Optimizing Febuxostat Intermediate Production Through Novel Mixed Acid Catalysis Technology
Optimizing Febuxostat Intermediate Production Through Novel Mixed Acid Catalysis Technology
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical active pharmaceutical ingredient (API) precursors, particularly for treatments addressing chronic conditions like gout. Patent CN102002017B introduces a significant advancement in the preparation of a key febuxostat intermediate, specifically 2-[3-formyl-4-hydroxyphenyl]-4-methylthiazol-5-carboxylic acid ethyl ester. This compound serves as a pivotal building block in the synthesis of febuxostat, a potent non-purine selective xanthine oxidase inhibitor used to manage hyperuricemia. The disclosed technology addresses longstanding challenges in the Duff reaction formylation process, traditionally plagued by either low yields or prohibitive solvent costs. By leveraging a novel mixed acid solvent system, this method achieves a delicate balance between economic feasibility and chemical efficiency, making it a highly attractive option for large-scale manufacturing.
![Chemical structure of the target febuxostat intermediate 2-[3-formyl-4-hydroxyphenyl]-4-methylthiazol-5-carboxylic acid ethyl ester](/insights/img/febuxostat-intermediate-synthesis-pharma-supplier-20260315110307-04.png)
The structural integrity and purity of this intermediate are paramount for the downstream synthesis of the final API. As illustrated in the molecular diagram, the presence of the aldehyde group at the 3-position and the hydroxyl group at the 4-position on the phenyl ring requires precise regioselective control during synthesis. Conventional methods often struggle to introduce the formyl group without affecting other sensitive functionalities or generating complex impurity profiles that are difficult to remove. The innovation presented in this patent provides a streamlined pathway that not only enhances the overall yield but also simplifies the purification process, thereby ensuring a reliable supply of high-purity pharmaceutical intermediates for global drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this specific heterocyclic aldehyde has relied heavily on the Duff reaction, which typically employs hexamethylenetetramine as the formylating agent. However, the choice of solvent in traditional protocols presents a significant dichotomy for process chemists. On one hand, the use of polyphosphoric acid (PPA) alone is economically attractive due to its low cost; however, it frequently results in lower product yields, often hovering around 70%, and generates a substantial amount of tarry impurities that complicate downstream processing. On the other hand, utilizing trifluoroacetic acid (TFA) as the solvent dramatically improves the yield to approximately 95%, but this comes at a steep financial premium. TFA is not only expensive to purchase in bulk but also poses significant challenges regarding recovery and recycling, leading to increased environmental waste and higher operational expenditures. These limitations create a bottleneck for manufacturers aiming to produce febuxostat intermediates in a cost-effective and environmentally sustainable manner.
The Novel Approach
The breakthrough detailed in patent CN102002017B lies in the strategic formulation of a mixed acid reaction solvent. Instead of relying on a single acidic medium, this method combines polyphosphoric acid with a secondary strong acid, such as concentrated hydrochloric acid, sulfuric acid, or even a reduced proportion of trifluoroacetic acid. This synergistic solvent system creates an optimal reaction environment that mimics the high reactivity of pure TFA while retaining the economic benefits of PPA. Experimental data from the patent demonstrates that this approach consistently delivers yields ranging from 90% to 93%, effectively bridging the gap between cost and performance. Furthermore, the mixed acid system significantly reduces the viscosity and complexity of the reaction mixture, facilitating easier workup procedures and minimizing the formation of intractable byproducts.
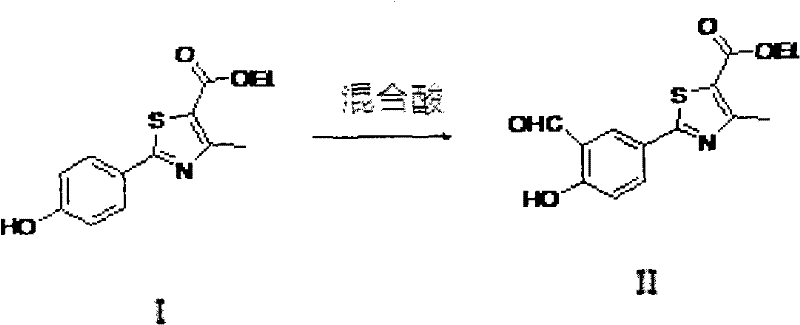
As depicted in the reaction scheme, the transformation involves the conversion of the phenolic precursor into the corresponding aldehyde through an electrophilic aromatic substitution mechanism mediated by the iminium ion derived from urotropine. The use of mixed acids likely enhances the generation of the active electrophilic species while maintaining the solubility of the organic substrate throughout the reaction course. This method allows for reaction temperatures between 40°C and 120°C and reaction times spanning from 1 to 36 hours, offering considerable flexibility for process optimization. The ability to tune the acid ratio, typically between 4:1 and 1:1, empowers manufacturers to customize the process based on their specific equipment capabilities and raw material availability, ensuring a robust and adaptable manufacturing protocol.
Mechanistic Insights into Mixed Acid-Catalyzed Duff Formylation
The core of this synthetic advancement relies on the nuanced interplay between the polyphosphoric acid matrix and the added mineral or organic acid. In the Duff reaction mechanism, the formation of the reactive iminium salt from hexamethylenetetramine is the rate-determining step, which is heavily influenced by the acidity and polarity of the medium. Polyphosphoric acid provides a highly polar, viscous environment that stabilizes charged intermediates, but its sheer viscosity can hinder mass transfer and lead to localized overheating or incomplete reactions. By introducing a secondary acid like hydrochloric acid or sulfuric acid, the overall viscosity of the system is modulated, improving the diffusion of reactants and ensuring homogeneous mixing. This enhanced mass transfer is critical for achieving the high conversion rates observed in the patent examples, where yields consistently exceed 90% under optimized conditions.
