Scalable Trifluoromethylation of Bromopyridines for High-Purity Pharmaceutical Intermediates
Scalable Trifluoromethylation of Bromopyridines for High-Purity Pharmaceutical Intermediates
The introduction of trifluoromethyl groups into organic molecules is a critical transformation in modern medicinal chemistry, significantly enhancing the lipophilicity, metabolic stability, and membrane permeability of drug candidates. Patent CN108239021B discloses a groundbreaking trifluoromethylation process specifically designed for bromopyridine and its derivatives, addressing long-standing challenges in the synthesis of these valuable building blocks. This technology leverages a specialized fluoro-S-(trifluoromethyl)-dibenzothiophene salt, often referred to as a Umemoto-type reagent, to achieve high-yield conversions under copper-mediated conditions. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this method represents a significant leap forward in process efficiency and cost management. By shifting the paradigm from expensive iodide substrates to more economical bromides, the invention offers a sustainable pathway for the commercial scale-up of complex fluorinated heterocycles essential for next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl pyridines has been plagued by economic and technical barriers that hinder large-scale manufacturing. Traditional approaches, such as those cited in Chinese patent CN101973829B, rely heavily on 4-iodopyridine as the starting material. While effective on a small scale, iodopyridines are substantially more expensive than their brominated counterparts due to the higher cost of iodination reagents and the scarcity of raw materials. Furthermore, the trifluoromethylating agents used in these legacy processes, such as [Ph2SCF3]+[OTf]-, require cumbersome multi-step syntheses involving up to five distinct reaction stages, inflating the overall cost of goods. Another significant drawback identified in academic literature, such as the method disclosed in Organic Letters (2014), is the lack of scalability; while micro-tests show promise, these copper-mediated perfluorination routes often fail to maintain yield and efficiency when transferred to ton-scale production environments, rendering them unsuitable for industrial application.
The Novel Approach
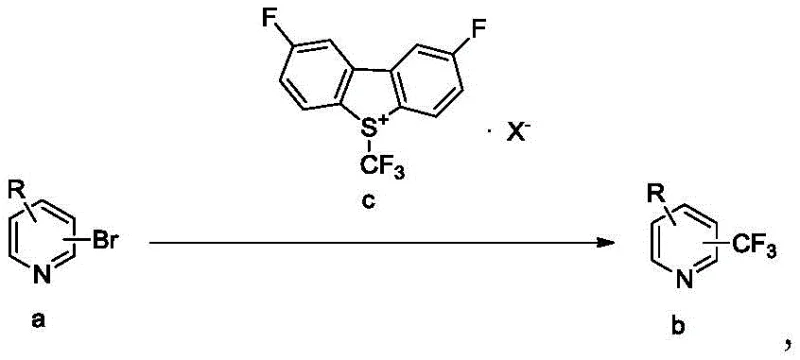
The patented process introduces a robust alternative that overcomes these economic and technical hurdles by utilizing bromopyridine compounds as the primary feedstock. As illustrated in the general reaction scheme below, the method employs a fluoro-S-(trifluoromethyl)-dibenzothiophene salt (Formula c) to effect the transformation of bromopyridines (Formula a) into the desired trifluoromethyl pyridines (Formula b). This approach not only capitalizes on the lower cost and wider availability of brominated precursors but also utilizes a trifluoromethyl reagent that can be synthesized in a single step, drastically simplifying the supply chain. The reaction proceeds efficiently in polar aprotic solvents like DMF or NMP at moderate temperatures, achieving fluorine spectrum yields as high as 93 percent. This technological advancement ensures that manufacturers can produce high-purity OLED material or pharmaceutical intermediates with greater economic viability and reduced environmental footprint compared to prior art.
Mechanistic Insights into Copper-Mediated Trifluoromethylation
The core of this innovation lies in the efficient activation of the carbon-bromine bond through a copper-mediated radical mechanism. Unlike traditional nucleophilic substitutions which may struggle with electron-deficient pyridine rings, this process utilizes copper powder to facilitate the generation of trifluoromethyl radicals from the sulfonium salt reagent. These reactive species then engage with the bromopyridine substrate, likely through a single-electron transfer (SET) pathway, to form the new carbon-carbon bond. The presence of the specific dibenzothiophene scaffold in the reagent enhances the stability and reactivity of the trifluoromethyl source, allowing for milder reaction conditions compared to harsher fluorinating agents. This mechanistic pathway is crucial for maintaining the integrity of sensitive functional groups often present in advanced drug intermediates, ensuring that the final product retains the necessary structural features for biological activity without degradation.
Impurity control is another critical aspect where this mechanism excels, particularly for R&D teams focused on purity specifications. By avoiding the use of iodide starting materials, the process eliminates the risk of iodine-containing impurities which can be difficult to remove and potentially toxic in final drug substances. The reaction conditions, typically maintained between 70 to 100°C for 3 to 5 hours, are optimized to maximize conversion while minimizing side reactions such as homocoupling or dehalogenation. The subsequent workup involves simple pH adjustment and filtration, which effectively removes copper residues and inorganic byproducts. This streamlined purification process results in a crude product of high quality, reducing the burden on downstream purification steps and ensuring that the commercial scale-up of complex polymer additives or active ingredients meets stringent regulatory standards.
