Scalable One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones via Palladium Catalysis
Introduction to Advanced Quinazolinone Manufacturing
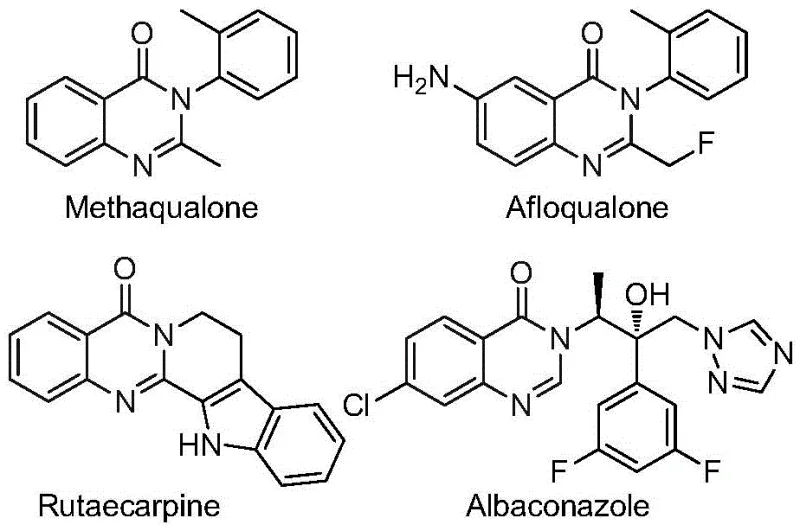
The pharmaceutical and agrochemical industries continuously seek robust synthetic routes for nitrogen-containing heterocycles due to their pervasive presence in bioactive molecules. Patent CN112480015B discloses a groundbreaking multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones, a structural motif found in numerous therapeutic agents ranging from antifungals to anticancer drugs. The introduction of the trifluoromethyl group is particularly strategic, as it markedly enhances the metabolic stability, lipophilicity, and bioavailability of the parent molecule, addressing key pharmacokinetic challenges in modern drug design. This technical insight report analyzes the proprietary methodology detailed in the patent, highlighting its potential to serve as a reliable platform for the commercial scale-up of complex pharmaceutical intermediates.
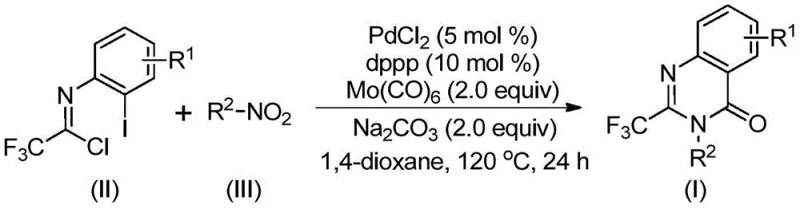
Traditionally, accessing these privileged scaffolds required harsh conditions or multi-step sequences that eroded overall yield and increased waste. The disclosed invention leverages a palladium-catalyzed carbonylation cascade that merges readily available nitro compounds with trifluoroethylimidoyl chloride. By utilizing molybdenum hexacarbonyl as a safe, solid carbon monoxide surrogate, the process circumvents the safety hazards associated with handling high-pressure CO gas. This innovation represents a significant leap forward in process chemistry, offering a streamlined pathway that aligns with the principles of green chemistry while maintaining high reaction efficiency and broad substrate compatibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has been plagued by significant synthetic hurdles that impede efficient manufacturing. Conventional strategies often rely on ruthenium or platinum-catalyzed reductive N-heterocyclization of nitro-substituted benzamides, which necessitates the use of high-pressure carbon monoxide atmospheres. Such conditions demand specialized autoclave equipment and rigorous safety protocols, drastically increasing capital expenditure and operational complexity. Furthermore, alternative iron-catalyzed condensation reactions or palladium-catalyzed cyclizations involving 2-bromoformylaniline frequently suffer from narrow substrate scopes and the requirement for expensive, pre-activated starting materials. These legacy methods often result in low yields and generate substantial chemical waste, making them economically unviable for the cost-sensitive production of high-volume pharmaceutical intermediates.
