Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
Introduction to Next-Generation Quinazolinone Synthesis
Quinazolinone derivatives represent a privileged scaffold in modern medicinal chemistry, widely recognized for their extensive spectrum of biological activities ranging from anti-inflammatory and antiviral to potent anticancer properties. The structural integrity of these fused-ring nitrogen-containing heterocycles serves as the backbone for numerous commercially successful pharmaceutical agents, including methaqualone and various kinase inhibitors. However, the introduction of a trifluoromethyl group at the 2-position of the quinazolinone ring significantly enhances the physicochemical profile of the parent molecule, improving metabolic stability, lipophilicity, and bioavailability. Despite the clear therapeutic value, synthetic access to these specific 2-trifluoromethyl substituted derivatives has historically been constrained by limited methodologies. Patent CN112125856A addresses this critical gap by disclosing a robust preparation method that leverages transition metal palladium catalysis to efficiently construct these complex architectures.
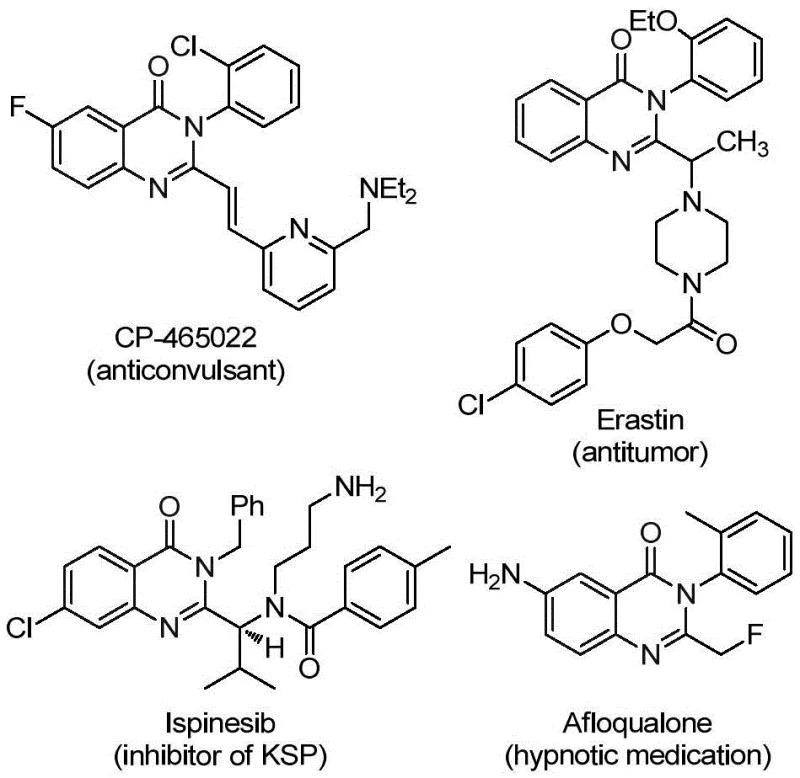
This groundbreaking technology not only streamlines the synthetic route but also aligns with modern green chemistry principles by replacing hazardous gaseous reagents with solid surrogates. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, understanding the nuances of this patent is essential for securing a competitive edge in API development. The method described herein offers a versatile platform for generating diverse libraries of quinazolinone derivatives, facilitating rapid structure-activity relationship (SAR) studies while maintaining high standards of purity and yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl substituted quinazolinones has relied on several established but flawed protocols that hinder large-scale commercial adoption. Common strategies include the cyclization of anthranilamide with ethyl trifluoroacetate or trifluoroacetic anhydride, often requiring harsh reaction conditions that compromise sensitive functional groups. Alternative approaches utilizing isatoic anhydride or T3P-promoted tandem reactions frequently suffer from low atom economy and the necessity for expensive, pre-activated substrates. Furthermore, many traditional carbonylation methods depend on the direct use of carbon monoxide gas, a toxic and colorless substance that necessitates specialized high-pressure reactor infrastructure and rigorous safety protocols. These factors collectively result in elevated production costs, narrow substrate scope, and significant operational hazards, making cost reduction in pharmaceutical intermediate manufacturing a persistent challenge for supply chain managers.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a palladium-catalyzed carbonylation tandem reaction that fundamentally reshapes the synthetic landscape. By employing 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide substitute, the method completely circumvents the handling of toxic CO gas, thereby drastically simplifying the engineering requirements for scale-up. The reaction proceeds under relatively mild thermal conditions (90°C) in standard organic solvents like tetrahydrofuran, utilizing cheap and commercially available o-iodoanilines and trifluoroethylimidoyl chlorides as starting materials. This shift not only enhances the safety profile of the manufacturing process but also broadens the functional group tolerance, allowing for the incorporation of diverse substituents such as halogens, nitro groups, and bulky aryl moieties without significant yield penalties. The operational simplicity and high efficiency of this route make it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Reaction
The efficacy of this synthesis relies on a sophisticated catalytic cycle initiated by the oxidative addition of a palladium(0) species into the carbon-iodine bond of the o-iodoaniline substrate. Following this activation, the solid CO surrogate TFBen undergoes thermal decomposition to release carbon monoxide in situ, which subsequently inserts into the carbon-palladium bond to form a reactive acyl-palladium intermediate. This step is critical as it introduces the carbonyl functionality required for the quinazolinone core without external gas feeders. Concurrently, the trifluoroethylimidoyl chloride participates in an intermolecular carbon-nitrogen bond coupling promoted by the base potassium tert-butoxide, generating a trifluoroacetamidine derivative that coordinates with the metal center. The resulting seven-membered ring palladium intermediate then undergoes reductive elimination to forge the final C-N bond, releasing the 2-trifluoromethyl substituted quinazolinone product and regenerating the active palladium catalyst for the next turnover.
