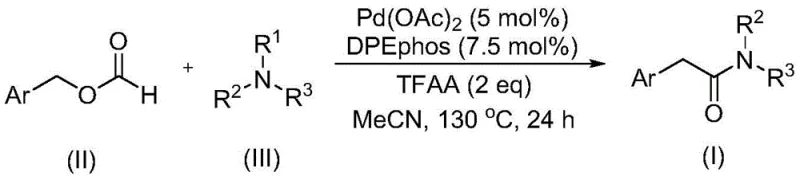
Advanced Palladium-Catalyzed Synthesis of Aryl Acetamides for Commercial Scale-Up
The landscape of amide synthesis is undergoing a significant transformation driven by the urgent need for safer, more efficient, and scalable methodologies in the production of high-value pharmaceutical intermediates. A pivotal advancement in this domain is detailed in patent CN111978194B, which discloses a robust preparation method for aryl acetamide compounds. This technology leverages a sophisticated palladium-catalyzed carbonylation strategy that fundamentally alters the traditional approach to constructing the amide bond. By utilizing benzyl formate as a dual-function reagent—acting simultaneously as a carbon monoxide source and a reactant—the process circumvents the inherent dangers associated with handling gaseous carbon monoxide. Furthermore, the method achieves the challenging activation and cleavage of tertiary amine C-N bonds without the necessity for external oxidants, a feat that has historically plagued synthetic chemists. This innovation not only streamlines the synthetic route but also opens new avenues for the design of complex molecular architectures required in modern drug discovery.
For R&D directors and process chemists, the implications of this patent are profound. It offers a reliable alternative to classical amidation reactions that often rely on pre-functionalized carboxylic acids or hazardous reagents. The ability to generate aryl acetamides directly from readily available benzyl formates and tertiary amines represents a paradigm shift in retrosynthetic planning. As we delve deeper into the technical specifics, it becomes clear that this methodology is not merely an academic curiosity but a viable industrial process capable of delivering high-purity products with exceptional efficiency. The following analysis explores the mechanistic nuances, operational parameters, and commercial viability of this cutting-edge synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl acetamide compounds has relied heavily on the direct amidation of phenylacetic acid and its derivatives. While conceptually straightforward, this classical approach is fraught with significant limitations that hinder its application in large-scale manufacturing. The activation of carboxylic acids typically requires stoichiometric amounts of coupling reagents, which generates substantial chemical waste and increases the overall cost of goods. Moreover, when transition metal-catalyzed carbonylation is employed as an alternative strategy, it predominantly utilizes primary and secondary amines as nitrogen sources. The activation of tertiary amines, which are ubiquitous in bioactive molecules, remains a formidable challenge due to the strength and steric hindrance of the C-N bond. Existing methods for tertiary amine carbonylation often necessitate harsh reaction conditions, including the use of explosive mixtures of carbon monoxide and oxygen, or excessive amounts of strong oxidants. These requirements introduce severe safety hazards and complicate the purification process, often leading to lower yields and compromised product quality due to over-oxidation side reactions.
The Novel Approach
In stark contrast to these conventional limitations, the method described in patent CN111978194B introduces a remarkably elegant solution. By employing benzyl formate as a liquid surrogate for carbon monoxide, the process eliminates the need for high-pressure gas equipment and the associated safety risks. The reaction proceeds under relatively mild thermal conditions, typically around 130°C, using a palladium catalyst system that is highly tolerant of diverse functional groups. Crucially, the system facilitates the cleavage of the tertiary amine C-N bond without the addition of external oxidants, relying instead on the intrinsic reactivity of the catalytic cycle. This results in a cleaner reaction profile with fewer by-products. The use of trifluoroacetic anhydride (TFAA) as an activator further enhances the efficiency of the transformation, ensuring high conversion rates even with sterically demanding substrates. This novel approach not only simplifies the operational workflow but also significantly improves the atom economy and environmental footprint of the synthesis.
Mechanistic Insights into Pd-Catalyzed Carbonylation and C-N Bond Activation
The success of this synthesis hinges on the intricate interplay between the palladium catalyst, the bidentate phosphine ligand (DPEphos), and the activator (TFAA). The catalytic cycle likely initiates with the oxidative addition of the benzyl formate to the palladium center, generating a palladium-hydride species and releasing carbon monoxide in situ. This generated CO then coordinates to the metal center, forming a palladium-carbonyl complex. The presence of the bulky and electron-rich DPEphos ligand is critical here; it stabilizes the active palladium species and prevents the formation of inactive palladium black, thereby maintaining catalytic turnover over the extended reaction period of 24 hours. The subsequent insertion of the CO into the palladium-alkyl bond forms an acyl-palladium intermediate, which is the key precursor to the amide bond formation.
The activation of the tertiary amine represents the most distinct mechanistic feature of this process. Unlike primary amines that can easily coordinate and undergo nucleophilic attack, tertiary amines require C-N bond cleavage to participate effectively. The proposed mechanism suggests that the palladium complex facilitates this cleavage through a concerted metalation-deprotonation or an oxidative pathway that does not require external oxidants. The trifluoroacetic anhydride likely plays a role in activating the amine or the acyl intermediate, making the nitrogen more nucleophilic or the carbonyl more electrophilic, respectively. This precise control over the reaction pathway minimizes the formation of impurities such as over-alkylated by-products or hydrolysis products. The result is a highly selective transformation that yields aryl acetamides with purity levels suitable for pharmaceutical applications, as evidenced by the clean NMR spectra and high isolated yields reported in the experimental data.
