Scalable Green Synthesis of Febuxostat Intermediates for Commercial API Production
The pharmaceutical industry's relentless pursuit of safer, more efficient manufacturing processes is exemplified by the technological breakthroughs detailed in Chinese Patent CN101665471B. This patent discloses a novel preparation method for 2-(3-formyl-4-hydroxyphenyl)-4-methyl-thiazole-5-carboxylic acid ethyl ester, a pivotal intermediate in the synthesis of Febuxostat, a potent non-purine xanthine oxidase inhibitor used for treating hyperuricemia and gout. As global demand for gout medications surges, the ability to produce high-purity intermediates without relying on hazardous reagents becomes a critical competitive advantage for any reliable pharmaceutical intermediate supplier. The disclosed methodology fundamentally shifts the paradigm from toxic, corrosive chemistries to a greener, scalable approach that utilizes low-toxicity paraformaldehyde in an acetonitrile solvent system. This transition not only addresses stringent environmental regulations but also resolves significant bottlenecks related to equipment corrosion and operator safety that have historically plagued the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Febuxostat intermediates has been fraught with significant safety and operational hazards, as illustrated by earlier patent literature. For instance, the route described in US Patent US5614520 relies heavily on the use of potassium cyanide and cuprous cyanide, reagents that pose extreme toxicity risks to personnel and require elaborate waste treatment protocols.
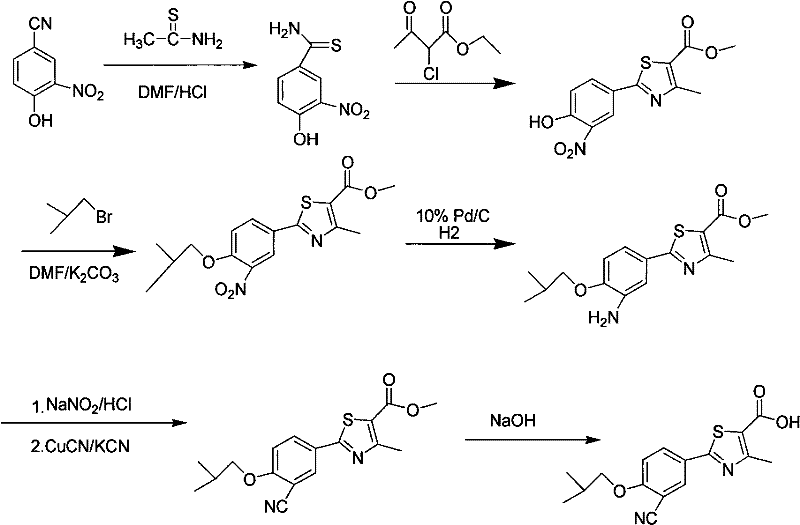
Furthermore, alternative industrial routes, such as those found in Japanese Patent JP1994329647, utilize trifluoroacetic acid (TFA) and urotropine for the critical formylation step. While effective chemically, TFA is notoriously corrosive to standard stainless steel reactors, often necessitating the use of expensive lined vessels or Hastelloy equipment, thereby driving up capital expenditure. Additionally, the use of hydrogen sulfide gas in other variations, as noted in US20050027128, introduces severe odor issues and environmental pollution concerns that are increasingly unacceptable in modern manufacturing hubs. These conventional methods collectively represent a high-risk operational profile that complicates supply chain reliability and inflates production costs through specialized safety measures and equipment maintenance.
The Novel Approach
In stark contrast to these legacy methods, the process outlined in CN101665471B introduces a robust and environmentally benign synthetic pathway. The core innovation lies in the formylation step, where the corrosive TFA and flammable urotropine are replaced by a system utilizing paraformaldehyde and anhydrous magnesium chloride in acetonitrile. This modification eliminates the aggressive acidic environment that degrades reactor integrity, allowing for smoother processing in standard equipment. The visual representation of this improved pathway highlights the streamlined conversion from the hydroxy-thiazole precursor to the formylated product.