The Novel Approach
In stark contrast, the method described in CN112480015B introduces a highly efficient, one-pot tandem reaction that fundamentally simplifies the synthetic landscape. By employing trifluoroethylimidoyl chloride and nitro compounds as the primary building blocks, the process achieves the simultaneous formation of multiple bonds in a single operational step. The use of Mo(CO)6 as an in situ source of carbon monoxide allows the reaction to proceed under standard atmospheric pressure conditions at a moderate temperature of 120°C. This approach not only eliminates the need for hazardous gas cylinders but also enhances the atom economy of the transformation. The reaction system exhibits remarkable tolerance for various functional groups, enabling the synthesis of diverse derivatives without the need for protecting group strategies, thereby reducing the total number of synthetic steps and associated costs.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a sophisticated example of transition metal catalysis driving a multi-component assembly. The reaction likely initiates with the reduction of the nitro compound to the corresponding amine by molybdenum hexacarbonyl, which serves a dual role as both a CO source and a reducing agent. Subsequently, a base-promoted intermolecular coupling occurs between the generated amine and trifluoroethylimidoyl chloride, forming a trifluoroacetamidine intermediate. The palladium catalyst then inserts into the carbon-iodine bond of the imidoyl chloride moiety, generating a reactive divalent palladium species. As the temperature rises, Mo(CO)6 decomposes to release carbon monoxide, which inserts into the carbon-palladium bond to form an acyl-palladium intermediate. This critical step sets the stage for the final cyclization, where the nitrogen atom attacks the acyl center to close the ring, followed by reductive elimination to release the stable 2-trifluoromethyl quinazolinone product.
Understanding this mechanism is crucial for R&D teams aiming to optimize impurity profiles and maximize yield. The careful selection of the ligand, specifically 1,3-bis(diphenylphosphino)propane (dppp), is essential for stabilizing the palladium center throughout the catalytic cycle and facilitating the difficult reductive elimination step. The base, typically sodium carbonate, plays a vital role in neutralizing the hydrochloric acid byproduct generated during the amidine formation, driving the equilibrium forward. This intricate balance of reagents ensures that side reactions, such as homocoupling or incomplete carbonylation, are minimized. The ability to control these mechanistic variables allows for the production of high-purity intermediates, which is a non-negotiable requirement for downstream API synthesis and regulatory compliance.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO source, and substrates in an aprotic solvent like 1,4-dioxane. The mixture is then heated to facilitate the cascade reaction. While the general procedure is straightforward, attention to detail regarding reagent purity and stoichiometry is paramount for achieving the reported high yields. For a comprehensive, step-by-step guide tailored to your specific facility capabilities, please refer to the standardized operating procedure below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for 16 to 30 hours to allow the carbonylation cascade to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthetic route offers compelling advantages that directly impact the bottom line and operational resilience. The shift from high-pressure gas reactions to a solution-phase process using solid reagents significantly lowers the barrier to entry for manufacturing partners. This reduces the dependency on specialized infrastructure, allowing for more flexible production scheduling and the utilization of a broader range of contract manufacturing organizations. Furthermore, the use of commodity chemicals like nitro compounds and sodium carbonate ensures a stable and cost-effective supply chain, mitigating the risks associated with sourcing exotic or unstable precursors.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the operational workflow. By consolidating multiple synthetic steps into a single one-pot reaction, manufacturers can drastically reduce labor costs, solvent consumption, and energy usage associated with intermediate isolation and purification. The elimination of high-pressure equipment maintenance and the use of inexpensive, commercially available catalysts further contribute to a leaner cost structure. Additionally, the high conversion rates observed across various substrates minimize raw material waste, enhancing the overall material efficiency of the production campaign.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on robust, shelf-stable starting materials. Nitro compounds and trifluoroethylimidoyl chlorides are widely produced industrial chemicals with established global supply networks, reducing the risk of shortages that often plague niche reagents. The tolerance of the reaction to various functional groups means that a single manufacturing platform can produce a wide array of derivatives, allowing for agile responses to changing market demands without the need for extensive process re-validation or equipment modification.
- Scalability and Environmental Compliance: The scalability of this method is evidenced by its successful demonstration at the gram level with clear pathways to kilogram and tonne scales. The use of 1,4-dioxane, a common industrial solvent, facilitates easy solvent recovery and recycling, aligning with modern environmental sustainability goals. Moreover, the avoidance of toxic carbon monoxide gas and heavy metal waste streams simplifies effluent treatment and regulatory reporting, ensuring that production facilities remain compliant with increasingly stringent environmental regulations while maintaining high throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this method into your existing production pipelines.
Q: What are the key advantages of this one-pot method over traditional quinazolinone synthesis?
A: This method eliminates the need for high-pressure carbon monoxide gas and expensive pre-activated substrates. It utilizes cheap nitro compounds and a solid CO source (Mo(CO)6), significantly simplifying the operational setup and improving safety profiles for industrial scale-up.
Q: What is the substrate scope for the R2 group in the nitro compound?
A: The process demonstrates excellent functional group tolerance. The R2 group can be varied widely to include C1-C10 alkyl chains, cycloalkyl groups like cyclohexyl, and various substituted or unsubstituted aryl groups, allowing for the rapid generation of diverse chemical libraries.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the patent explicitly states that the method can be expanded to the gram level and beyond. The use of commercially available reagents and standard organic solvents like dioxane makes it highly amenable to commercial scale-up without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking to secure their supply chains.
We invite you to leverage our technical expertise to optimize your production costs and accelerate your time to market. By collaborating with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the competitive landscape of fine chemical intermediates.
