Advanced Nickel-Catalyzed Alkylamine Synthesis for Scalable Pharmaceutical Manufacturing
Introduction to Next-Generation Alkylamine Synthesis
The synthesis of alkylamine derivatives represents a cornerstone challenge in modern organic chemistry, particularly for the development of complex pharmaceutical intermediates and functional materials. A groundbreaking methodology disclosed in patent CN113105374A introduces a highly efficient nickel-catalyzed reductive coupling strategy that fundamentally shifts the paradigm of amine alkylation. This innovative approach utilizes a metallic nickel catalyst to facilitate hydrogen transfer directly from the reactant amine, thereby enabling the coupling of amines and olefins to construct diverse alkylamine structures under a unified catalytic system. Unlike conventional pathways that often rely on pre-functionalized substrates or stoichiometric reducing agents, this method achieves atom economy by integrating the hydrogen source within the reaction mechanism itself. The technology demonstrates exceptional compatibility with a broad spectrum of amine compounds and aryl or alkyl olefins, offering a versatile platform for generating high-purity pharmaceutical intermediates. Furthermore, the protocol has been validated to maintain robust yields even upon scaling to gram-level quantities, signaling its strong potential for industrial adoption and reliable supply chain integration.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of alkylamine derivatives has been fraught with significant synthetic hurdles that impact both cost and operational efficiency in pharmaceutical manufacturing. Traditional routes frequently involve the reduction of imines or enamines, a process complicated by the inherent instability of these substrates which are prone to decomposition and difficult to store. Moreover, these methods typically necessitate the use of excess reducing agents and often depend on expensive noble metal catalysts such as ruthenium, rhodium, or iridium to achieve chirality, driving up the cost of goods significantly. Another prevalent method involves the nucleophilic addition of metal reagents to imines, which generates substantial metal waste and requires costly chiral reagents, making it less economically viable and environmentally unfriendly. Additionally, hydroamination of olefins usually demands the addition of excess silane as a hydrogen source, which negatively affects atom economy and introduces safety concerns regarding silane handling. These legacy processes collectively suffer from narrow substrate applicability, difficult amplification operations, and poor alignment with green chemistry principles, creating bottlenecks for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy limitations, the novel nickel-catalyzed method presented in the patent data offers a streamlined, one-step solution that directly alkylates the alpha position of amines using readily available olefins. This approach eliminates the need for pre-synthesizing unstable imine intermediates or introducing additional guiding groups, thereby simplifying the synthetic route and reducing the overall number of unit operations. By utilizing a zero-valent or divalent nickel catalyst in conjunction with affordable monophosphine ligands, the process avoids the reliance on precious metals, leading to substantial cost savings in raw material procurement. The reaction proceeds efficiently in common nonpolar solvents like toluene at moderate temperatures ranging from 80°C to 120°C, ensuring operational safety and energy efficiency. Crucially, the method does not require an external reducing agent, as the hydrogen necessary for the reduction is transferred internally from the amine substrate, enhancing the atom economy and minimizing waste generation. This robust methodology supports a wide range of substrates, including those with electron-donating or withdrawing groups, and has been proven to maintain high yields during scale-up, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Nickel-Catalyzed Reductive Coupling
The core of this technological breakthrough lies in the unique ability of the nickel catalyst to mediate a hydrogen transfer process that couples amine dehydrogenation with olefin reduction in a single catalytic cycle. The reaction initiates with the coordination of the nickel center to the amine substrate, facilitating the activation of the C-H bond at the alpha position through a dehydrogenation step. This generates a reactive nickel-hydride species and an electrophilic imine intermediate in situ, which subsequently undergoes insertion of the olefin substrate. The presence of arylboronic acid additives, such as phenylboronic acid, plays a critical role in modulating the electronic environment of the catalyst and stabilizing key intermediates, thereby significantly improving reaction yields. In cases where yields are initially moderate, the addition of pivalaldehyde serves as a secondary promoter to further drive the reaction forward, ensuring high conversion rates even for sterically hindered substrates. The use of strong bases like potassium tert-butoxide is essential for maintaining the catalytic turnover by regenerating the active nickel species and neutralizing acidic byproducts. This intricate balance of catalyst, ligand, and additive allows for precise control over the reaction pathway, minimizing side reactions and ensuring the formation of the desired alkylamine structure with high selectivity.
