Scalable Manufacturing of Polysubstituted Azatricyclic Azine Derivatives via Base-Metal Catalysis
Scalable Manufacturing of Polysubstituted Azatricyclic Azine Derivatives via Base-Metal Catalysis
The landscape of organic synthesis for complex heterocyclic scaffolds is undergoing a significant transformation, driven by the urgent need for greener, more cost-effective methodologies. A pivotal advancement in this domain is detailed in patent CN108484602B, which discloses a robust preparation method for polysubstituted azatricyclic azine derivatives. These compounds serve as critical building blocks in the construction of bioactive molecules, finding extensive application in pharmaceutical production and natural product synthesis. The disclosed technology leverages a novel cyclization strategy that merges substituted aminopyridines with butynedioic acid esters under remarkably mild conditions. By utilizing air as the oxidant and inexpensive base-metal catalysts, this approach circumvents the stringent requirements of traditional noble-metal catalysis. This report analyzes the technical merits of this innovation, highlighting its potential to redefine supply chain economics for high-value pharmaceutical intermediates.
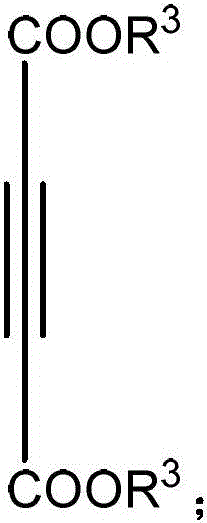
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of azatricyclic frameworks has relied heavily on transition-metal-catalyzed cyclizations involving imidazo[1,2-a]pyridines and disubstituted alkynes or benzynes. While effective in specific contexts, these legacy pathways are fraught with inefficiencies that hinder large-scale adoption. A primary bottleneck is the dependence on precious metal catalysts, such as palladium or rhodium complexes, which not only inflate raw material costs but also introduce significant challenges in residual metal removal—a critical quality attribute for pharmaceutical intermediates. Furthermore, conventional routes often necessitate the pre-synthesis of complex starting materials, adding multiple steps to the overall process flow. The requirement for inert atmospheres and harsh reaction conditions further complicates operational safety and energy consumption. These cumulative factors result in prolonged lead times and reduced overall process mass intensity, making traditional methods less attractive for cost-sensitive commercial manufacturing environments.
The Novel Approach
The methodology outlined in the patent data represents a paradigm shift towards sustainable and economical synthesis. By employing a direct cyclization between readily available substituted aminopyridines and butynedioic acid esters, the process eliminates the need for pre-functionalized precursors. The use of abundant base metals like manganese, iron, or copper drastically reduces catalyst costs while maintaining high catalytic activity. Crucially, the reaction proceeds efficiently under an air atmosphere, removing the capital and operational expenditures associated with nitrogen or argon blanketing systems. The protocol demonstrates exceptional substrate versatility, accommodating a wide array of electronic and steric environments on the pyridine ring. This flexibility allows chemists to access a diverse library of derivatives from a single, unified platform, thereby accelerating lead optimization campaigns in drug discovery while simplifying inventory management for procurement teams.
Mechanistic Insights into Base-Metal Catalyzed Cyclization
The core of this synthetic breakthrough lies in the intricate interplay between the base-metal catalyst, the ligand system, and the oxidant. The reaction initiates with the coordination of the metal center to the alkyne moiety of the butynedioic acid ester, activating it towards nucleophilic attack by the amino group of the pyridine substrate. Unlike precious metal cycles that often rely on oxidative addition/reductive elimination manifolds, this base-metal system likely operates through a radical or single-electron transfer pathway facilitated by the oxidant, such as di-tert-butyl peroxide. The ligand, typically a bidentate nitrogen donor like 2,2'-bipyridine or 1,10-phenanthroline, stabilizes the metal oxidation state and modulates its electrophilicity, ensuring high turnover numbers. This mechanistic pathway is inherently robust, tolerating moisture and oxygen, which contributes to the observed high yields and reproducibility across different substrate classes.
From an impurity control perspective, the specificity of this cyclization is remarkable. The reaction conditions are tuned to favor the formation of the fused tricyclic core over potential side reactions such as polymerization of the alkyne or over-oxidation of the amine. The use of mild temperatures, typically around 70°C, minimizes thermal degradation of sensitive functional groups like halogens or ethers, which are often present in advanced intermediates. Post-reaction workup is straightforward, usually involving simple concentration and chromatographic purification, which suggests that the reaction mixture is relatively clean with minimal byproduct formation. This high level of chemoselectivity is vital for maintaining the purity profiles required by regulatory bodies, reducing the burden on downstream purification processes and enhancing the overall economic viability of the manufacturing route.
