Advanced One-Pot Synthesis Strategy for High-Purity Lapatinib Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for critical oncology therapeutics, and patent CN102295638A presents a significant breakthrough in the manufacturing of Lapatinib, a potent small molecule targeted drug used extensively in the treatment of HER2-positive breast cancer. This intellectual property discloses a novel preparation method for the compound of Formula I, addressing long-standing challenges related to yield, purity, and environmental safety that have plagued previous synthetic routes. The core innovation lies in a streamlined one-pot strategy that cleverly utilizes imine formation to protect sensitive functional groups, thereby enabling a seamless transition through Suzuki coupling and final reduction without the need for intermediate isolation. For R&D directors and process chemists, this represents a paradigm shift towards more atom-economical and operationally simple methodologies that can be directly translated into commercial scale-up. By integrating this advanced chemistry into our portfolio, we offer a reliable lapatinib intermediate supplier capability that aligns with modern green chemistry principles while ensuring the highest standards of product quality required for global regulatory submissions.
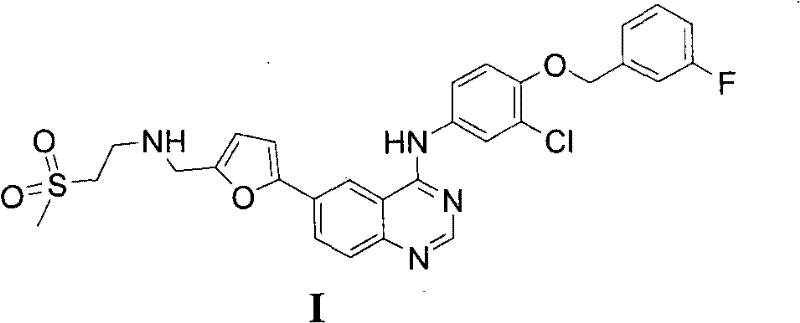
The limitations of conventional methods for synthesizing Lapatinib are well-documented in the prior art, presenting substantial hurdles for efficient commercial manufacturing. Early approaches, such as those described in WO9935146A1, relied heavily on the use of highly toxic organotin reagents for the coupling steps, which pose severe risks to both production safety and environmental compliance due to the difficulty in removing trace tin residues from the final active pharmaceutical ingredient. Furthermore, alternative routes like those in WO2008024439 necessitated the use of nitrogen protecting groups, specifically Boc protection and deprotection sequences, which inherently extend the production cycle and increase operational costs through additional reaction steps and purification requirements. Another significant drawback found in existing literature, such as CN1440403A, involves the instability of the aldehyde group in key intermediates; this functional group is prone to oxidation under high temperatures or varying pH conditions, leading to the formation of carboxylic acid impurities that compromise yield and necessitate rigorous, multi-step purification protocols. These cumulative inefficiencies result in prolonged lead times, excessive solvent consumption, and a complex impurity profile that challenges quality control teams.
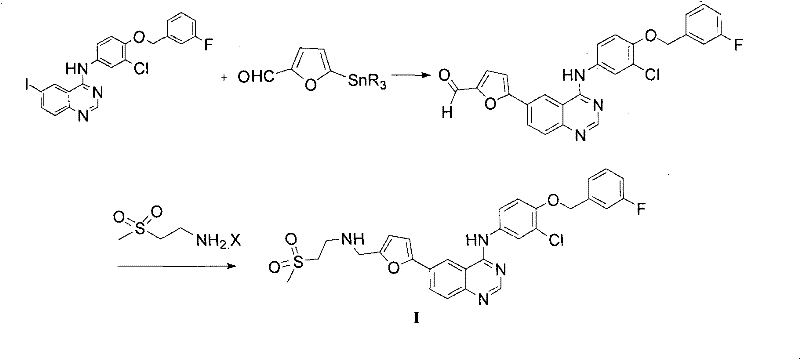
The novel approach disclosed in the patent overcomes these deficiencies through an ingenious one-pot synthesis that eliminates the need for external protecting groups and toxic reagents. The process initiates with the reaction of 2-(methylsulfonyl)ethylamine (Compound II) and 5-formyl-2-furylboronic acid (Compound III) to form an imine intermediate, which serves as a transient protecting group for the otherwise unstable aldehyde functionality. This strategic move allows the subsequent Suzuki coupling with the iodo-quinoline derivative (Compound IV) to proceed under elevated temperatures without the risk of aldehyde oxidation, a common failure point in traditional methods. Following the coupling, the imine is directly reduced in the same reaction vessel using a mild reducing agent to yield the final Lapatinib base, bypassing the need for isolation of the sensitive aldehyde intermediate. This telescoped process not only shortens the reaction sequence significantly but also drastically reduces the volume of organic solvents required, offering a cleaner and more cost-effective pathway for cost reduction in pharmaceutical intermediates manufacturing. The ability to perform these transformations sequentially in a single pot minimizes material handling and exposure, enhancing overall process safety and throughput.