How to Synthesize 2-Trifluoromethyl Isonicotinic Acid Tert-Butyl Ester Efficiently
To demonstrate the practical application of this technology, the patent details a specific embodiment for the synthesis of 2-trifluoromethyl isonicotinic acid tert-butyl ester, a valuable intermediate in medicinal chemistry. The procedure begins with the protection of the carboxylic acid group, followed by the key trifluoromethylation step using the novel reagent system. This sequence highlights the compatibility of the method with ester functionalities and showcases the operational simplicity required for industrial adoption. The detailed standardized synthesis steps provided below outline the precise stoichiometry, temperature controls, and workup procedures necessary to replicate the high yields reported in the patent data, serving as a reliable guide for process chemists aiming to implement this route.
- Under nitrogen protection, mix 2-bromoisonicotinic acid tert-butyl ester, DMF solvent, and copper powder in a reactor, cooling to 0-5°C.
- Add the plum reagent (fluoro-S-(trifluoromethyl)-dibenzothiophene salt) and stir in an ice-water bath before heating to 80°C for 3 hours.
- Dilute the reaction mixture with isopropyl acetate, adjust pH with KH2PO4 solution, filter, separate the organic layer, and concentrate to obtain the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this bromide-based trifluoromethylation process offers substantial strategic benefits beyond mere technical performance. The primary advantage stems from the drastic reduction in raw material costs; by substituting expensive iodopyridines with readily available bromopyridines, the direct material cost of the synthesis is significantly lowered. Furthermore, the trifluoromethylating reagent itself is economically superior, requiring only a one-step synthesis compared to the multi-step preparations of competing reagents found in prior art. This simplification of the reagent supply chain reduces dependency on niche chemical suppliers and mitigates the risk of supply disruptions, ensuring a more stable and predictable procurement landscape for long-term manufacturing contracts.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven by the elimination of costly iodine reagents and the simplification of the trifluoromethyl source synthesis. By utilizing a reagent that can be prepared in a single step, the manufacturing overhead associated with reagent production is drastically simplified, leading to substantial cost savings in the overall process. Additionally, the high yields achieved (up to 93 percent by NMR) minimize waste and maximize the throughput of the reactor, further enhancing the cost-efficiency of the operation. This makes the process highly attractive for cost reduction in electronic chemical manufacturing or large-scale API production where margin pressure is intense.
- Enhanced Supply Chain Reliability: The reliance on bromopyridines, which are commodity chemicals with robust global supply chains, significantly enhances the reliability of raw material sourcing. Unlike specialized iodinated compounds that may suffer from availability fluctuations, brominated precursors are produced in vast quantities for various industries, ensuring consistent access. Moreover, the ability to recover and reuse certain components of the reaction mixture, as suggested by the patent's emphasis on sustainability, adds another layer of security to the supply chain. This reliability is critical for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to meet tight delivery schedules without compromising on quality.
- Scalability and Environmental Compliance: From an environmental and operational standpoint, the process is designed for seamless scalability from laboratory benchtop to multi-ton production. The use of standard solvents like DMF and common equipment (four-mouth bottles or standard reactors) means that existing infrastructure can be utilized without major capital investment. The post-treatment steps involve simple aqueous workups and filtrations, which generate less hazardous waste compared to processes requiring exotic quenching agents. This alignment with green chemistry principles not only simplifies waste management but also ensures compliance with increasingly stringent environmental regulations, facilitating smoother regulatory approvals for commercial scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethylation technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is this bromopyridine trifluoromethylation process more cost-effective than iodide methods?
A: This process utilizes bromopyridines, which are significantly cheaper and more readily available than the corresponding iodopyridines required by older methods. Additionally, the trifluoromethylating reagent used here requires only a one-step synthesis, compared to the five or six steps needed for reagents in prior art, drastically reducing raw material costs.
Q: What is the scalability potential of this copper-mediated reaction?
A: The patent explicitly addresses the limitations of micro-test methods found in literature, demonstrating that this process maintains high yields (up to 93% by NMR) even when scaled. The use of robust conditions (80°C in DMF) and simple workup procedures makes it highly suitable for ton-scale commercial production.
Q: Does this method tolerate various functional groups on the pyridine ring?
A: Yes, the process exhibits excellent functional group tolerance. The patent data confirms successful trifluoromethylation on substrates containing esters, nitriles, methyl groups, and protected acids, making it versatile for synthesizing diverse pharmaceutical and agrochemical intermediates without extensive protecting group strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented trifluoromethylation technology for the production of high-value fluorinated intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of trifluoromethyl pyridine delivered meets the exacting standards required by the global pharmaceutical and agrochemical industries. We are committed to leveraging this innovative chemistry to provide our clients with a competitive edge in the market.
We invite you to collaborate with us to explore how this cost-effective and scalable process can optimize your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fluorine chemistry can drive value and efficiency in your next project.
