Advanced Manufacturing of Magnolol: Leveraging Niobium Catalysis for Commercial Scale-Up
Advanced Manufacturing of Magnolol: Leveraging Niobium Catalysis for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and economically viable synthetic routes for high-value bioactive compounds. A pivotal development in this domain is documented in patent CN109053387B, which discloses a highly efficient method for the synthesis of Magnolol, a potent bioactive neolignan found in Magnolia officinalis. This patent introduces a streamlined two-step protocol that fundamentally alters the production landscape by replacing harsh thermal conditions with a sophisticated Lewis acid-catalyzed rearrangement. By utilizing 1,1'-biphenyl-2,2'-diol as the starting scaffold, the process achieves exceptional purity and yield through a controlled O-allylation followed by a niobium pentachloride (NbCl5) mediated [3,3]-sigmatropic rearrangement. For global procurement teams and R&D directors, this technology represents a significant leap forward, offering a reliable magnolol supplier pathway that mitigates the risks of isomer contamination and excessive waste generation inherent in legacy methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Magnolol and its isomers has been plagued by significant thermodynamic and kinetic challenges that hinder cost-effective manufacturing. Traditional approaches often rely on thermal Claisen rearrangements, which necessitate extremely high temperatures to overcome the activation energy barrier for the migration of the allyl group. These harsh thermal conditions frequently result in the degradation of the sensitive biphenyl backbone and the polymerization of allyl moieties, leading to complex reaction mixtures that are notoriously difficult to separate. Furthermore, the lack of regioselectivity in uncatalyzed thermal processes often generates substantial quantities of unwanted isomers, such as honokiol or other positional variants, which drastically reduces the effective yield of the target API intermediate. The downstream purification required to remove these impurities involves multiple chromatographic steps or recrystallizations, inflating both the operational expenditure and the environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast, the methodology outlined in CN109053387B employs a catalytic strategy that operates under remarkably mild conditions, effectively bypassing the limitations of thermal activation. By introducing Niobium Pentachloride as a potent Lewis acid catalyst, the reaction barrier for the rearrangement is significantly lowered, allowing the transformation to proceed efficiently at cryogenic temperatures ranging from -20°C to -78°C. This低温 (low-temperature) operation not only preserves the structural integrity of the molecule but also exerts precise control over the regioselectivity of the allyl migration, ensuring that the allyl groups install exclusively at the desired positions to form Magnolol. The result is a process that delivers high-purity magnolol with minimal byproduct formation, simplifying the isolation procedure to a straightforward aqueous workup and crystallization, thereby offering substantial cost savings in pharmaceutical intermediates manufacturing.
Mechanistic Insights into NbCl5-Catalyzed Rearrangement
The core innovation of this synthetic route lies in the unique electronic interaction between the Niobium Pentachloride catalyst and the ether oxygen atoms of the intermediate. In the first stage, 1,1'-biphenyl-2,2'-diol undergoes nucleophilic substitution with an allyl halide in the presence of a base, such as potassium carbonate, to form the bis-allyl ether intermediate. This step is critical for setting up the substrate for the subsequent rearrangement, and the patent specifies that using anhydrous conditions and specific molar ratios ensures near-quantitative conversion.

Following the formation of the ether, the addition of NbCl5 initiates the catalytic cycle. The niobium center, being highly electrophilic, coordinates with the lone pair electrons of the ether oxygen, thereby weakening the C-O bond and facilitating the concerted [3,3]-sigmatropic shift. This coordination stabilizes the transition state, allowing the allyl group to migrate from the oxygen to the ortho-position of the aromatic ring with high fidelity. Unlike thermal rearrangements that rely on entropy-driven chaos, this Lewis acid-mediated pathway is enthalpy-driven and highly ordered, which explains the observed suppression of side reactions. The reaction is typically quenched by pouring into cold water, which hydrolyzes the niobium complexes and precipitates the product, demonstrating a mechanism that is not only chemically elegant but also operationally simple for commercial scale-up of complex pharmaceutical intermediates.
