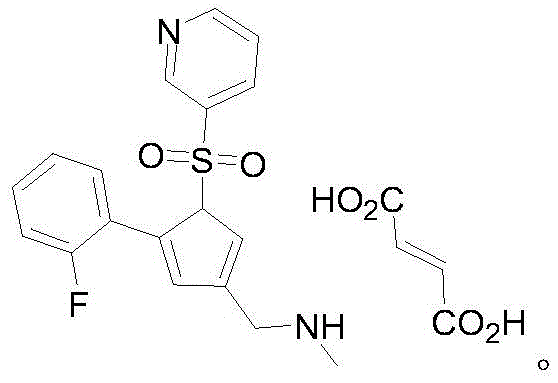
Revolutionizing Vonoprazan Manufacturing: A Safer, Scalable Synthetic Route for Global Supply Chains
The pharmaceutical industry is constantly seeking robust, scalable, and safe manufacturing processes for high-value Active Pharmaceutical Ingredients (APIs). Patent CN114539216A introduces a groundbreaking preparation method for Vonoprazan and its fumarate salt, addressing critical bottlenecks found in legacy synthetic routes. This innovation centers on the utilization of a novel intermediate, methyl (5-(2-fluorophenyl)-1-(pyridine-3-sulfonyl)-1H-pyrrol-3-yl)-pyridine-3-sulfonate, which serves as a pivotal precursor for efficient amination. By shifting away from hazardous redox chemistry and toxic methylation agents, this technology offers a pathway to higher yields and superior product quality. For R&D directors and procurement specialists, understanding this mechanistic shift is crucial for securing a reliable API intermediate supplier capable of meeting global demand without compromising on safety or regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Vonoprazan has been plagued by complex multi-step sequences that introduce significant operational risks and cost inefficiencies. As illustrated in prior art such as patent CN101300229A, traditional routes often involve the reduction of ester groups to hydroxyls followed by re-oxidation to aldehydes, creating a cumbersome cycle of functional group interconversions. 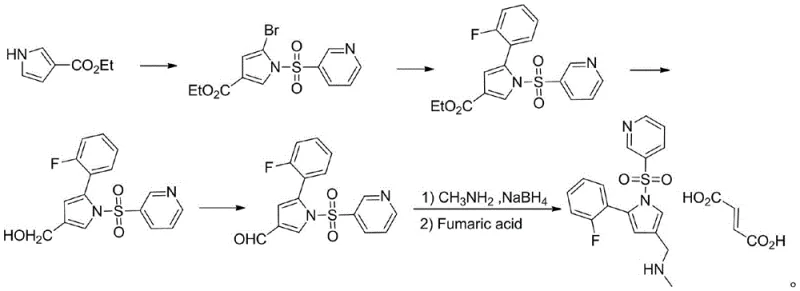 Furthermore, other documented methods, like those in CN105085484A, rely on the protection and deprotection of amine groups using tert-butoxycarbonyl (Boc) groups, which adds unnecessary steps and reduces overall atom economy.
Furthermore, other documented methods, like those in CN105085484A, rely on the protection and deprotection of amine groups using tert-butoxycarbonyl (Boc) groups, which adds unnecessary steps and reduces overall atom economy. 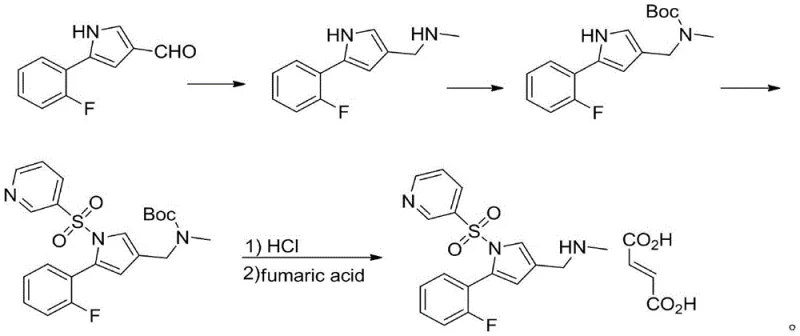 Perhaps most critically, routes described in CN104860923A necessitate the use of precious metal catalysts such as palladium, platinum, or rhodium for nitrile reduction, alongside formaldehyde for methylation.
