Advanced Manganese-Catalyzed Cyclization for High-Purity Azatricyclic Azine Intermediates
Introduction to Next-Generation Azatricyclic Synthesis
The pharmaceutical industry continuously demands efficient routes to complex heterocyclic scaffolds, and patent CN108484602B introduces a transformative method for preparing polysubstituted azatricyclic azine derivatives. These compounds serve as critical building blocks in the development of novel bioactive agents, yet their synthesis has historically been plagued by harsh conditions and low atom economy. This new technology leverages a cost-effective manganese-catalyzed oxidative cyclization strategy that operates under mild thermal conditions and ambient air pressure. By utilizing readily available substituted aminopyridines and dialkyl butynedioates, the process achieves high regioselectivity and yield without the need for noble metals. This represents a significant leap forward for reliable pharmaceutical intermediate supplier networks seeking to optimize their supply chains.
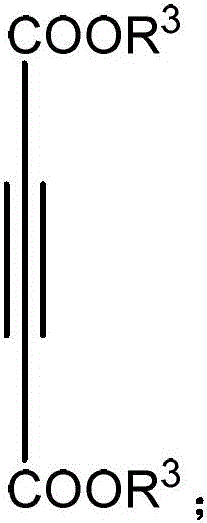
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing the imidazo[5,1,2-cd]indolizine core often rely heavily on transition metal catalysts such as palladium or rhodium, which are not only prohibitively expensive but also pose significant environmental hazards due to heavy metal residue concerns. Furthermore, legacy methods frequently necessitate multi-step sequences involving the pre-functionalization of starting materials, which drastically increases the overall processing time and reduces the final throughput. The requirement for strict inert atmospheres, such as nitrogen or argon shielding, adds another layer of operational complexity and capital expenditure for manufacturing facilities. These factors collectively contribute to higher production costs and longer lead times, creating bottlenecks for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN108484602B utilizes a base-metal manganese catalyst system that functions efficiently in the presence of oxygen, effectively turning a traditional liability into a reactive asset. The reaction proceeds through a direct oxidative annulation between the aminopyridine and the alkyne diester, streamlining the synthesis into a single pot operation. This approach eliminates the need for pre-activated substrates and allows for the direct installation of diverse functional groups at multiple positions on the tricyclic framework. The robustness of this system is evidenced by its compatibility with a wide range of solvents and oxidants, providing process chemists with substantial flexibility.
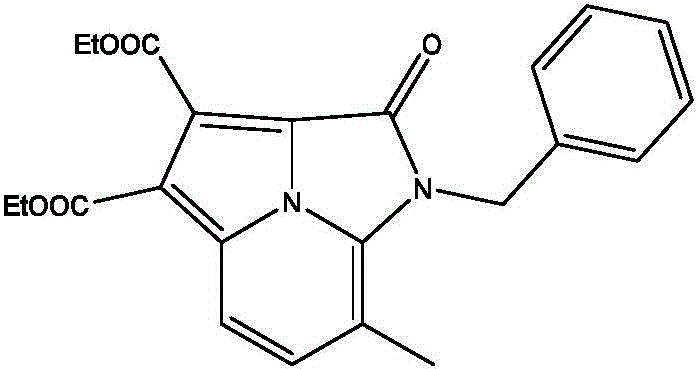
Mechanistic Insights into Manganese-Catalyzed Oxidative Cyclization
The catalytic cycle likely initiates with the coordination of the manganese species to the nitrogen atom of the aminopyridine substrate, facilitating the activation of the adjacent C-H bond. In the presence of oxidants like di-tert-butyl peroxide (DTBP), a radical pathway is proposed where the manganese center mediates the generation of reactive radical intermediates from the alkyne component. This radical species then undergoes an intramolecular addition to the activated pyridine ring, followed by oxidative aromatization to restore the stable tricyclic system. The choice of ligands, such as 2,2'-bipyridine or phenanthroline derivatives, plays a crucial role in stabilizing the active manganese oxidation state and preventing catalyst deactivation. Understanding this mechanistic nuance is vital for R&D directors aiming to troubleshoot potential side reactions or optimize reaction kinetics for specific substrate classes.
