Advanced One-Pot Synthesis of 5-Substituted 8-Acylaminoquinolines for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex heterocyclic scaffolds, particularly quinoline derivatives which serve as privileged structures in medicinal chemistry. Patent CN106938984B introduces a transformative approach to synthesizing 5-substituted arylation and heterocycle 8-acylamino quinoline compounds through a sophisticated one-pot tandem sequence. This methodology elegantly bypasses the traditional limitations of multi-step syntheses by integrating a metal-free halogenation reaction with a subsequent transition-metal catalyzed coupling, all performed without the isolation of intermediates. For research and development teams focused on API intermediate production, this represents a significant leap forward in process intensification, offering a route that minimizes waste generation while maximizing atom economy. The strategic design of this protocol ensures that the harsh conditions required for the initial functionalization do not compromise the integrity of the sensitive catalytic cycle required for the final carbon-carbon bond formation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of substituted quinoline frameworks has relied heavily on classical cycloaddition reactions involving precursor anilines and carbonyl compounds or alkynes, which often necessitate the preparation of expensive and highly substituted starting materials. These traditional routes are frequently plagued by poor regioselectivity, requiring extensive purification efforts to separate desired isomers from complex reaction mixtures. Furthermore, conventional methods for introducing aryl groups at the 5-position of the quinoline ring typically involve discrete, isolated steps where the halogenated intermediate must be purified before undergoing cross-coupling. This sequential approach not only increases the overall processing time and solvent consumption but also leads to substantial material loss during each isolation event. Additionally, many prior art methods utilize metal salts as halogen sources, which can leave residual metal contaminants that act as potent poisons for the palladium or nickel catalysts used in subsequent coupling reactions, drastically reducing overall yield and complicating downstream purification.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the technology disclosed in CN106938984B utilizes a streamlined one-pot strategy that seamlessly merges halogenation and arylation into a single operational unit. By employing common halides such as N-bromosuccinimide (NBS) or molecular iodine for the initial activation, the process avoids the introduction of interfering metal ions during the critical first stage. This allows the reaction mixture to proceed directly to the coupling phase upon the simple addition of the boronic acid, base, and catalyst, maintaining a continuous flow of chemical transformation. The ability to perform this sequence without intermediate work-up represents a paradigm shift in efficiency, effectively collapsing two distinct synthetic operations into one. This novel approach not only simplifies the operational workflow for chemists but also inherently reduces the environmental footprint of the manufacturing process by eliminating the need for multiple solvent exchanges and purification columns between steps.
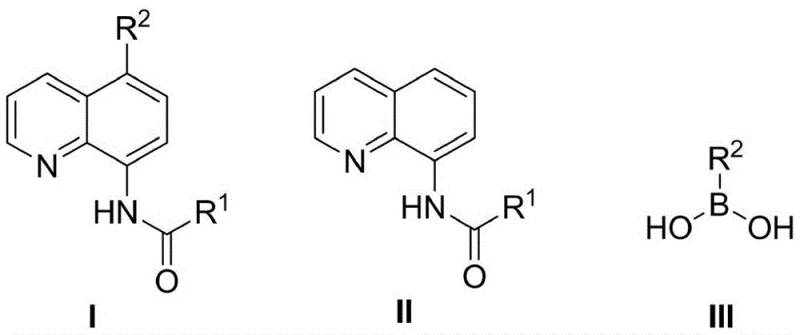
Mechanistic Insights into Metal-Free Halogenation and Pd/Ni-Catalyzed Coupling
The success of this tandem reaction relies heavily on the orthogonal compatibility of the two distinct mechanistic phases occurring within the same reactor vessel. The first phase involves an electrophilic aromatic substitution or radical halogenation mechanism where the electron-rich quinoline ring is selectively functionalized at the 5-position. Crucially, this step is designed to be metal-free, utilizing organic halides or elemental halogens that do not introduce transition metal residues. This is a vital design feature because trace amounts of certain metal salts from the halogenation step could coordinate strongly with the phosphine ligands of the palladium or nickel catalyst introduced in the second phase, rendering them inactive. By keeping the first step devoid of metals, the system preserves the catalytic activity required for the subsequent Suzuki-Miyaura type cross-coupling. The second phase then proceeds via the classic oxidative addition, transmetallation, and reductive elimination cycle typical of zero-valent palladium or nickel catalysis, efficiently forging the carbon-carbon bond between the quinoline core and the diverse array of aryl or heteroaryl boronic acids.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity pharmaceutical intermediates. Since the halogenation is highly selective for the 5-position due to the directing effects of the 8-acylamino group, the formation of regioisomeric byproducts is minimized. Furthermore, the one-pot nature means that any unreacted halogenating agent is consumed or quenched during the basic conditions of the coupling step, preventing the carryover of reactive halogen species into the final product. The use of robust catalysts such as tetrakis(triphenylphosphine)palladium or various nickel-phosphine complexes ensures that the coupling proceeds even in the presence of the polar solvents like DMSO or DMF used in the first step. This mechanistic robustness allows for a broader scope of substrates, including those with sensitive functional groups, to be tolerated without degradation, resulting in a cleaner crude reaction profile that facilitates easier final purification via column chromatography or crystallization.
