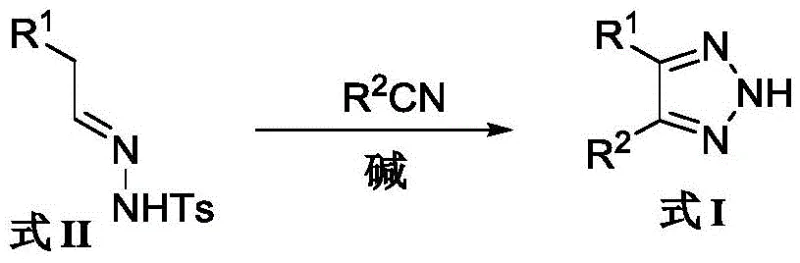
Advanced Base-Promoted Cyclization for High-Purity 4,5-Diaryl-1,2,3-Triazole Intermediates
The pharmaceutical and agrochemical industries continuously demand robust synthetic routes for heterocyclic scaffolds, particularly the 1,2,3-triazole motif, which serves as a critical bioisostere in modern drug design. Patent CN108794412B introduces a transformative preparation method for 4,5-diaryl-2H-1,2,3-triazole compounds, addressing long-standing challenges in purity and safety associated with traditional synthesis. This technology leverages a base-promoted cyclization between aryl aldehyde sulfonyl hydrazones and aromatic nitriles, bypassing the need for hazardous azide reagents. For R&D directors and procurement specialists, this innovation represents a pivotal shift towards safer, more efficient manufacturing of high-value oncology intermediates, such as analogs of combretastatin A-4. By eliminating toxic reagents and simplifying purification, this method significantly enhances the commercial viability of producing complex triazole derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,3-triazole ring has relied heavily on the Huisgen cycloaddition between alkynes and sodium azide. While chemically effective, this classical approach is fraught with severe safety liabilities due to the explosive nature of azides, necessitating expensive containment infrastructure and rigorous safety protocols that inflate operational costs. Furthermore, alternative methods involving functionalized alkenes often suffer from poor atom economy and require multi-step precursor synthesis. A more recent approach reported by Subhankar Panda utilized cesium carbonate to promote coupling; however, this method is critically flawed by the formation of self-coupling byproducts when unsymmetrical substrates are employed. These self-coupled impurities possess polarity and molecular weights nearly identical to the target product, rendering standard chromatographic separation inefficient and drastically reducing overall isolated yield to below 50 percent in many cases.
The Novel Approach
The methodology disclosed in CN108794412B circumvents these pitfalls by employing strong, non-nucleophilic bases such as potassium tert-butoxide (t-BuOK) or sodium bis(trimethylsilyl)amide (NaHMDS). This strategic choice of reagents facilitates a clean cyclization between the sulfonyl hydrazone and the nitrile without generating the problematic self-coupling side reactions observed with weaker bases. Experimental data within the patent demonstrates a dramatic improvement in efficiency; for instance, the synthesis of 4,5-diphenyl-2H-1,2,3-triazole achieves a yield of 89.5 percent using t-BuOK, compared to merely 45.6 percent when using cesium carbonate under similar conditions. This substantial increase in conversion efficiency directly translates to reduced raw material consumption and simplified downstream processing, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Base-Promoted Cyclization
The core of this synthetic breakthrough lies in the precise activation of the sulfonyl hydrazone moiety by strong alkoxide or amide bases. Upon deprotonation by t-BuOK or NaHMDS, the sulfonyl hydrazone generates a reactive diazo species in situ, which subsequently undergoes a [3+2] cycloaddition with the electrophilic carbon of the nitrile group. Unlike the cesium carbonate-mediated pathway, which allows for competitive dimerization of the hydrazone, the stronger basicity and specific coordination properties of t-BuOK drive the reaction kinetics preferentially towards the cross-coupled triazole product. This mechanistic selectivity is crucial for maintaining a clean impurity profile, as it prevents the formation of symmetrical triazole byproducts that are notoriously difficult to separate from the desired asymmetrical target molecule.