Impurity control in this system is inherently superior due to the high chemoselectivity of the manganese catalyst towards the specific C-H activation site. Unlike non-selective radical processes that might lead to polymerization of the alkyne or over-oxidation of the amine, this coordinated cycle directs the reaction exclusively towards the desired cyclization. The mild reaction temperature of approximately 70°C further suppresses thermal degradation pathways that are common in high-energy processes. Consequently, the crude reaction mixtures are cleaner, which significantly reduces the burden on downstream purification units like column chromatography or recrystallization. This purity profile is essential for meeting the stringent specifications required for high-purity pharmaceutical intermediates intended for clinical applications.
How to Synthesize Polysubstituted Azatricyclic Azine Derivatives Efficiently
To implement this synthesis effectively, process engineers should focus on the precise stoichiometric balance between the aminopyridine and the butynedioate ester, typically maintaining a molar ratio between 1:2 and 1:5 to drive the reaction to completion. The selection of the solvent is also critical, with polar aprotic solvents like acetonitrile or dimethyl sulfoxide showing optimal performance in dissolving both the organic substrates and the inorganic catalyst salts. Operators must ensure adequate oxygen mass transfer by using open vessels or controlled air sparging to sustain the oxidative cycle without creating safety hazards. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility across different batch sizes.
- Mix substituted aminopyridine, dialkyl butynedioate, manganese catalyst (e.g., Mn(OAc)2·2H2O), ligand (e.g., 2,2'-bipyridine), oxidant (e.g., DTBP), and organic solvent in a reaction vessel.
- Heat the mixture to approximately 70°C under an air atmosphere and maintain stirring for about 16 hours to facilitate the cyclization reaction.
- Cool the reaction mixture to room temperature, concentrate the solution, and purify the crude product via column chromatography to obtain the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift from precious metal catalysts to abundant manganese salts offers a dramatic reduction in raw material expenditure, directly impacting the cost of goods sold. The elimination of inert gas requirements simplifies the reactor setup, allowing for the utilization of standard glass-lined or stainless steel equipment without specialized modifications for air-sensitive chemistry. This operational simplicity translates to faster turnaround times between batches and increased overall equipment effectiveness (OEE) for manufacturing plants. Furthermore, the use of green oxidants and the generation of less toxic waste streams align with increasingly strict environmental regulations, reducing the costs associated with waste treatment and compliance auditing.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or rhodium catalysts with inexpensive manganese acetate drastically lowers the catalyst cost per kilogram of product. Additionally, the simplified workup procedure reduces solvent consumption and labor hours required for purification, leading to substantial overall cost savings in API intermediate manufacturing.
- Enhanced Supply Chain Reliability: Since the starting materials such as substituted aminopyridines and dialkyl acetylenedicarboxylates are commodity chemicals available from multiple global vendors, the risk of supply chain disruption is minimized. The robustness of the reaction conditions ensures consistent quality output regardless of minor fluctuations in ambient manufacturing conditions, securing a steady flow of materials for downstream drug production.
- Scalability and Environmental Compliance: The reaction exhibits excellent scalability potential due to its exothermic profile being manageable at 70°C, avoiding the need for cryogenic cooling or high-pressure reactors. The green nature of the process, characterized by the use of air as an oxidant and the absence of heavy metal contaminants, facilitates easier regulatory approval and supports sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this manganese-catalyzed protocol. These insights are derived directly from the experimental data and scope analysis presented in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances helps in making informed decisions about process integration and resource allocation.
Q: What are the advantages of using manganese catalysts over precious metals for this synthesis?
A: Using inexpensive manganese catalysts significantly reduces raw material costs compared to traditional palladium or rhodium systems. Additionally, manganese is more environmentally benign, aligning with green chemistry principles and simplifying waste disposal protocols.
Q: Does this method require inert gas protection?
A: No, the process operates effectively under an air atmosphere. This eliminates the need for expensive nitrogen or argon purging systems, reducing equipment complexity and operational overhead for large-scale production.
Q: What is the substrate scope of this cyclization reaction?
A: The method demonstrates broad substrate tolerance, accommodating various substituents on the aminopyridine ring including alkyl, alkenyl, aryl, and halogen groups, allowing for the synthesis of diverse derivative libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Azatricyclic Azine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic methodologies like the one described in CN108484602B to enhance our portfolio of fine chemical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of azatricyclic azine derivatives meets the exacting standards required by top-tier pharmaceutical companies globally.
We invite you to collaborate with us to leverage this advanced technology for your specific drug development programs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can accelerate your project timelines while reducing overall expenditure.
