Scalable Palladium-Catalyzed Carbonylation for High-Purity 2,3-Dihydroquinolone Intermediates
Introduction to Next-Generation Heterocyclic Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more efficient, and scalable synthetic routes. A significant breakthrough in this domain is detailed in patent CN112239456B, which discloses a robust preparation method for substituted 2,3-dihydroquinolone compounds. These nitrogen-containing carbonyl heterocycles are not merely academic curiosities; they serve as critical scaffolds in medicinal chemistry, underpinning the biological activity of numerous therapeutic agents. As illustrated in the provided structural data, derivatives of this core skeleton have demonstrated potent antitumor activity against human cells and significant analgesic properties, making them highly sought-after targets for drug discovery programs globally.
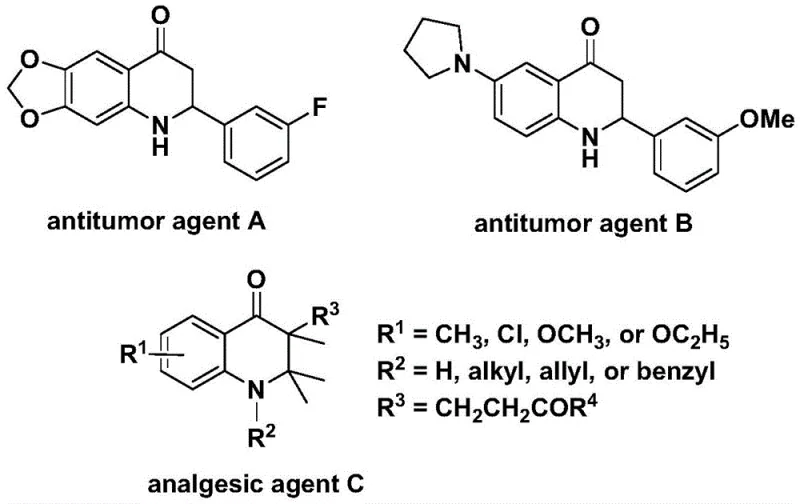
The innovation presented in this patent addresses a long-standing challenge in organic synthesis: the efficient construction of the quinolone core via carbonylation without the logistical and safety nightmares associated with traditional carbon monoxide sources. By leveraging a transition metal palladium catalyst system in conjunction with a solid CO surrogate, this methodology offers a streamlined pathway that is both operationally simple and chemically versatile. For R&D directors and procurement specialists alike, understanding the nuances of this technology is essential for securing a reliable supply of high-quality intermediates while mitigating the risks associated with hazardous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles like 2,3-dihydroquinolones has often relied on direct carbonylation reactions using carbon monoxide gas. While chemically effective, the use of gaseous CO presents severe limitations for industrial application. The toxicity of carbon monoxide requires specialized high-pressure equipment, rigorous safety protocols, and dedicated infrastructure that many fine chemical facilities lack. Furthermore, controlling the stoichiometry of a gas in a liquid-phase reaction can be technically demanding, often leading to inconsistent results or incomplete conversions. Alternative methods involving pre-functionalized acid chlorides or anhydrides can introduce additional synthetic steps, increasing the overall cost and reducing the atom economy of the process. These factors collectively create bottlenecks in the supply chain, extending lead times and inflating the cost of goods sold for the final active pharmaceutical ingredients.