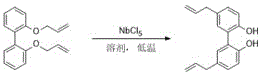
How to Synthesize Magnolol Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to maximize the benefits of the niobium catalysis. The process begins with the preparation of the bis-allyl ether precursor, followed by the critical low-temperature rearrangement step where the catalyst loading and reaction time must be optimized. Detailed standardized operating procedures regarding reagent grades, solvent drying, and quenching protocols are essential for reproducibility. For a comprehensive guide on the specific molar ratios and workup techniques described in the patent, please refer to the technical breakdown below.
- React 1,1'-biphenyl-2,2'-diol with allyl halide in the presence of a base (e.g., K2CO3) to form 2,2'-bisallyloxy-1,1'-biphenyl.
- Dissolve the intermediate in a solvent like dichloromethane and cool to -78°C.
- Add Niobium Pentachloride (NbCl5) catalyst and stir to effect rearrangement into Magnolol.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this NbCl5-catalyzed route offers transformative advantages that extend far beyond simple yield improvements. The shift from high-energy thermal processes to low-temperature catalytic ones fundamentally alters the cost structure of production by reducing energy consumption and minimizing the need for expensive, corrosion-resistant high-temperature reactors. Furthermore, the high selectivity of the reaction means that raw material utilization is maximized, as less feedstock is lost to the formation of inseparable isomers or polymeric tars. This efficiency translates directly into a more stable and predictable supply chain, reducing the lead time for high-purity pharmaceutical intermediates and ensuring consistent quality batches for downstream formulation.
- Cost Reduction in Manufacturing: The elimination of high-temperature heating phases results in significantly reduced energy costs per kilogram of product. Additionally, because the reaction produces fewer byproducts, the downstream purification burden is drastically simplified, removing the need for costly preparative HPLC or multiple recrystallization cycles. The use of common solvents like dichloromethane and toluene, which are easily recovered and recycled, further enhances the economic viability of the process, driving down the overall cost of goods sold (COGS) without compromising on quality standards.
- Enhanced Supply Chain Reliability: The starting materials, specifically 1,1'-biphenyl-2,2'-diol and allyl halides, are commodity chemicals with robust global supply chains, mitigating the risk of raw material shortages. The mild reaction conditions also reduce the wear and tear on processing equipment, leading to lower maintenance downtime and higher asset utilization rates. This operational stability ensures that manufacturers can meet tight delivery schedules and maintain continuous production runs, which is critical for just-in-time inventory models employed by major pharmaceutical companies.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates less hazardous waste due to the absence of thermal degradation products and the ability to recycle solvents efficiently. The workup procedure, which involves a simple aqueous quench and phase separation, is inherently safer and easier to scale than complex distillation or chromatographic separations. This aligns perfectly with modern green chemistry principles and regulatory requirements, facilitating smoother regulatory approvals and reducing the environmental compliance burden on the manufacturing site.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into existing production lines. The following questions address common concerns regarding catalyst handling, safety, and scalability, derived directly from the technical specifications of the patent. These insights are intended to provide clarity on the operational feasibility and commercial potential of this advanced manufacturing route.
Q: Why is Niobium Pentachloride preferred over thermal rearrangement for Magnolol synthesis?
A: Traditional thermal Claisen rearrangements require high temperatures which often lead to decomposition and isomer mixtures. NbCl5 catalysis allows the reaction to proceed at low temperatures (-20°C to -78°C), ensuring high selectivity and easier purification.
Q: What is the expected yield of this synthetic route?
A: According to patent data, the O-allylation step can achieve yields up to 99%, and the subsequent rearrangement step yields over 81%, resulting in a highly efficient overall process.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents and standard unit operations. The mild conditions reduce energy consumption and safety risks associated with high-temperature reactions, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Magnolol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting cutting-edge synthetic methodologies like the NbCl5-catalyzed route for Magnolol production. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle moisture-sensitive Lewis acid chemistry with precision, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this innovative technology for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable, high-quality supply of this critical intermediate.