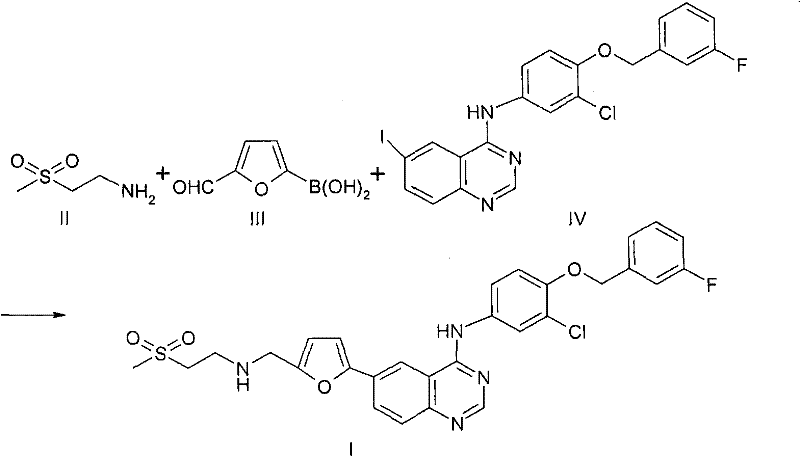
Mechanistic Insights into Imine-Protection Assisted Suzuki Coupling
The mechanistic elegance of this synthesis lies in the dual role of the amine component, which acts both as a reactant for the final side chain and as a protecting group provider during the critical cross-coupling phase. Initially, the primary amine of Compound II condenses with the aldehyde of Compound III to form a Schiff base or imine linkage; this transformation is thermodynamically favorable and effectively masks the electrophilic carbonyl carbon, rendering it inert to oxidative degradation during the subsequent palladium-catalyzed step. Once the imine is formed, the boronic acid moiety remains available for transmetallation with the palladium catalyst, facilitating the Suzuki-Miyaura coupling with the aryl iodide of Compound IV. The presence of the imine does not interfere with the catalytic cycle, allowing the biaryl bond formation to occur efficiently at temperatures ranging from 40°C to 130°C, depending on the solvent system employed. This compatibility is crucial, as it ensures that the fragile furan ring and the aldehyde equivalent survive the harsh conditions typically required for cross-coupling reactions, thereby maintaining the structural integrity of the molecule throughout the synthesis.
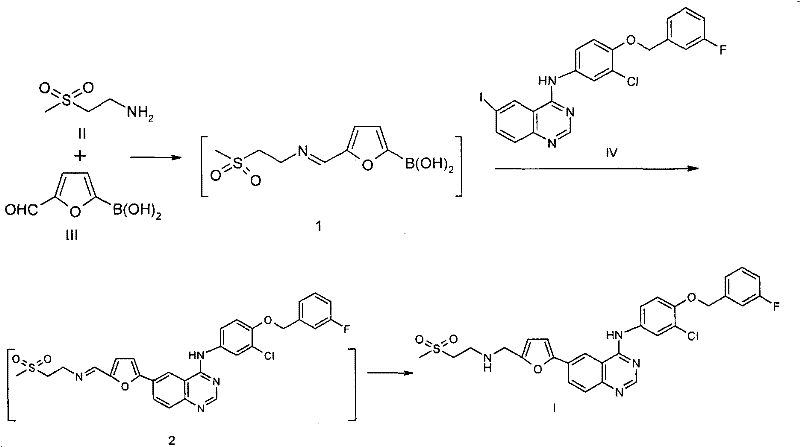
Impurity control is inherently built into this mechanism through the avoidance of free aldehyde intermediates and the elimination of tin-based reagents. In traditional routes, the free aldehyde is susceptible to over-oxidation to the corresponding carboxylic acid, a difficult-to-remove impurity that often requires recrystallization or chromatography to eliminate. By keeping the aldehyde masked as an imine until the very end of the sequence, the potential for this specific oxidative degradation pathway is virtually eliminated. Furthermore, the final reduction step using sodium triacetoxyborohydride is highly selective for the imine double bond, reducing it to the secondary amine without affecting other sensitive functionalities such as the chloro-substituent on the aniline ring or the ether linkage. This selectivity ensures a high-purity lapatinib intermediate profile, with patent examples demonstrating HPLC purities exceeding 98% directly after crystallization. The absence of heavy metal contaminants like tin also simplifies the downstream purification process, as there is no need for specialized scavenging resins or extensive aqueous workups designed to chelate and remove toxic metals, resulting in a cleaner final product suitable for stringent pharmaceutical applications.
How to Synthesize Lapatinib Efficiently
The implementation of this synthetic route requires precise control over reaction parameters to maximize the efficiency of the telescoped steps. The process begins by dissolving the starting materials, including the amine, boronic acid, and iodo-quinoline derivative, along with a palladium catalyst and base in a suitable organic solvent such as ethanol, THF, or DMF. The mixture is heated to facilitate both the initial imine formation and the subsequent Suzuki coupling, with temperature profiles carefully managed to ensure complete conversion of the halide starting material. Once the coupling is confirmed complete, typically via TLC or HPLC monitoring, the reaction mixture is cooled, and a reducing agent is introduced to convert the imine intermediate into the final amine product. The detailed standardized synthesis steps see the guide below for specific molar ratios, temperature ramps, and workup procedures optimized for kilogram-scale production.