Furthermore, the mixed acid system plays a crucial role in impurity control and regioselectivity. The phenolic substrate contains multiple potential sites for electrophilic attack, but the ortho-position relative to the hydroxyl group is the desired target for formylation. The specific acidity profile of the mixed solvent likely promotes the formation of a hydrogen-bonded complex between the phenolic hydroxyl group and the acid, which directs the incoming electrophile to the correct position on the aromatic ring. This directed ortho-metalation-like effect minimizes the formation of meta- or para-substituted byproducts, which are common contaminants in less controlled Duff reactions. Consequently, the crude product obtained from this method possesses a cleaner impurity profile, reducing the burden on subsequent crystallization or chromatography steps and ultimately enhancing the overall process efficiency and product quality.
How to Synthesize 2-[3-formyl-4-hydroxyphenyl]-4-methylthiazol-5-carboxylic acid ethyl ester Efficiently
Implementing this novel synthetic route requires careful attention to the preparation of the mixed acid solvent and the control of reaction parameters. The process begins with the dissolution of the starting thiazole derivative in the pre-mixed acid solution, ensuring complete solubilization before the addition of the formylating agent. The addition of urotropine should be managed to prevent excessive exothermicity, although the reaction is generally conducted under heated conditions to drive the equilibrium towards the product. Following the reaction period, the mixture is typically cooled and poured into ice water to precipitate the product, a standard quenching procedure for acid-catalyzed reactions that allows for easy filtration and isolation. The detailed standardized synthesis steps see the guide below.
- Dissolve the starting material, 2-[4-hydroxyphenyl]-4-methylthiazol-5-carboxylic acid ethyl ester, into a prepared mixed acid solvent system consisting of polyphosphoric acid and a secondary strong acid.
- Introduce hexamethylenetetramine (urotropine) to the reaction mixture, ensuring the molar ratio is between 0.5 to 10 times that of the substrate to facilitate formylation.
- Heat the reaction mixture to a temperature range of 40-120°C and maintain stirring for 1 to 36 hours to complete the Duff reaction, followed by standard aqueous workup to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this mixed acid methodology represents a strategic opportunity to optimize the cost structure of febuxostat production. The primary economic driver is the substantial reduction in solvent costs. By replacing expensive, pure trifluoroacetic acid with a blend dominated by low-cost polyphosphoric acid and commodity acids like hydrochloric or sulfuric acid, the raw material expenditure per kilogram of intermediate is drastically lowered. Additionally, the simplified workup procedure reduces the consumption of water and energy required for solvent recovery and waste treatment, further contributing to the overall cost efficiency. This economic advantage is compounded by the high yield, which maximizes the output from every batch of starting material, effectively lowering the cost of goods sold (COGS) for the final API.
- Cost Reduction in Manufacturing: The shift from pure TFA to a mixed acid system eliminates the need for expensive solvent recovery units dedicated solely to trifluoroacetic acid. Since PPA and mineral acids are significantly cheaper and easier to handle, the operational overhead is reduced. The high yield of over 90% ensures that valuable starting materials are not wasted, directly translating to improved margin potential for the manufacturing facility.
- Enhanced Supply Chain Reliability: The reagents required for this process, including polyphosphoric acid, hydrochloric acid, and urotropine, are commodity chemicals with stable and abundant global supply chains. Unlike specialized catalysts or exotic solvents that may face supply disruptions, these materials are readily available from multiple vendors. This diversity in sourcing mitigates the risk of production delays caused by raw material shortages, ensuring a consistent and reliable flow of intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The reaction conditions are mild, operating at temperatures up to 120°C, which are easily achievable in standard glass-lined or stainless steel reactors without the need for specialized high-pressure equipment. The reduction in hazardous waste generation, particularly the avoidance of large volumes of fluorinated waste associated with pure TFA processes, simplifies environmental compliance and waste disposal logistics. This makes the process highly scalable from pilot plant to commercial production volumes while adhering to increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows.
Q: What are the advantages of using a mixed acid solvent system over pure trifluoroacetic acid?
A: While pure trifluoroacetic acid offers high yields, it is expensive and difficult to recover. The mixed acid system (polyphosphoric acid combined with acids like HCl or H2SO4) maintains high yields (up to 93%) while significantly reducing raw material costs and simplifying post-reaction treatment.
Q: What is the optimal temperature range for this formylation reaction?
A: The patent specifies a broad operational window between 40°C and 120°C. Specific embodiments demonstrate successful outcomes at 60°C, 80°C, 90°C, and 100°C, allowing flexibility based on available heating infrastructure and desired reaction kinetics.
Q: How does this method impact the purity of the final intermediate?
A: By optimizing the acidity and solvation properties through the mixed acid approach, the reaction minimizes the formation of polymeric byproducts often seen with pure polyphosphoric acid, resulting in a cleaner crude product that requires less rigorous purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Febuxostat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive landscape of pharmaceutical manufacturing. Our team of expert chemists has thoroughly analyzed the technology described in CN102002017B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art facilities capable of handling corrosive acid systems safely and efficiently, ensuring that the transition from laboratory scale to industrial production is seamless. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs that guarantee every batch of febuxostat intermediate meets the highest international standards.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this cost-effective synthesis technology. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this optimized process can enhance your supply chain resilience and profitability. Let us help you engineer a more efficient future for your gout medication production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