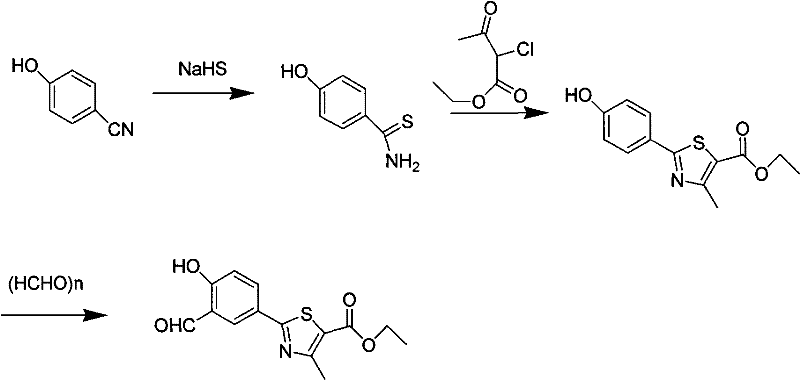
Moreover, the upstream synthesis of the thioamide precursor avoids the generation of hydrogen sulfide gas by employing sodium hydrosulfide in the presence of magnesium chloride hexahydrate. This closed-system approach effectively contains sulfur species, mitigating odor complaints and reducing the burden on scrubber systems. By integrating these safer reagents, the novel approach achieves a formylation yield exceeding 95% and a total synthesis yield of over 72%, demonstrating that safety improvements do not come at the expense of efficiency. This balance makes the process highly attractive for cost reduction in API manufacturing, as it simplifies downstream purification and reduces the frequency of equipment replacement.
Mechanistic Insights into Magnesium-Catalyzed Formylation
The success of this novel route hinges on the precise mechanistic interplay between the Lewis acid catalyst and the formylating agent in a polar aprotic solvent. In the critical formylation step, anhydrous magnesium chloride acts as a Lewis acid to activate the paraformaldehyde, generating an electrophilic species capable of attacking the electron-rich aromatic ring of the thiazole substrate. The use of acetonitrile as the solvent is particularly strategic; it provides excellent solubility for both the organic substrate and the inorganic catalyst while maintaining a thermal profile that supports reflux conditions without decomposing the sensitive aldehyde product. Triethylamine is subsequently introduced to facilitate the rearomatization of the intermediate sigma complex, ensuring the stable incorporation of the formyl group at the ortho-position relative to the hydroxyl group. This specific regioselectivity is crucial for the biological activity of the final Febuxostat molecule, and the mild conditions prevent the formation of poly-formylated byproducts that often plague harsher acid-catalyzed reactions.
From an impurity control perspective, the avoidance of strong mineral acids and halogenated solvents significantly simplifies the impurity profile of the crude product. Traditional methods using TFA often lead to halogenated impurities or ester hydrolysis side reactions due to the acidic environment, requiring rigorous chromatographic purification that lowers overall yield. In this magnesium-catalyzed system, the primary impurities are typically unreacted starting materials or minor oligomers of formaldehyde, which are easily removed via aqueous workup and recrystallization. The patent data indicates that simple filtration and washing with water are sufficient to isolate the product with high purity, suggesting that the reaction generates minimal tarry byproducts. This clean reaction profile is essential for meeting the stringent purity specifications required by regulatory bodies for pharmaceutical intermediates, reducing the analytical burden on quality control laboratories and accelerating batch release times.
How to Synthesize Ethyl 2-(3-formyl-4-hydroxyphenyl)-4-methyl-thiazole-5-carboxylate Efficiently
The practical implementation of this synthesis involves a sequential three-step protocol that begins with the safe generation of the thioamide building block. Operators first react p-cyanophenol with sodium hydrosulfide in DMF, catalyzed by magnesium chloride hexahydrate, to obtain p-hydroxythiobenzamide with a reported yield of 95%. This intermediate is then cyclized with ethyl 2-chloroacetoacetate in ethanol to form the thiazole ring, followed by the critical formylation step using paraformaldehyde in acetonitrile. The detailed standardized operating procedures, including specific temperature ramps, addition rates, and quenching protocols necessary to replicate the >95% yield in the final step, are critical for technology transfer.
- Synthesize p-hydroxythiobenzamide by reacting p-cyanophenol with sodium hydrosulfide in DMF using magnesium chloride hexahydrate as a catalyst.