Furthermore, the reaction conditions are optimized to balance reactivity with stability. The patent specifies a temperature range of 60°C to 80°C, which provides sufficient thermal energy to overcome the activation barrier for cyclization without inducing thermal decomposition of the sensitive diazo intermediates. The use of polar aprotic solvents like DMF or toluene ensures adequate solubility of both the organic substrates and the inorganic base salts, facilitating homogeneous reaction conditions. This homogeneity is vital for heat transfer and mass transfer during scale-up, ensuring that the reaction proceeds uniformly throughout the reactor volume, thereby minimizing localized hot spots that could lead to degradation or safety incidents.
How to Synthesize 4,5-Diaryl-2H-1,2,3-Triazole Efficiently
To implement this synthesis effectively, operators must adhere to strict stoichiometric controls and temperature monitoring. The process begins with the dissolution of the aryl aldehyde sulfonyl hydrazone and the aromatic nitrile in a dry, oxygen-free solvent system. The addition of the base must be controlled to manage the exotherm associated with deprotonation. Following the reaction period, a careful quenching procedure using saturated ammonium chloride is required to neutralize excess base and decompose any remaining reactive intermediates safely. The detailed standardized synthesis steps are outlined in the guide below.
- Dissolve the aryl aldehyde sulfonyl hydrazone (Formula II) and aromatic nitrile (R2CN) in a polar aprotic solvent such as DMF or toluene.
- Add a strong non-nucleophilic base, specifically potassium tert-butoxide (t-BuOK) or NaHMDS, maintaining a molar ratio of substrate to base between 1: 2.5 and 1:3.0.
- Heat the reaction mixture to 60-80°C for 3-4 hours, then quench with saturated ammonium chloride and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible benefits beyond mere chemical yield. The elimination of sodium azide removes a major regulatory and safety burden, significantly lowering the cost of compliance and insurance for manufacturing facilities. Additionally, the high selectivity of the reaction reduces the load on purification units, allowing for faster batch turnover and increased throughput without the need for additional capital investment in separation equipment.
- Cost Reduction in Manufacturing: The switch from low-yielding cesium carbonate protocols to high-efficiency t-BuOK catalysis results in a drastic reduction in raw material waste. By achieving yields approaching 90 percent, the effective cost per kilogram of the active intermediate is significantly lowered. Moreover, the avoidance of expensive transition metal catalysts or specialized azide-handling equipment further contributes to substantial cost savings in the overall production budget.
- Enhanced Supply Chain Reliability: The reagents required for this process, including aromatic nitriles and sulfonyl hydrazones, are commodity chemicals with robust global supply chains. Unlike specialized azide reagents which may face shipping restrictions or supply bottlenecks, the inputs for this synthesis are readily available from multiple vendors. This diversity of supply sources mitigates the risk of production delays caused by raw material shortages, ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures (60-80°C) and atmospheric pressure, making it inherently safer and easier to scale from laboratory to pilot and commercial plants. The solvent systems used, such as toluene and DMF, are well-understood in industrial waste management, allowing for efficient recovery and recycling. This aligns with modern green chemistry principles, reducing the environmental footprint of the manufacturing process and simplifying the permitting process for new production lines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for process evaluation.
Q: How does this method improve upon previous triazole synthesis routes?
A: Unlike traditional azide-alkyne cycloadditions which involve hazardous explosives, or cesium carbonate methods that generate difficult-to-remove self-coupling impurities, this patent utilizes t-BuOK or NaHMDS to achieve high conversion with minimal byproduct formation.
Q: What represents the optimal reaction temperature for this cyclization?
A: The patent data indicates an optimal temperature range of 60°C to 80°C. Operating within this window maximizes yield (up to 96%) while minimizing reaction time to approximately 3-4 hours.
Q: Is this process scalable for commercial API production?
A: Yes, the use of common organic solvents like toluene and DMF, combined with commercially available bases and mild thermal conditions, makes this route highly amenable to kilogram-scale manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,5-Diaryl-2H-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4,5-diaryl-2H-1,2,3-triazole meets the exacting standards required for clinical and commercial applications.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the economic benefits of switching to this superior method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific project requirements.