How to Synthesize Polysubstituted Azatricyclic Azine Derivatives Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The general procedure involves combining the substituted aminopyridine and the dialkyl butynedioate in a suitable organic solvent such as acetonitrile or toluene. A catalytic amount of a manganese or iron salt is added along with a chelating ligand and a stoichiometric oxidant. The mixture is then heated under open-air conditions, allowing the cyclization to proceed to completion. The simplicity of the setup makes it ideal for both laboratory screening and pilot plant operations. Below is a representative example of the reaction scheme demonstrating the transformation of specific substrates into the target tricyclic scaffold.
- Mix substituted aminopyridine, butynedioic acid ester, base-metal catalyst (e.g., manganese acetate), ligand, oxidant, and organic solvent in a reaction vessel.
- Heat the mixture under an air atmosphere at moderate temperatures (e.g., 70°C) to facilitate the cyclization reaction.
- Upon completion, cool the reaction to room temperature, concentrate the solution, and purify the crude product via column chromatography.
![Reaction scheme for Example 1 showing synthesis of 1-benzyl-7-methyl-2-carbonyl-1,2-dihydroimidazo[5,1,2-cd]indolizine](/insights/img/azatricyclic-azine-synthesis-pharma-supplier-20260303023904-04.png)
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this base-metal catalyzed process offers tangible strategic benefits beyond mere technical novelty. The elimination of expensive precious metals directly translates to a significant reduction in the bill of materials, insulating the supply chain from the volatility of commodity prices for metals like palladium. Moreover, the use of air as the terminal oxidant removes the logistical complexity and cost associated with sourcing and handling high-purity inert gases. The mild reaction conditions imply lower energy consumption for heating and cooling, contributing to a smaller carbon footprint and aligning with corporate sustainability goals. These factors collectively enhance the resilience of the supply chain, ensuring a more stable and predictable flow of critical intermediates.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with abundant base metals like manganese or iron creates a profound impact on the cost structure of the final product. Precious metals not only carry a high unit price but also require specialized recovery processes to meet strict residual metal limits in pharmaceuticals. By avoiding these costly metals entirely, the process eliminates the need for expensive scavenging resins or complex extraction protocols. Furthermore, the high atom economy of the cyclization reaction ensures that a greater proportion of the starting materials end up in the final product, minimizing waste disposal costs. This lean manufacturing approach allows for substantial margin improvement, making the final API or intermediate more competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on commercially available, commodity-grade reagents significantly de-risks the supply chain. Substituted aminopyridines and butynedioic acid esters are widely produced chemicals with multiple global suppliers, preventing single-source bottlenecks. The robustness of the reaction under air atmosphere means that production is less susceptible to disruptions caused by utility failures or gas supply issues. Additionally, the short reaction times reported in the patent data suggest higher throughput capacity for existing reactor trains. This increased velocity allows manufacturers to respond more agilely to fluctuations in demand, reducing inventory holding costs and improving cash flow dynamics for the entire value chain.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this methodology is inherently designed for scale-up. The absence of pyrophoric reagents or high-pressure hydrogenation steps simplifies the engineering controls required for large-scale reactors. The use of common organic solvents facilitates easy recycling and recovery, aligning with green chemistry principles. From a regulatory standpoint, the cleaner reaction profile reduces the burden of validating impurity clearance, accelerating the timeline for technology transfer from R&D to commercial production. This ease of scale-up ensures that the supply of these complex heterocycles can be ramped up rapidly to meet clinical or commercial needs without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: This method utilizes inexpensive base-metal catalysts like manganese or iron instead of precious metals, operates under mild air atmospheres without inert gas protection, and offers a broad substrate scope with high yields and simplified post-treatment.
Q: Can this process be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the reaction conditions are mild (e.g., 70°C) and utilize common solvents like acetonitrile or toluene, making it highly suitable for scale-up from laboratory to multi-ton commercial manufacturing without complex safety hazards.
Q: What types of substituents are tolerated in this cyclization reaction?
A: The protocol demonstrates excellent tolerance for various groups including alkyl, alkenyl, aryl, substituted arenes (such as fluoro, bromo, methoxy, and trifluoromethyl groups), allowing for diverse functionalization of the azatricyclic core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Azatricyclic Azine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this base-metal catalyzed cyclization technology for the pharmaceutical industry. As a premier CDMO partner, we possess the technical expertise to adapt and optimize this pathway for your specific molecular targets. Our facilities are equipped to handle complex organic syntheses with precision, ensuring that every batch meets the rigorous quality standards demanded by global health authorities. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art infrastructure to deliver consistent results. Our commitment to excellence is underpinned by stringent purity specifications and operated by rigorous QC labs that utilize advanced analytical techniques to verify identity and potency.
We invite you to collaborate with us to unlock the full commercial potential of your azatricyclic projects. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By partnering with us, you gain access to a reliable supply chain capable of supporting your growth from early-stage development through to full-scale commercialization. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your next generation of therapeutic candidates.