How to Synthesize Aryl Acetamide Efficiently
The practical implementation of this synthesis is designed for reproducibility and scalability. The protocol involves charging a sealed reaction vessel with the palladium catalyst, ligand, benzyl formate, tertiary amine, and trifluoroacetic anhydride in an aprotic solvent like acetonitrile. The mixture is then heated to 130°C for 24 hours. Post-reaction, the workup is straightforward, involving filtration and silica gel treatment followed by column chromatography.
- Combine palladium acetate catalyst, DPEphos ligand, benzyl formate, tertiary amine, and trifluoroacetic anhydride in an organic solvent such as acetonitrile.
- Heat the reaction mixture to 130°C in a sealed vessel and maintain stirring for 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity aryl acetamide product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this technology offers tangible strategic advantages that extend beyond mere chemical efficiency. The shift from gaseous carbon monoxide to liquid benzyl formate fundamentally alters the safety profile of the manufacturing facility. By removing the requirement for high-pressure CO cylinders and specialized gas handling infrastructure, companies can significantly reduce capital expenditure and insurance costs. This simplification of the process equipment allows for greater flexibility in plant utilization and reduces the regulatory burden associated with handling toxic gases. Furthermore, the raw materials employed—benzyl formate, tertiary amines, and palladium acetate—are commercially available commodity chemicals with stable supply chains. This ensures consistent availability and mitigates the risk of production delays caused by raw material shortages.
- Cost Reduction in Manufacturing: The elimination of external oxidants and the use of a single liquid reagent for both carbonylation and alkylation drastically simplifies the material input list. This reduction in reagent complexity translates directly to lower procurement costs and simplified inventory management. Additionally, the high reaction efficiency and yield minimize the loss of valuable starting materials, optimizing the overall cost per kilogram of the final API intermediate. The avoidance of expensive and hazardous coupling reagents further contributes to substantial cost savings in the production budget.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against various functional groups means that a single standardized protocol can be applied to a wide range of substrates. This versatility reduces the need for multiple specialized production lines or extensive process re-optimization for different analogues. Consequently, lead times for new product introductions can be significantly shortened. The stability of the reagents also allows for longer storage periods without degradation, providing a buffer against supply chain disruptions and ensuring continuous production capability.
- Scalability and Environmental Compliance: The use of acetonitrile as a solvent and the absence of heavy metal oxidants simplify the waste treatment process. The post-treatment involves standard filtration and chromatography, which are well-established unit operations in the fine chemical industry. This ease of scale-up from gram to multi-ton quantities ensures that the process can meet commercial demand without encountering the typical bottlenecks associated with exotic chemistries. Moreover, the greener profile of the reaction aligns with increasingly stringent environmental regulations, reducing the liability and cost associated with waste disposal and emissions control.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams.
Q: What is the primary advantage of using benzyl formate in this synthesis?
A: Benzyl formate serves a dual role as both the carbon monoxide source and a reactant, eliminating the need for hazardous high-pressure CO gas and external oxidants, thereby significantly enhancing operational safety.
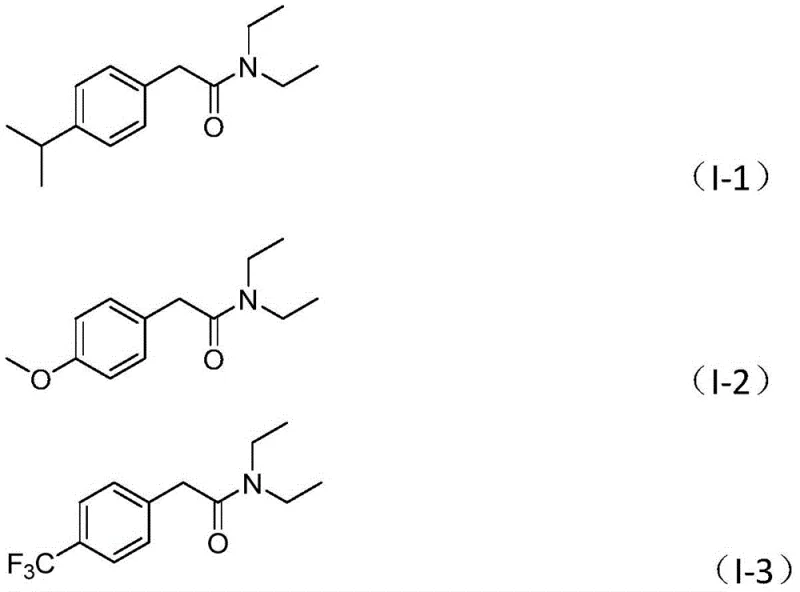
Q: Does this method tolerate diverse functional groups on the aryl ring?
A: Yes, the protocol demonstrates excellent substrate tolerance, successfully accommodating electron-withdrawing groups like trifluoromethyl and cyano, as well as electron-donating methoxy and alkyl groups, without significant yield loss.
Q: Is the cleavage of the tertiary amine C-N bond challenging in this process?
A: Unlike conventional methods that struggle with tertiary amine activation, this palladium-catalyzed system efficiently cleaves the C-N bond without requiring additional oxidants, offering a streamlined pathway to complex amide structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Acetamide Supplier
The technological breakthroughs encapsulated in patent CN111978194B represent a significant leap forward in the synthesis of aryl acetamide compounds, offering a safer and more efficient route for the production of critical pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage this advanced chemistry for your specific project needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest standards of quality required by the global pharmaceutical industry.
We invite you to collaborate with us to explore the full potential of this synthesis for your pipeline. Whether you require custom synthesis of specific aryl acetamide derivatives or a comprehensive Customized Cost-Saving Analysis for your existing supply chain, our technical procurement team is ready to assist. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
