Advanced One-Pot Synthesis of 5-Substituted 8-Acylaminoquinolines for Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic methodologies to access complex heterocyclic scaffolds efficiently, and patent CN106938984B presents a significant breakthrough in the synthesis of 5-substituted arylation and heterocycle 8-acylamino quinoline compounds. These quinoline derivatives serve as critical building blocks in the development of novel therapeutic agents, owing to their privileged structural status in medicinal chemistry. The disclosed technology introduces a streamlined one-pot preparation method that elegantly bypasses the traditional need for isolating unstable halogenated intermediates. By strategically employing a metal-free halogenation reaction as the initial step, the process ensures that the subsequent coupling reaction with boric acid compounds proceeds without interference from residual metal salts. This innovation not only enhances the overall yield and selectivity of the target products but also addresses key pain points in process chemistry regarding waste generation and operational complexity. For R&D directors and procurement managers alike, understanding the nuances of this tandem reaction sequence is vital for evaluating its potential integration into existing supply chains for high-value API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of selectively substituted quinoline derivatives has been fraught with challenges, particularly when attempting to introduce functional groups at the 5-position of the quinoline ring. Conventional strategies often rely on the cycloaddition of precursor anilines with carbonyl compounds or alkynes, a route that necessitates the prior preparation of expensive and often difficult-to-synthesize polysubstituted aniline precursors. Furthermore, traditional methods for direct C-H functionalization or halogenation of the quinoline nucleus frequently employ metal catalysts or metal salts as halogen sources, which can leave behind trace metal impurities. These residues are detrimental to downstream reactions, particularly palladium-catalyzed cross-couplings, as they can poison the active catalytic species or lead to unpredictable side reactions. The requirement for multiple isolation and purification steps between halogenation and coupling further exacerbates material loss, increases solvent consumption, and prolongs the overall production timeline, making these legacy processes economically unviable for large-scale commercial manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the methodology described in patent CN106938984B utilizes a clever one-pot tandem strategy that merges halogenation and arylation into a single continuous operation. The core innovation lies in the execution of the first halogenation step under metal-free conditions, utilizing reagents such as N-bromosuccinimide (NBS) or molecular halogens, which ensures that the reaction mixture remains free of transition metals that could interfere with the subsequent catalytic cycle. Once the halogenation is complete, the arylboronic acid, base, and a zero-valent metal catalyst (such as tetrakis(triphenylphosphine)palladium) are introduced directly into the same reactor without cooling or isolating the intermediate. This seamless transition allows for the immediate formation of the carbon-carbon bond at the 5-position, delivering the 5-substituted 8-acylamino quinoline product with impressive efficiency. 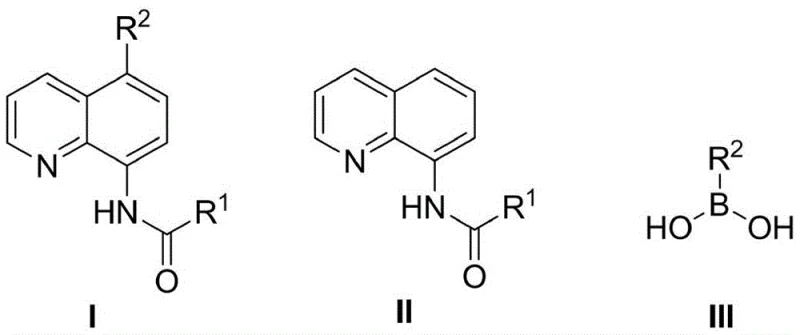
Mechanistic Insights into Metal-Free Halogenation and Suzuki-Miyaura Coupling
The mechanistic elegance of this process stems from the orthogonal compatibility of the two distinct reaction phases within a single pot. In the first phase, the electrophilic halogenation occurs selectively at the 5-position of the 8-acylaminoquinoline substrate, driven by the electronic activation provided by the nitrogen atom and the acylamino directing group. Because this step avoids the use of metal catalysts, there is no risk of generating metal-halide byproducts that typically complicate purification or deactivate sensitive catalysts in subsequent steps. The absence of metal ions during halogenation preserves the integrity of the reaction medium, creating an ideal environment for the introduction of the palladium or nickel catalyst in the second phase. Upon addition of the catalyst and the boronic acid coupling partner, the standard Suzuki-Miyaura catalytic cycle initiates, involving oxidative addition of the in-situ generated aryl halide to the metal center, transmetallation with the boronate species, and reductive elimination to forge the new C-C bond. This sequential yet integrated mechanism ensures high turnover numbers and minimizes the formation of homocoupling byproducts often seen in less optimized systems.
From an impurity control perspective, this one-pot design offers substantial advantages over stepwise approaches. In traditional multi-step syntheses, the isolation of the halogenated intermediate often leads to decomposition or the carryover of unreacted halogenating agents, which can degrade the catalyst in the next step. By maintaining the reaction in a closed system, the concentration of reactive species is managed dynamically, and the immediate consumption of the halogenated intermediate drives the equilibrium forward. Furthermore, the choice of solvents such as DMF or DMSO, which are effective for both the halogenation and the high-temperature coupling phases, ensures homogeneous reaction conditions throughout the process. This homogeneity is crucial for heat transfer and mass transfer in larger reactors, reducing the likelihood of hot spots that could lead to thermal degradation of the sensitive quinoline scaffold. The result is a cleaner crude reaction profile, which simplifies the final column chromatography or crystallization steps required to meet the stringent purity specifications demanded by pharmaceutical customers.