The Novel Approach
The methodology described in patent CN112239456B represents a paradigm shift by replacing hazardous CO gas with 1,3,5-trimesic acid phenol ester (TFBen), a stable and easy-to-handle solid surrogate. This substitution fundamentally alters the risk profile of the reaction, allowing it to be conducted in standard glassware or reactors without the need for high-pressure gas lines. The reaction proceeds smoothly in the presence of a palladium catalyst, specifically bis(acetylacetone)palladium, and a dppp ligand, facilitating the insertion of the carbonyl group directly into the molecular framework. This approach not only enhances safety but also simplifies the workflow, as the solid reagent can be weighed and added with precision, ensuring reproducible reaction outcomes. The ability to generate the reactive acyl-palladium species in situ from a benign precursor is a key enabler for cost reduction in pharmaceutical intermediate manufacturing, as it removes the need for expensive safety mitigation systems.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
To fully appreciate the robustness of this synthetic route, one must examine the underlying catalytic cycle. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This step generates a crucial aryl-palladium intermediate, setting the stage for the subsequent carbonylation event. Unlike traditional methods where CO gas diffuses into the solution, here the carbon monoxide is released controllably from the decomposition of the trimesic acid phenol ester additive. This released CO then inserts into the aryl-palladium bond to form an acyl-palladium intermediate. This precise mechanism ensures that the concentration of free CO remains low, further enhancing safety while maintaining high reaction efficiency.
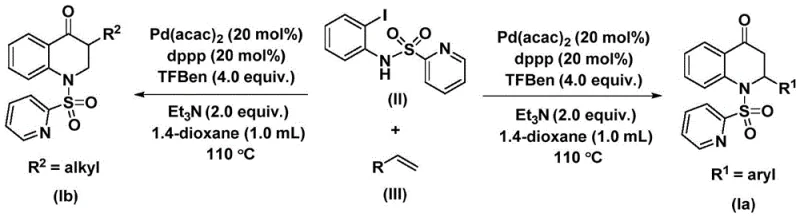
Following the carbonyl insertion, the olefin substrate coordinates with the palladium center and undergoes migratory insertion into the acyl-palladium bond. This step forms a new carbon-carbon bond and generates a palladium-alkyl intermediate. The cycle concludes with a reductive elimination step, which releases the final substituted 2,3-dihydroquinolone product and regenerates the active palladium(0) catalyst. This elegant cycle tolerates a wide range of functional groups on both the aniline and the olefin components. Whether the olefin bears electron-donating groups like methyl or methoxy, or electron-withdrawing groups like chlorine or fluorine, the catalytic system maintains high activity. This broad substrate scope is vital for medicinal chemists who need to rapidly explore structure-activity relationships (SAR) without being constrained by synthetic limitations.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
Implementing this synthesis in a practical setting requires adherence to specific parameters to maximize yield and purity. The process involves mixing the palladium catalyst, ligand, base, CO surrogate, and substrates in an aprotic organic solvent such as 1,4-dioxane. The reaction is typically heated to 110 °C for a duration of 48 hours to ensure complete conversion. Post-reaction processing is straightforward, involving filtration and standard column chromatography purification. For a detailed breakdown of the specific molar ratios, solvent volumes, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- Combine bis(acetylacetone)palladium, dppp ligand, triethylamine, 1,3,5-trimesic acid phenol ester (CO source), N-pyridine sulfonyl-o-iodoaniline, and the desired olefin in 1,4-dioxane solvent.
- Heat the reaction mixture to 110 °C and maintain stirring for 48 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, load onto silica gel, and purify via column chromatography to isolate the target substituted 2,3-dihydroquinolone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic methodology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the manufacturing environment. By eliminating the requirement for high-pressure carbon monoxide infrastructure, facilities can repurpose existing reactor trains for this chemistry, thereby increasing asset utilization and flexibility. This adaptability translates directly into enhanced supply chain reliability, as production is no longer bottlenecked by the availability of specialized high-pressure equipment or the scheduling constraints of hazardous gas delivery. Furthermore, the use of commercially available and inexpensive starting materials, such as simple olefins and readily synthesized iodoanilines, ensures a stable and cost-effective raw material supply base.