Perhaps most critically, routes described in CN104860923A necessitate the use of precious metal catalysts such as palladium, platinum, or rhodium for nitrile reduction, alongside formaldehyde for methylation. 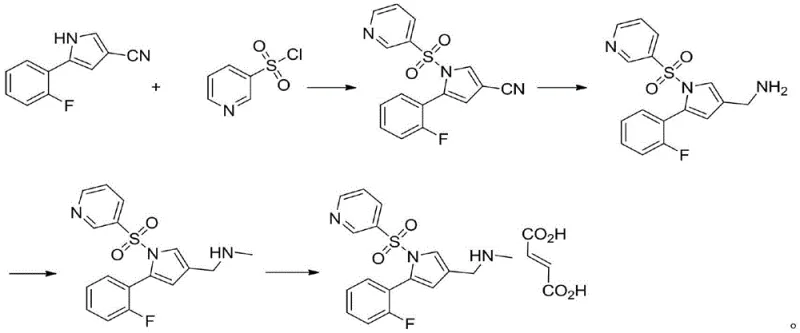 These reliance on heavy metals introduces the risk of residual metal contamination, while formaldehyde poses severe genotoxicity concerns, complicating purification and regulatory approval for cost reduction in pharmaceutical manufacturing.
These reliance on heavy metals introduces the risk of residual metal contamination, while formaldehyde poses severe genotoxicity concerns, complicating purification and regulatory approval for cost reduction in pharmaceutical manufacturing.
The Novel Approach
The methodology disclosed in CN114539216A represents a paradigm shift by streamlining the synthesis into a more direct and chemically elegant sequence. Instead of navigating through hazardous redox states or toxic alkylating agents, this novel approach leverages the high reactivity of a sulfonate ester intermediate. The process begins with the formation of methyl (5-(2-fluorophenyl)-1-(pyridine-3-sulfonyl)-1H-pyrrol-3-yl)-pyridine-3-sulfonate, which acts as an excellent electrophile. This intermediate undergoes a clean nucleophilic substitution with methylamine to directly install the required methylamino methyl group. 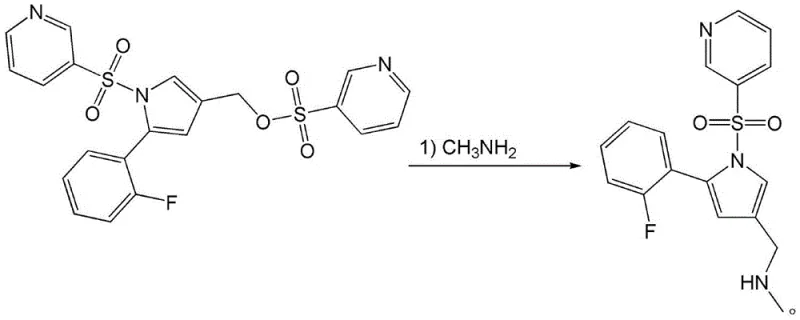 This strategy effectively bypasses the need for catalytic hydrogenation and formaldehyde usage, thereby eliminating entire classes of impurities. The subsequent salt formation with fumaric acid is straightforward, yielding high-purity Vonoprazan fumarate suitable for immediate downstream processing, demonstrating a clear advantage for the commercial scale-up of complex pharmaceutical intermediates.
This strategy effectively bypasses the need for catalytic hydrogenation and formaldehyde usage, thereby eliminating entire classes of impurities. The subsequent salt formation with fumaric acid is straightforward, yielding high-purity Vonoprazan fumarate suitable for immediate downstream processing, demonstrating a clear advantage for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Sulfonate Ester Aminolysis
The core chemical transformation in this patent relies on the nucleophilic substitution capability of the pyridine-3-sulfonate group. In organic synthesis, sulfonate esters are renowned for their superior leaving group ability compared to hydroxyl groups or halides in certain contexts. The electron-withdrawing nature of the pyridine ring attached to the sulfonyl group further activates the sulfur center, facilitating the displacement by the nucleophilic methylamine. This reaction proceeds under mild thermal conditions, typically ranging from -5°C to 40°C, which minimizes the formation of thermal degradation byproducts. The use of solvents like methanol or ethanol ensures good solubility of both the organic substrate and the amine reagent, promoting homogeneous reaction kinetics. This mechanistic simplicity is a key factor in achieving the high reaction efficiency noted in the patent data, as it reduces the likelihood of side reactions such as over-alkylation or polymerization that often plague more aggressive methylation protocols.