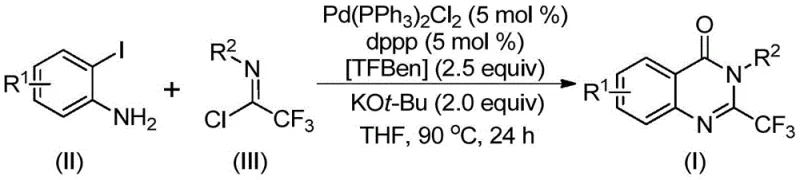
From an impurity control perspective, the use of a well-defined palladium catalyst system combined with the specific ligand 1,3-bis(diphenylphosphine)propane (dppp) ensures high regioselectivity and minimizes side reactions such as homocoupling or dehalogenation. The basic conditions provided by KOt-Bu facilitate the necessary deprotonation steps while maintaining the stability of the trifluoromethyl group, which is susceptible to nucleophilic attack under strongly acidic or overly nucleophilic conditions. This precise balance of reactivity allows for the synthesis of high-purity products where the impurity profile is dominated by easily removable byproducts rather than structurally similar analogs. For quality assurance teams, this mechanistic clarity translates to predictable batch-to-batch consistency and simplified purification workflows, typically involving standard filtration and silica gel chromatography.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical implementation of this synthesis protocol is designed for straightforward execution in standard laboratory or pilot plant settings. The process begins with the precise charging of reagents into a reaction vessel, followed by heating and stirring for a defined period to ensure complete conversion. The use of common solvents and commercially sourced catalysts eliminates the need for custom synthesis of exotic reagents, further lowering the barrier to entry for production. While the general procedure is robust, optimization of reaction time and temperature may be required for specific substrate combinations to maximize yield. For detailed operational parameters and stoichiometric ratios tailored to specific derivatives, please refer to the standardized synthesis guide below.
- Charge a reaction vessel with palladium catalyst Pd(PPh3)2Cl2, ligand dppp, base KOt-Bu, solid CO surrogate TFBen, trifluoroethylimidoyl chloride, and o-iodoaniline in THF solvent.
- Heat the reaction mixture to 90°C and maintain stirring for 16 to 30 hours to allow the carbonylation and cyclization to proceed to completion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl-substituted quinazolinone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the substantial cost savings achieved through the utilization of inexpensive, commodity-grade starting materials. Unlike previous methods that required specialized anhydrides or activated acids, this route leverages o-iodoanilines and trifluoroethylimidoyl chlorides, which are widely available in the global chemical market. This abundance ensures a stable supply chain and mitigates the risk of raw material shortages that often plague niche pharmaceutical syntheses. Furthermore, the elimination of high-pressure carbon monoxide equipment significantly reduces capital expenditure (CAPEX) for manufacturing facilities, allowing for faster deployment of production lines and reduced lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the replacement of hazardous and expensive reagents with cost-effective alternatives. By avoiding the use of T3P coupling reagents and unstable trifluoroacetylating agents, the overall material cost per kilogram of product is significantly lowered. Additionally, the simplified post-treatment process, which avoids complex extraction or distillation steps in favor of straightforward filtration and chromatography, reduces labor and utility costs. The high yields reported across a broad substrate scope mean less waste generation and higher throughput, directly contributing to improved profit margins for API manufacturers seeking cost reduction in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-hazardous reagents that do not require special shipping permits or storage conditions associated with toxic gases. The solid nature of the CO surrogate (TFBen) allows for easy handling and inventory management, reducing the logistical complexity often associated with gaseous reagents. Moreover, the compatibility of the reaction with a wide variety of substituents means that a single manufacturing platform can produce multiple derivatives, allowing for flexible production scheduling and rapid response to changing market demands. This versatility ensures a reliable quinazolinone derivative supplier capability that can adapt to diverse client needs without extensive process re-validation.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns perfectly with increasingly stringent global safety standards. The avoidance of toxic carbon monoxide gas eliminates the risk of accidental exposure and simplifies environmental permitting for new facilities. The reaction generates minimal hazardous waste, and the use of standard organic solvents like THF facilitates solvent recovery and recycling programs. The scalability of the process is evidenced by its successful application across a range of substrates with consistent performance, indicating that translation from gram-scale laboratory synthesis to multi-ton commercial production can be achieved with minimal technical risk. This makes it an ideal solution for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the safety advantages of using TFBen over traditional carbon monoxide sources?
A: Traditional methods often require toxic, colorless carbon monoxide gas under high pressure, posing significant safety risks. This patented method utilizes 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide surrogate, which decomposes in situ to release CO safely, eliminating the need for specialized high-pressure gas equipment and enhancing operational safety.
Q: Does this synthesis method support a wide range of substrate substituents?
A: Yes, the method demonstrates excellent substrate compatibility. It tolerates various substituents on both the o-iodoaniline and the imidoyl chloride components, including halogens (F, Cl, Br), alkyl groups, nitro groups, and bulky aryl systems like naphthyl, allowing for the design of diverse quinazolinone libraries.
Q: What represents the primary cost driver reduction in this new protocol?
A: The primary cost reductions stem from the use of cheap and readily available starting materials such as o-iodoanilines and trifluoroethylimidoyl chlorides. Additionally, the avoidance of expensive pre-activated substrates and the simplification of post-treatment processes contribute to a more economically viable manufacturing route compared to prior art methods involving T3P or unstable anhydrides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. We are committed to delivering high-purity 2-trifluoromethyl quinazolinone derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality and safety makes us a trusted partner for multinational pharmaceutical companies seeking to secure their supply chains for key oncology and anti-inflammatory intermediates.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and efficiency in your supply chain. Let us help you transform innovative chemistry into commercial success.