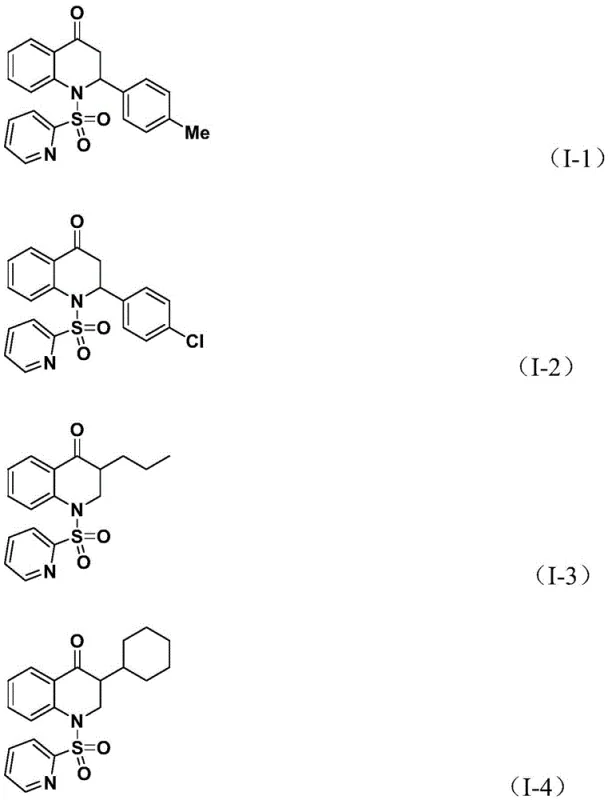
- Cost Reduction in Manufacturing: The economic implications of switching to a solid CO surrogate are profound. Traditional carbonylation processes often incur hidden costs related to safety compliance, gas monitoring, and specialized maintenance. By removing these overheads, the overall cost of production is significantly optimized. Additionally, the high reaction efficiency and good yields reported across various substrates mean less raw material waste and higher throughput per batch. The elimination of expensive transition metal scavengers, often required when using less selective catalysts, further contributes to substantial cost savings in the downstream purification stages.
- Enhanced Supply Chain Reliability: Supply continuity is paramount in the pharmaceutical industry. This method relies on reagents that are either commodity chemicals or easily synthesized from common precursors, reducing the risk of supply disruptions caused by niche reagent shortages. The robustness of the reaction conditions—tolerating air-sensitive steps better than some alternative low-valent metal protocols—means that batch failures due to minor procedural deviations are minimized. This reliability allows for more accurate forecasting and inventory planning, ensuring that downstream API synthesis schedules are met without delay.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is superior. The avoidance of toxic CO gas reduces the facility's environmental footprint and simplifies regulatory compliance. The reaction generates minimal hazardous waste compared to multi-step alternatives, aligning with green chemistry principles. The scalability is evidenced by the successful gram-level synthesis described in the patent, indicating a clear path to kilogram and ton-scale production. This ease of scale-up ensures that as demand for the final drug product grows, the supply of the intermediate can expand seamlessly without requiring fundamental process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a clear picture of what partners can expect when adopting this route for their own manufacturing needs.
Q: What is the primary safety advantage of this synthesis method?
A: This method utilizes 1,3,5-trimesic acid phenol ester as a solid carbon monoxide surrogate, eliminating the need for handling toxic and hazardous CO gas cylinders, thereby significantly improving operational safety in the laboratory and plant.
Q: What types of olefin substrates are compatible with this protocol?
A: The protocol demonstrates excellent substrate compatibility, successfully accommodating both aryl-substituted olefins (such as styrenes with various electron-donating or withdrawing groups) and alkyl-substituted olefins, allowing for diverse structural modifications.
Q: Is this process suitable for industrial scale-up?
A: Yes, the invention explicitly states that the method is simple to operate, uses cheap and easily obtainable raw materials, and has been validated for gram-level synthesis, providing a solid foundation for industrial large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
The technological advancements detailed in patent CN112239456B represent a significant opportunity for pharmaceutical companies to optimize their supply chains for bioactive heterocycles. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage this cutting-edge chemistry for your specific projects. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project phase. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 2,3-dihydroquinolone intermediate meets the highest quality standards required for GMP manufacturing.
We invite you to discuss how this innovative synthesis route can be integrated into your pipeline to achieve faster timelines and lower costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals in drug development and commercialization.