How to Synthesize 5-Substituted 8-Acylaminoquinoline Efficiently
The practical implementation of this synthesis route requires careful attention to reaction parameters to maximize yield and reproducibility. The process begins with the suspension of the 8-acylaminoquinoline starting material in a polar aprotic solvent, followed by the controlled addition of the halogenating agent under an inert nitrogen atmosphere to prevent oxidation of sensitive reagents. The mixture is then heated to temperatures ranging between 50°C and 160°C, depending on the reactivity of the specific substrate and halogen source, and maintained for a period of 10 to 24 hours to ensure complete conversion to the halogenated intermediate. Once the first stage is confirmed complete, the reaction mixture is cooled slightly, and the coupling partners—specifically the arylboronic acid, a mild inorganic base like sodium carbonate, and the phosphine-ligated palladium or nickel catalyst—are added directly. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined in the guide below.
- Initiate the reaction by combining the aminoquinoline substrate with a halogenating agent in a suitable organic solvent under inert atmosphere.
- Upon completion of the halogenation phase, directly introduce the arylboronic acid coupling partner, base, and transition metal catalyst into the same vessel.
- Maintain elevated temperatures to facilitate the cross-coupling reaction, followed by standard workup and chromatographic purification to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot synthetic methodology translates into tangible operational efficiencies and cost optimizations that extend beyond simple yield improvements. The elimination of the intermediate isolation step is a game-changer for manufacturing economics, as it removes the need for filtration, drying, and re-dissolution of the halogenated quinoline species. This reduction in unit operations directly correlates to a significant decrease in solvent usage, energy consumption for heating and cooling cycles, and labor hours required for handling materials. Consequently, the overall cost of goods sold (COGS) for these high-value pharmaceutical intermediates can be drastically reduced, offering a competitive pricing advantage in the global market. Moreover, the streamlined process reduces the physical footprint required for production, allowing manufacturers to increase batch sizes or throughput within existing facility constraints without the need for capital-intensive infrastructure upgrades.
- Cost Reduction in Manufacturing: The metal-free nature of the initial halogenation step eliminates the expense associated with purchasing and recovering expensive metal catalysts for that specific transformation. Furthermore, by avoiding the isolation of the intermediate, the process minimizes material losses that typically occur during transfer and purification stages, leading to a higher overall mass balance. The ability to use common, commodity-grade solvents like DMF and toluene throughout the entire sequence further simplifies solvent recovery and recycling protocols, contributing to substantial long-term cost savings in raw material procurement and waste disposal management.
- Enhanced Supply Chain Reliability: The robustness of this one-pot method enhances supply chain continuity by reducing the number of critical control points where production delays could occur. Traditional multi-step syntheses are vulnerable to bottlenecks at each isolation stage, where equipment availability or quality control hold-ups can stall the entire batch. By consolidating the synthesis into a single reactor train, the risk of such interruptions is minimized, ensuring more predictable lead times for the delivery of high-purity quinoline intermediates. Additionally, the broad substrate scope demonstrated in the patent suggests that the same platform technology can be adapted for various analogues, providing flexibility to respond quickly to changing customer demands for different substituted derivatives without requalifying entirely new processes.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the reduction in solvent volume and the avoidance of heavy metal waste streams align perfectly with modern green chemistry principles and increasingly stringent regulatory standards. The process generates less hazardous waste per kilogram of product, simplifying the compliance burden associated with wastewater treatment and solid waste disposal. This environmental efficiency not only mitigates regulatory risks but also enhances the sustainability profile of the supply chain, a factor that is becoming increasingly important for multinational pharmaceutical companies seeking eco-conscious partners. The scalability of the reaction conditions, which operate at moderate to high temperatures without requiring cryogenic conditions or ultra-high pressures, further facilitates a smooth transition from laboratory bench scale to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific project needs.
Q: What is the primary advantage of the metal-free halogenation step in this synthesis?
A: The initial halogenation step avoids the use of metal catalysts, preventing residual metal contamination that could poison the subsequent palladium or nickel catalyst used in the coupling phase, thereby ensuring higher reaction efficiency and yield.
Q: Can this one-pot method be scaled for industrial production of quinoline intermediates?
A: Yes, the elimination of intermediate isolation steps significantly simplifies the process flow, reducing solvent consumption and operational time, which makes it highly suitable for commercial scale-up in pharmaceutical manufacturing.
Q: What types of substituents are compatible with this synthetic route?
A: The method demonstrates broad substrate scope, tolerating various aryl and heteroaryl groups at the 5-position and diverse acyl groups at the 8-position, including electron-withdrawing and electron-donating substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Substituted 8-Acylaminoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN106938984B for accelerating drug discovery and development pipelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-substituted 8-acylaminoquinoline delivered meets the highest industry standards for potency and impurity profiles. We are committed to leveraging our technical expertise to optimize these one-pot processes further, ensuring maximum efficiency and cost-effectiveness for our global partners.
We invite R&D directors and procurement leaders to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this streamlined manufacturing process. We encourage you to contact us today to request specific COA data for our available quinoline derivatives and to schedule a consultation for comprehensive route feasibility assessments that will secure your supply chain for the future.
