaction vessel and heat the mixture to 140°C under an atmospheric atmosphere with magnetic stirring for approximately 6 hours to facilitate the radical coupling reaction.
- Upon completion, cool the reaction, extract the product with ethyl acetate, wash with water to remove DMSO, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this iron-catalyzed sulfoxide alkylation method presents a compelling value proposition centered on cost stability and operational resilience. By eliminating the dependency on volatile precious metal markets for palladium or rhodium catalysts, manufacturers can significantly decouple their production costs from fluctuating commodity prices. Furthermore, the dual functionality of DMSO as both reagent and solvent drastically reduces the volume of raw materials required, leading to substantial reductions in logistics costs and storage requirements. The ability to run the reaction under atmospheric pressure without the need for specialized high-pressure reactors or inert gas manifolds further lowers the barrier to entry for contract manufacturing organizations (CMOs), enabling faster technology transfer and more flexible production scheduling. These factors collectively contribute to a more robust and predictable supply chain for critical olefinic building blocks.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with commodity-grade iron salts represents a direct and significant decrease in catalyst expenditure, which is often a major cost driver in fine chemical synthesis. Additionally, the use of DMSO as the alkylating agent removes the need for purchasing specialized alkyl halides or organometallic reagents, which are typically priced at a premium due to their complex synthesis and handling requirements. The simplified workup procedure, which avoids the need for extensive heavy metal scavenging steps, further reduces the consumption of auxiliary chemicals and processing time. Consequently, the overall cost of goods sold (COGS) for the final olefin product is markedly lower, enhancing profit margins for downstream applications in pharmaceutical and agrochemical formulations.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit immensely from the use of ubiquitous raw materials such as iron chloride, hydrogen peroxide, and DMSO, all of which are produced on a massive global scale with stable supply lines. Unlike specialized ligands or custom-synthesized halides that may have single-source suppliers and long lead times, these commodity chemicals are readily available from multiple vendors worldwide. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction conditions allows for manufacturing in a wider range of facilities, increasing the available capacity pool and ensuring consistent delivery schedules for reliable pharmaceutical intermediate supplier networks.
- Scalability and Environmental Compliance: The environmental footprint of this process is significantly reduced compared to traditional methods, aligning with increasingly strict global regulations on waste disposal and emissions. The absence of halogenated byproducts and heavy metal residues simplifies wastewater treatment and reduces the burden on EHS (Environment, Health, and Safety) departments. From a scalability standpoint, the exothermic nature of the radical reaction is manageable under the described conditions, and the lack of sensitive reagents allows for safe scaling from kilogram to multi-ton batches. This ease of scale-up ensures that the technology can seamlessly transition from pilot plant validation to full commercial production without the need for extensive re-engineering of the process equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this C-H coupling technology. These insights are derived directly from the experimental data and optimization studies presented in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: What are the primary advantages of using DMSO in this alkylation process?
A: DMSO serves a dual role as both the benign solvent and the alkylating reagent, eliminating the need for expensive, pre-functionalized alkyl halides and reducing overall material costs while simplifying waste treatment.
Q: Can this method be applied to sterically hindered substrates?
A: Yes, unlike many traditional palladium-catalyzed methods, this iron-catalyzed radical process demonstrates excellent tolerance for sterically hindered tri-substituted olefins and various functional groups including halogens and esters.
Q: Is the reaction sensitive to air or moisture?
A: The process is robust and operates effectively under an atmospheric atmosphere without the need for rigorous inert gas protection or anhydrous conditions, significantly lowering operational complexity for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Olefin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed C-H coupling technology in streamlining the production of complex olefinic intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of radical reactions, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We are committed to leveraging such innovative synthetic methodologies to deliver superior value to our global clientele.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your target molecule. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in your supply chain for high-performance chemical intermediates.