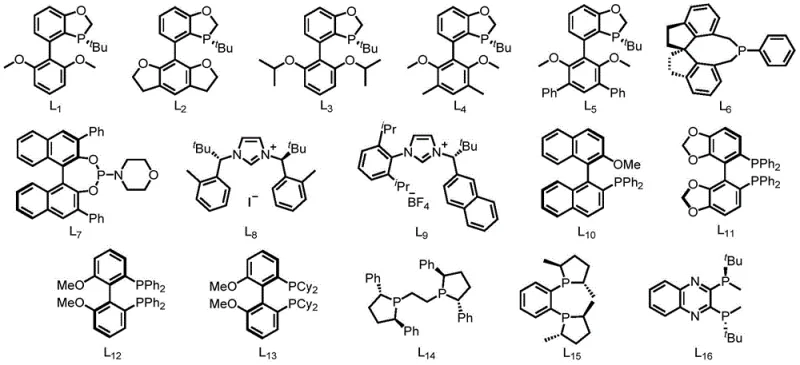
For the synthesis of chiral alkylamines, which are vital for the production of enantiomerically pure drugs, the system employs specialized chiral monophosphine ligands designated as L1 through L16. Among these, ligand L1 has demonstrated superior catalytic performance, enabling the construction of chiral centers with high optical purity. The steric and electronic properties of these ligands dictate the facial selectivity of the olefin insertion step, effectively transferring chirality from the ligand to the product. This capability allows manufacturers to access valuable chiral building blocks without the need for resolution steps or expensive chiral pool starting materials. The mechanistic flexibility of the nickel system ensures that it can accommodate various substitution patterns on both the amine and olefin components, providing a versatile tool for medicinal chemists. By understanding these mechanistic nuances, process engineers can optimize reaction conditions to maximize yield and purity, ensuring that the final product meets the stringent quality specifications required for pharmaceutical applications.
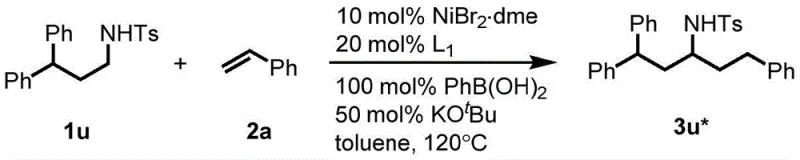
How to Synthesize N-(1,3-Diphenylpropyl)-4-methylbenzenesulfonamide Efficiently
The practical implementation of this nickel-catalyzed protocol is exemplified by the synthesis of N-(1,3-Diphenylpropyl)-4-methylbenzenesulfonamide, a representative alkylamine derivative with significant utility in organic synthesis. The process begins by charging a reaction vessel with the benzylamine starting material and styrene olefin under an inert nitrogen atmosphere to prevent catalyst oxidation. Key to the success of this transformation is the precise addition of the nickel catalyst, tricyclohexylphosphine ligand, and phenylboronic acid additive, which work synergistically to drive the coupling reaction. The mixture is then heated to 120°C in toluene solvent, a condition that provides sufficient thermal energy to overcome the activation barrier for C-H bond cleavage while maintaining solvent stability. After a reaction period of approximately 20 hours, the mixture is cooled, concentrated, and subjected to standard purification techniques to isolate the target compound in high yield. This straightforward procedure highlights the operational simplicity of the method, making it accessible for both laboratory research and pilot plant operations.
- Mix raw material I (benzylamine derivative), raw material II (olefin), metal catalyst (Ni(cod)2), ligand (PCy3), base (KOtBu), and additive (PhB(OH)2) in a solvent like Toluene under nitrogen.
- Stir the reaction mixture at elevated temperatures between 80°C and 120°C for approximately 20 hours to ensure complete conversion via hydrogen transfer.