How to Synthesize 5-Substituted 8-Acylaminoquinoline Efficiently
The practical execution of this synthesis is remarkably straightforward, making it highly attractive for both laboratory scale optimization and eventual commercial manufacturing. The process begins by charging a reaction vessel with the 8-acylaminoquinoline substrate and the chosen halogenating agent in a suitable polar aprotic solvent, followed by heating to initiate the halogenation. Once this initial transformation is complete, the reaction mixture is cooled slightly, and the coupling partners—specifically the arylboronic acid, an inorganic base like sodium carbonate, and the transition metal catalyst—are added directly to the pot. The detailed standardized synthesis steps, including specific molar ratios, temperature gradients, and reaction times optimized for different substituents, are outlined in the guide below to ensure reproducibility and maximum yield.
- Combine the 8-acylaminoquinoline substrate with a halogenating agent (such as NBS, NIS, or molecular halogens) in a polar aprotic solvent like DMF or DMSO.
- Heat the reaction mixture to temperatures between 50°C and 160°C for 10 to 24 hours to effect selective halogenation at the 5-position without metal catalysts.
- Without isolating the halogenated intermediate, directly add the arylboronic acid, a base such as sodium carbonate, and a zero-valent palladium or nickel catalyst to the same vessel.
- Continue heating the mixture at 50°C to 160°C for an additional 4 to 16 hours to complete the cross-coupling, followed by standard filtration and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot synthesis technology offers compelling economic and logistical benefits that extend far beyond simple yield improvements. By eliminating the isolation and purification of the halogenated intermediate, the process drastically reduces the total volume of solvents required and the associated costs of solvent recovery and disposal. This consolidation of steps also translates to a significant reduction in man-hours and equipment occupancy time, allowing manufacturing facilities to increase their throughput capacity without the need for additional capital investment in reactors. The reliance on commercially available and inexpensive reagents such as NBS, common boronic acids, and standard palladium or nickel catalysts ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or proprietary reagents. Furthermore, the robustness of the reaction conditions allows for greater flexibility in raw material sourcing, as the process can tolerate slight variations in reagent quality without compromising the final product specification.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the elimination of the intermediate isolation step, which traditionally accounts for a significant portion of manufacturing expenses due to solvent usage, filtration, and drying operations. By performing the reaction in a telescoped manner, the facility saves substantially on utility costs associated with heating and cooling cycles for multiple vessels, as well as the labor costs for handling and transferring materials between steps. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, reducing the burden on downstream purification units and increasing the overall recovery of the valuable active pharmaceutical ingredient intermediate. These cumulative efficiencies result in a leaner manufacturing process that delivers a lower cost of goods sold while maintaining high margins.
- Enhanced Supply Chain Reliability: The synthetic route utilizes commodity chemicals that are widely produced and available from multiple global suppliers, reducing the risk of supply disruptions caused by single-source dependencies. The tolerance of the reaction to various solvents, including DMF, DMSO, and toluene, provides procurement teams with the flexibility to switch solvents based on market availability and price fluctuations without needing to revalidate the entire process. Moreover, the stability of the reaction intermediates within the pot means that the process is less sensitive to minor delays or interruptions compared to multi-step sequences requiring the storage of unstable intermediates. This inherent robustness ensures a more predictable production schedule and reliable delivery timelines for downstream customers.
- Scalability and Environmental Compliance: Scaling this one-pot process to industrial levels is facilitated by the simplicity of the operation, which involves standard heating and stirring protocols compatible with existing glass-lined or stainless steel reactors. The reduction in solvent waste and the avoidance of heavy metal salts in the initial step align perfectly with increasingly stringent environmental regulations regarding waste discharge and heavy metal limits in pharmaceutical products. The process generates less hazardous waste per kilogram of product, simplifying the permitting process for new manufacturing lines and reducing the long-term liability associated with waste management. This green chemistry profile not only enhances the corporate sustainability image but also future-proofs the supply chain against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this specific quinoline synthesis pathway. These answers are derived directly from the experimental data and scope defined in the patent literature, providing a reliable foundation for process development decisions. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: Why is the metal-free halogenation step critical for this synthesis?
A: The initial halogenation is performed without metal catalysts to prevent the accumulation of residual metal salts that could poison the sensitive palladium or nickel catalysts required for the subsequent Suzuki-Miyaura type coupling step, thereby ensuring higher overall yields and cleaner reaction profiles.
Q: What types of substituents are compatible with this one-pot protocol?
A: The methodology demonstrates broad substrate scope, tolerating diverse R1 groups including alkyl, aryl, and heteroaryl moieties, as well as varied R2 substituents such as halophenyls, methoxyphenyls, and heterocycles like thienyl and pyridyl groups.
Q: Does this process require intermediate purification between steps?
A: No, the core innovation of this patent is the true one-pot nature of the reaction, allowing the halogenated intermediate to react directly with the boronic acid in the same vessel, which significantly reduces solvent usage and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Substituted 8-Acylaminoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation pharmaceuticals. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement complex one-pot tandem reactions like the one described in CN106938984B demonstrates our technical prowess in process intensification and our dedication to providing cost-effective solutions for our global partners.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this streamlined synthesis can optimize your budget. Please contact us today to request specific COA data for related quinoline derivatives and to discuss route feasibility assessments for your target molecules, ensuring a secure and efficient supply chain for your critical drug development programs.
