Advanced Palladium-Catalyzed Synthesis of Aryl Acetamides for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously demand efficient, scalable, and safe methodologies for constructing amide bonds, a ubiquitous motif in bioactive molecules. Patent CN111978194B introduces a transformative approach to synthesizing aryl acetamide compounds, addressing long-standing challenges in transition metal-catalyzed carbonylation. This technology leverages a palladium-catalyzed system that utilizes benzyl formate not merely as a solvent or additive, but as a dual-function reagent serving as both a carbon monoxide source and a reactant. By employing readily available tertiary amines as the nitrogen source, this method bypasses the limitations of traditional amidation which often relies on pre-functionalized acids or hazardous gaseous reagents. The significance of this development lies in its operational simplicity and robust substrate tolerance, offering a reliable aryl acetamide supplier pathway for complex molecule synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl acetamides has relied heavily on the direct amidation of phenylacetic acid derivatives, a process that frequently suffers from poor atom economy and requires harsh activation conditions. Alternatively, transition metal-catalyzed carbonylation has emerged as a powerful tool; however, conventional protocols typically utilize primary or secondary amines, leaving the activation of tertiary amines largely unexplored due to the high energy barrier associated with C-N bond cleavage. Furthermore, existing methods for tertiary amine carbonylation often necessitate the use of excessive oxidants or dangerous mixtures of carbon monoxide and oxygen, posing significant safety risks and complicating the purification process. These factors collectively hinder the cost reduction in pharmaceutical intermediates manufacturing, as they increase both the raw material costs and the safety infrastructure requirements for production facilities.
The Novel Approach
In stark contrast to these legacy methods, the disclosed invention utilizes a sophisticated palladium catalytic system comprising palladium acetate and the bidentate ligand DPEphos to facilitate the reaction under mild yet effective conditions. The use of benzyl formate as a liquid CO surrogate eliminates the need for high-pressure gas cylinders, drastically improving the safety profile of the operation. Crucially, the system achieves the cleavage of the tertiary amine C-N bond without the addition of external oxidants, a feat that streamlines the reaction mixture and minimizes byproduct formation. This novel strategy allows for the high-efficiency synthesis of diverse aryl acetamide structures, making it an ideal candidate for the commercial scale-up of complex polymer additives and fine chemical intermediates.
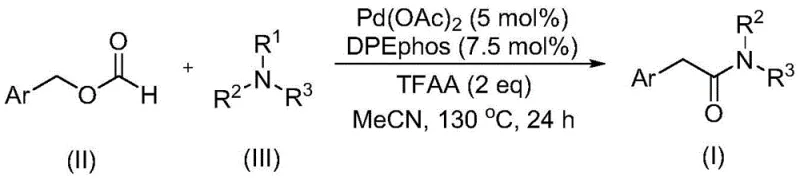
Mechanistic Insights into Pd-Catalyzed Carbonylative C-N Cleavage
The core of this technological advancement lies in the unique mechanistic pathway enabled by the Pd(OAc)2/DPEphos catalyst system. The reaction initiates with the activation of the benzyl formate, which undergoes decarbonylation to generate the active palladium-carbonyl species in situ, effectively serving as a controlled release mechanism for carbon monoxide. Simultaneously, the catalyst facilitates the coordination and subsequent activation of the tertiary amine. Unlike traditional pathways that might require stoichiometric oxidants to drive the reaction forward, this system leverages the intrinsic reactivity of the trifluoroacetic anhydride additive to promote the cleavage of the carbon-nitrogen bond in the tertiary amine. This results in the formation of the desired amide bond with high selectivity, preserving sensitive functional groups on the aromatic ring that might otherwise be compromised under oxidative conditions.
From an impurity control perspective, the absence of strong external oxidants significantly reduces the formation of over-oxidized byproducts, which are common pitfalls in amine functionalization. The reaction conditions, specifically the use of acetonitrile as a polar aprotic solvent at 130°C, ensure that the starting materials are fully dissolved and that the kinetic energy is sufficient to overcome the activation barrier for C-N bond scission. The broad tolerance for substituents such as halogens, cyano groups, and trifluoromethyl groups indicates that the catalytic cycle is robust against electronic variations on the aryl ring. This level of precision is critical for producing high-purity OLED material precursors or pharmaceutical intermediates where trace impurities can compromise biological activity or material performance.