Impurity control is inherently built into this design by avoiding the introduction of extraneous reactive species. In traditional routes, the use of formaldehyde can lead to the formation of bis-methylated amines or Mannich base byproducts, which are difficult to remove and potentially genotoxic. Similarly, heavy metal catalysts can leach into the product stream, requiring expensive scavenging resins to meet ICH Q3D guidelines. By utilizing a sulfonate ester amination strategy, the impurity profile is significantly simplified. The primary byproducts are typically pyridine-3-sulfonic acid derivatives, which are polar and easily removed during the aqueous workup and crystallization steps. This inherent cleanliness reduces the burden on analytical QC labs and ensures that the final high-purity API intermediate meets stringent specifications without the need for extensive chromatographic purification, thereby enhancing supply chain reliability.
How to Synthesize Vonoprazan Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process is divided into the preparation of the key sulfonate intermediate followed by the amination and salt formation steps. Detailed standard operating procedures regarding stoichiometry, temperature control, and workup conditions are essential for replicating the high yields reported.
- React 5-(2-fluorophenyl)-1H-pyrrole-3-methanol with 3-pyridinesulfonyl chloride in acetonitrile using DMAP and triethylamine to form the key sulfonate ester intermediate.
- Perform nucleophilic substitution by reacting the sulfonate ester with methylamine in methanol at controlled temperatures (-5°C to 35°C) to yield Vonoprazan base.
- Dissolve the crude Vonoprazan in ethyl acetate, add fumaric acid, and crystallize the final Vonoprazan fumarate salt through cooling and filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The elimination of precious metal catalysts removes a significant variable cost and supply risk associated with fluctuating prices of palladium and platinum. Furthermore, avoiding genotoxic reagents like formaldehyde simplifies the environmental, health, and safety (EHS) compliance landscape, reducing the need for specialized containment equipment and hazardous waste disposal protocols. This translates directly into cost reduction in pharmaceutical manufacturing by lowering overheads related to safety management and waste treatment. The robustness of the reaction conditions also implies a wider operating window, which is critical for maintaining consistent batch-to-batch quality in large-scale reactors.
- Cost Reduction in Manufacturing: The streamlined nature of this process significantly lowers the cost of goods sold (COGS) by reducing the number of unit operations. By avoiding the need for expensive hydrogenation equipment and precious metal catalysts, capital expenditure (CAPEX) requirements are minimized. Additionally, the high atom economy of the direct amination step means less raw material is wasted as byproduct, leading to substantial cost savings on starting materials. The simplified purification process further reduces solvent consumption and energy usage during concentration and drying phases, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Dependence on specialized reagents like borane complexes or specific heterogeneous catalysts can create supply chain bottlenecks. This new route utilizes commodity chemicals such as methylamine, triethylamine, and common organic solvents like acetonitrile and ethyl acetate, which are readily available from multiple global suppliers. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Moreover, the stability of the sulfonate intermediate allows for potential stockpiling or semi-finished goods inventory strategies, providing greater flexibility in responding to market demand fluctuations for reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Industrial scalability is often hindered by exothermic hazards or difficult-to-control gas evolution. The reactions described in this patent operate under relatively mild thermal conditions with manageable exotherms, making them ideal for scaling from kilogram to tonnage production. The absence of heavy metal waste streams simplifies wastewater treatment and aligns with green chemistry principles, facilitating easier permitting and regulatory approval in environmentally sensitive jurisdictions. This environmental compatibility ensures long-term operational continuity and protects the manufacturer's reputation as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this specific preparation method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implementation of this technology.
Q: How does this new route improve safety compared to traditional methods?
A: This method eliminates the need for high-risk catalytic hydrogenation using expensive heavy metals like palladium or platinum, and avoids the use of genotoxic formaldehyde reagents for methylation, significantly reducing toxic impurity profiles.
Q: What are the key advantages for large-scale production?
A: The process features a shorter synthetic route with fewer unit operations, avoids complex oxidation-reduction steps, and utilizes readily available raw materials, making it highly suitable for industrial scale-up and cost-effective manufacturing.
Q: What is the purity profile of the final product?
A: Experimental data indicates that the final Vonoprazan fumarate obtained via this route achieves high purity levels (e.g., >99% HPLC purity) with simplified post-processing, ensuring compliance with stringent pharmaceutical quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vonoprazan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to stay competitive in the global API market. Our team of expert chemists has thoroughly analyzed the route disclosed in CN114539216A and validated its potential for robust commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of Vonoprazan meets stringent purity specifications required by major regulatory bodies worldwide.
We invite you to collaborate with us to leverage this innovative technology for your supply chain needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of Vonoprazan intermediates that drives efficiency and profitability for your organization.