- Cool the reaction to room temperature, concentrate the mixture, and purify the resulting alkylamine derivative using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this nickel-catalyzed synthesis route offers transformative advantages for procurement managers and supply chain leaders seeking to optimize their manufacturing networks. The most significant benefit is the drastic reduction in raw material costs achieved by replacing expensive noble metal catalysts like ruthenium and rhodium with abundant and inexpensive nickel complexes. This substitution not only lowers the direct cost of goods but also mitigates the supply risk associated with volatile precious metal markets, ensuring greater supply chain reliability and continuity. Furthermore, the elimination of external reducing agents and the use of stable, commercially available starting materials simplify the procurement process, reducing the administrative burden and lead time for sourcing critical reagents. The robustness of the reaction conditions allows for the use of standard reactor equipment without the need for specialized high-pressure hydrogenation apparatus, facilitating easier technology transfer and scale-up. These factors collectively contribute to a more resilient and cost-effective supply chain, enabling companies to respond more agilely to market demands.
- Cost Reduction in Manufacturing: The transition from noble metal catalysts to nickel-based systems results in significant cost savings by removing the need for expensive ruthenium, rhodium, or iridium complexes. Additionally, the avoidance of stoichiometric reducing agents and silane additives reduces the consumption of hazardous and costly reagents, further lowering the overall production expense. The simplified workup procedure, which often requires only concentration and chromatography, minimizes solvent usage and waste disposal costs, contributing to a leaner manufacturing process. By streamlining the synthetic route and reducing the number of steps, manufacturers can achieve higher throughput and lower operational expenditures, directly impacting the bottom line.
- Enhanced Supply Chain Reliability: Utilizing widely available nickel catalysts and common phosphine ligands ensures a stable supply of critical reagents, reducing the risk of production delays caused by material shortages. The method's tolerance for a wide range of substrates means that alternative starting materials can be sourced easily if primary suppliers face disruptions, enhancing the flexibility of the supply chain. Moreover, the ability to scale the reaction to gram levels without loss of yield demonstrates the process's robustness, providing confidence in its ability to meet large-volume production schedules. This reliability is crucial for maintaining consistent delivery timelines to downstream customers and avoiding penalties associated with late shipments.
- Scalability and Environmental Compliance: The process aligns well with green chemistry principles by improving atom economy and reducing the generation of heavy metal waste, which simplifies environmental compliance and waste treatment protocols. The use of common solvents like toluene and the absence of highly toxic silanes make the process safer to operate on a large scale, reducing the regulatory burden and insurance costs associated with hazardous chemical handling. The straightforward scale-up characteristics allow for seamless transition from pilot batches to commercial production volumes, ensuring that capacity can be expanded rapidly to meet growing demand. This scalability, combined with improved environmental performance, positions the technology as a sustainable choice for long-term manufacturing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed alkylamine synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing accurate guidance for process development teams. Understanding these details is essential for evaluating the feasibility of adopting this method for specific project requirements and ensuring successful technology transfer. The information covers aspects ranging from catalyst selection to substrate scope, offering a comprehensive overview of the method's capabilities.
Q: What are the primary advantages of this nickel-catalyzed method over traditional imine reduction?
A: Unlike traditional methods requiring unstable imine substrates and expensive noble metals like ruthenium or rhodium, this nickel-catalyzed approach utilizes stable amine and olefin starting materials directly. It eliminates the need for external reducing agents by leveraging intramolecular hydrogen transfer, significantly simplifying the process and reducing raw material costs.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent data explicitly demonstrates that the reaction maintains high yields even when scaled up to the gram level. The use of commercially available ligands and common solvents like toluene, combined with a simple workup procedure involving concentration and chromatography, supports robust commercial scale-up of complex pharmaceutical intermediates.
Q: Is it possible to achieve chiral alkylamines using this protocol?
A: Absolutely. By substituting the standard monophosphine ligand with specific chiral monophosphine ligands, such as the L1 ligand described in the patent, the system can effectively construct chiral alkylamines with high optical purity, which is critical for active pharmaceutical ingredient (API) synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkylamine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this nickel-catalyzed technology to revolutionize the production of high-purity pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your projects move seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alkylamine derivative delivered meets the highest industry standards. We are committed to leveraging our technical expertise to optimize this novel synthetic route, delivering cost-effective and reliable solutions tailored to your specific needs.
We invite you to collaborate with our technical procurement team to explore how this advanced synthesis method can enhance your supply chain efficiency and reduce manufacturing costs. Please contact us to request a Customized Cost-Saving Analysis specific to your target molecules. Our experts are ready to provide specific COA data and detailed route feasibility assessments to support your decision-making process. Let us be your trusted partner in bringing high-quality alkylamine derivatives to market faster and more efficiently.