How to Synthesize Aryl Acetamide Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves a straightforward one-pot procedure where all reagents, including the palladium catalyst, ligand, trifluoroacetic anhydride, benzyl formate, and the specific tertiary amine, are combined in an organic solvent. The mixture is then heated in a sealed vessel to maintain the necessary pressure and temperature for the duration of the reaction. Detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and workup procedures required to achieve optimal yields comparable to the patent examples.
- Combine palladium acetate catalyst, DPEphos ligand, trifluoroacetic anhydride, benzyl formate, and tertiary amine in an organic solvent such as acetonitrile.
- Heat the reaction mixture to 130°C in a sealed tube and maintain stirring for approximately 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target aryl acetamide compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits beyond mere chemical efficiency. The reliance on benzyl formate and commercially available tertiary amines as starting materials ensures a stable and diversified supply base, reducing the risk of bottlenecks associated with specialized or hazardous reagents. Furthermore, the elimination of high-pressure carbon monoxide gas removes the need for expensive specialized reactors and rigorous safety monitoring systems, leading to significant capital expenditure savings during facility setup and operation. The simplified post-treatment process, involving basic filtration and chromatography, reduces the time and solvent consumption typically associated with complex workups, thereby enhancing overall throughput.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive and widely available raw materials such as benzyl formate and formic acid derivatives. By avoiding the use of stoichiometric oxidants and hazardous gas feeds, the process inherently lowers the cost of goods sold through reduced material waste and lower safety compliance costs. The high reaction efficiency and yield reported in the patent data suggest that raw material utilization is maximized, minimizing the financial impact of unreacted starting materials. Additionally, the simplified purification requirements reduce the consumption of silica gel and elution solvents, contributing to further operational cost savings.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical factor for long-term production planning, and this method excels by utilizing commodity chemicals that are less susceptible to market volatility compared to exotic catalysts or gases. The robustness of the reaction across a wide range of substrates means that a single manufacturing line can be adapted to produce various derivatives without extensive retooling or process re-validation. This flexibility allows for rapid response to changing market demands and ensures continuity of supply even if specific precursor availability fluctuates. The ability to synthesize diverse aryl acetamides from a common platform enhances the resilience of the supply chain against disruptions.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mass transfer, but the homogeneous nature of this reaction in acetonitrile facilitates smooth scale-up from gram to kilogram scales. The absence of explosive gas mixtures significantly lowers the environmental and safety risks, simplifying the permitting process for new production lines. Moreover, the reduced generation of hazardous waste streams aligns with increasingly stringent global environmental regulations, supporting sustainable manufacturing practices. This green chemistry profile not only mitigates regulatory risk but also enhances the brand value of the final product in markets that prioritize sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction scope, safety, and operational parameters. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this method into their existing synthetic workflows.
Q: How does this method improve safety compared to traditional carbonylation?
A: This protocol utilizes benzyl formate as a safe, liquid carbon monoxide surrogate, eliminating the need for handling hazardous high-pressure CO gas or explosive CO/O2 mixtures often required in conventional oxidative carbonylation processes.
Q: Can tertiary amines be used directly as the nitrogen source?
A: Yes, a key innovation of this technology is the ability to activate and cleave the C-N bond of tertiary amines directly without requiring additional external oxidants, which simplifies the reaction system and reduces waste.
Q: What is the substrate scope for the aryl group?
A: The method demonstrates excellent functional group tolerance, successfully accommodating various substituted aryl groups including those with alkyl, alkoxy, trifluoromethyl, cyano, and halogen substituents at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Acetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and material science applications. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to bulk manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of aryl acetamide intermediate delivered meets the highest industry standards for quality and consistency.
We invite you to collaborate with our technical team to explore how this innovative palladium-catalyzed route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits tailored to your volume needs. We encourage potential partners to contact our technical procurement team to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