- Cyclize the thioamide with ethyl 2-chloroacetoacetate in ethanol under reflux to form the thiazole ring structure.
- Perform formylation using paraformaldehyde in acetonitrile with anhydrous magnesium chloride and triethylamine to introduce the aldehyde group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates directly into tangible operational efficiencies and risk mitigation. The elimination of highly toxic cyanides and corrosive trifluoroacetic acid removes several high-cost variables from the manufacturing equation. By shifting to reagents like paraformaldehyde and sodium hydrosulfide, the facility reduces its dependency on tightly controlled substances that require special licensing, storage, and transportation logistics. This simplification of the raw material portfolio enhances supply chain resilience, as the sourcing of commodity chemicals is generally more stable and less prone to regulatory disruption than specialty toxic reagents. Furthermore, the mild reaction conditions reduce the energy load associated with heating and cooling cycles, contributing to a lower carbon footprint and reduced utility costs per kilogram of product produced.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the extension of equipment lifespan and the reduction of maintenance downtime. By avoiding trifluoroacetic acid, manufacturers eliminate the rapid corrosion of reactor walls and agitators, which traditionally necessitates frequent lining repairs or the purchase of exotic alloy vessels. This qualitative shift allows for the utilization of standard glass-lined or stainless steel reactors, drastically lowering capital depreciation costs. Additionally, the high yield of the formylation step (>95%) minimizes the loss of valuable starting materials, ensuring that the cost of goods sold (COGS) remains competitive even at large production scales. The simplified workup procedure, which avoids complex extractions with halogenated solvents, further reduces solvent recovery costs and waste disposal fees.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted supply to downstream API manufacturers. The use of stable, non-volatile reagents like paraformaldehyde reduces the risk of shipment delays caused by hazardous material transport restrictions. Moreover, the process tolerance to minor variations in reaction parameters means that production schedules are less likely to be disrupted by failed batches or out-of-specification results. This reliability allows supply chain planners to operate with leaner inventory buffers, knowing that the manufacturing process is predictable and scalable from pilot plant to multi-ton commercial production without significant re-optimization.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the ability to demonstrate a "green" manufacturing process is a distinct market advantage. This method avoids the generation of hydrogen sulfide gas and cyanide-containing wastewater, two of the most challenging effluent streams to treat in the fine chemical industry. The aqueous waste streams generated are significantly easier to neutralize and process, reducing the load on onsite wastewater treatment plants. This compliance ease facilitates faster permitting for capacity expansion and reduces the risk of regulatory fines or shutdowns. Consequently, the process is inherently scalable, supporting the transition from 100 kgs to 100 MT/annual commercial production with minimal environmental impact, aligning perfectly with the sustainability goals of major pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific technical advantages and operational data presented in the patent documentation, providing clarity for potential partners evaluating this technology for their supply chains.
Q: How does this new process improve safety compared to traditional cyanide routes?
A: Unlike the US5614520 route which utilizes highly toxic potassium cyanide and cuprous cyanide, this patented method employs sodium hydrosulfide and paraformaldehyde, significantly reducing operator exposure risks and hazardous waste disposal costs.
Q: What are the equipment advantages of avoiding trifluoroacetic acid (TFA)?
A: Traditional methods using TFA cause severe corrosion to reactor vessels, necessitating expensive Hastelloy equipment. This new acetonitrile-based process is non-corrosive, allowing for the use of standard stainless steel reactors and extending equipment lifespan.
Q: What is the reported yield for the critical formylation step?
A: According to the patent data, the formylation reaction using paraformaldehyde in acetonitrile achieves a yield of over 95%, contributing to a robust total synthesis yield exceeding 72%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Febuxostat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the technical elegance of a patent must be matched by manufacturing excellence to deliver real value. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this green synthesis route are fully realized at an industrial level. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of Febuxostat intermediate we produce. We understand that consistency is key in the pharmaceutical supply chain, and our process engineers are dedicated to maintaining the high yields and safety standards outlined in the patent data.
We invite forward-thinking procurement leaders to collaborate with us to optimize their supply chains. By leveraging this advanced manufacturing technology, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and compliant chemical solution available.