- React 2-(methylsulfonyl)ethylamine with 5-formyl-2-furylboronic acid to form a protective imine intermediate.
- Perform Suzuki coupling with the iodo-quinoline derivative in the same pot using a palladium catalyst.
- Reduce the resulting imine intermediate using sodium triacetoxyborohydride to yield the final Lapatinib base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic methodology offers transformative benefits regarding cost structure and supply reliability. The elimination of toxic organotin reagents removes a significant bottleneck in the supply chain, as sourcing high-purity tin reagents can be volatile and their disposal incurs substantial environmental compliance costs. By shifting to a boron-based coupling strategy that utilizes commercially abundant and stable boronic acids, the raw material supply becomes more robust and less susceptible to geopolitical or regulatory disruptions. Additionally, the one-pot nature of the reaction drastically reduces the number of unit operations required; fewer isolation and purification steps mean less equipment occupancy time, lower labor costs, and reduced consumption of auxiliary materials like filtration media and solvents. This streamlining translates directly into substantial cost savings in manufacturing overheads, allowing for more competitive pricing models without compromising on the quality of the high-purity pharmaceutical intermediates supplied to downstream API manufacturers.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the telescoping of three distinct chemical transformations into a single reactor vessel. Traditional multi-step syntheses require quenching, extraction, drying, and concentration between each step, all of which consume energy, solvents, and time. By performing the imine formation, coupling, and reduction sequentially without isolating intermediates, the process significantly lowers the variable cost per kilogram. Furthermore, the avoidance of nitrogen protection groups like Boc eliminates the cost of purchasing protecting group reagents and the acids required for their removal, alongside the waste treatment costs associated with these additional chemicals. The simplified workup procedure, which often involves direct crystallization from the reaction mixture, further reduces the reliance on expensive chromatographic purification techniques, ensuring that the cost reduction in pharma intermediate manufacturing is realized through fundamental process intensification rather than mere supplier negotiation.
- Enhanced Supply Chain Reliability: Supply continuity is critically dependent on the robustness of the chemical process and the availability of starting materials. This novel route utilizes commodity chemicals such as ethanol or THF as solvents and widely available palladium catalysts, reducing the risk of supply shortages associated with exotic or highly regulated reagents. The stability of the intermediates within the reaction pot means that the process is less sensitive to minor fluctuations in processing times or temperatures compared to routes involving isolated unstable aldehydes. This robustness ensures consistent batch-to-batch quality and yield, minimizing the risk of production failures that could disrupt the supply of critical oncology medications. For supply chain planners, this predictability allows for tighter inventory management and more accurate forecasting, ensuring that production schedules for the final API can be met reliably even in the face of market volatility.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden complexities, particularly regarding heat transfer and waste management. The one-pot design of this synthesis is inherently scalable because it minimizes the number of physical transfers of hazardous materials between vessels, reducing the potential for spills or exposure incidents. The absence of toxic tin waste simplifies the environmental permitting process for manufacturing facilities, as the effluent streams are easier to treat and do not require specialized heavy metal removal infrastructure. This alignment with green chemistry principles not only reduces the environmental footprint of the manufacturing site but also future-proofs the supply chain against increasingly stringent global environmental regulations. The ability to produce high volumes of lapatinib intermediates with minimal waste generation makes this route an ideal candidate for sustainable commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is crucial for partners looking to integrate this technology into their existing manufacturing frameworks or source intermediates produced via this method. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating the feasibility of this approach for their specific supply chain needs.
Q: How does this method address the instability of the furan aldehyde intermediate?
A: The process utilizes an in situ imine formation between the amine and the aldehyde group, which effectively protects the unstable aldehyde from oxidation during the subsequent high-temperature Suzuki coupling step.
Q: What are the environmental advantages over prior art methods?
A: Unlike previous routes that relied on toxic organotin reagents requiring complex removal processes, this novel method avoids heavy metal contamination entirely, significantly simplifying waste treatment and environmental compliance.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the one-pot design eliminates multiple isolation and purification steps associated with Boc protection strategies, reducing solvent consumption and operational time, making it highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lapatinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and state-of-the-art infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this one-pot synthesis are fully realized in a GMP-compliant environment. Our facility is equipped with advanced reactors capable of handling the precise temperature controls and inert atmosphere conditions required for palladium-catalyzed couplings, supported by rigorous QC labs that enforce stringent purity specifications on every batch released. We understand that for oncology drugs like Lapatinib, consistency and impurity control are non-negotiable, and our quality systems are designed to detect and mitigate even trace levels of process-related impurities, guaranteeing a product that meets the highest global regulatory standards.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthetic route can be tailored to your specific volume and timeline requirements. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient methodology for your specific supply chain context. We encourage you to contact us to request specific COA data from our pilot batches and comprehensive route feasibility assessments, allowing you to make informed decisions that optimize both the quality and cost-efficiency of your Lapatinib supply chain.
