Advanced N-Acyl Protection Strategy for Commercial Scale Alfuzosin Manufacturing
The pharmaceutical industry continuously demands higher standards for Active Pharmaceutical Ingredients (APIs), particularly regarding impurity profiles and batch-to-batch consistency. Patent CN101687859A introduces a transformative approach to the synthesis of alfuzosin, a selective alpha-1 adrenergic receptor antagonist used for treating benign prostatic hyperplasia. This technology addresses critical bottlenecks in traditional manufacturing by utilizing novel N-acyl substituted intermediates. Unlike legacy processes that often result in complex impurity spectra requiring extensive purification, this method leverages a protective group strategy to achieve exceptional purity levels exceeding 99.9%. For R&D directors and procurement specialists, understanding this shift from direct synthesis to intermediate protection is vital for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent ICH guidelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of alfuzosin has relied on routes disclosed in earlier patents such as US 4,315,007, which involve the hydrogenation of nitrile precursors using Raney nickel followed by amidation. These conventional pathways suffer from significant drawbacks, including the generation of difficult-to-remove metal residues and the necessity of isolating the alfuzosin free base in solid form. The isolation of the free base is particularly problematic because it can introduce variability and potential degradation products if not handled under strictly controlled conditions. Furthermore, prior art methods like those in WO 2006/030449A1 often require excessive solvent usage and multi-step crystallizations to separate the base, leading to suboptimal yields and increased environmental waste. These inefficiencies create substantial cost reduction challenges in API manufacturing and complicate the supply chain continuity for global pharmaceutical companies.
The Novel Approach
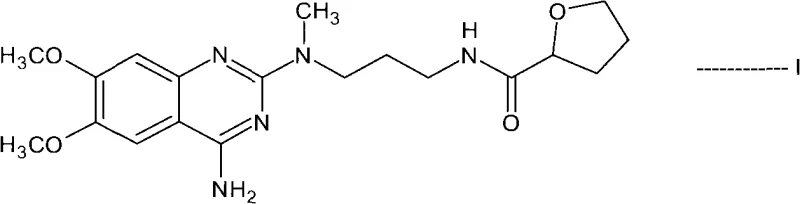
The innovative process described in CN101687859A circumvents these issues by introducing a transient N-acyl protection step. Instead of isolating the free base, the process generates the alfuzosin base in situ and immediately reacts it with an activating reagent to form a stable N-acyl derivative, designated as Formula II. This structural modification fundamentally alters the physicochemical properties of the molecule, allowing impurities to be washed away while the intermediate remains in solution or crystallizes in a highly pure form. By avoiding the isolation of the sensitive free base and utilizing robust acylation chemistry, the process achieves a streamlined workflow that is inherently safer and more efficient. This approach not only enhances the purity of the final product but also significantly simplifies the operational complexity required for commercial scale-up of complex quinazoline derivatives.
Mechanistic Insights into N-Acyl Protection and Deprotection
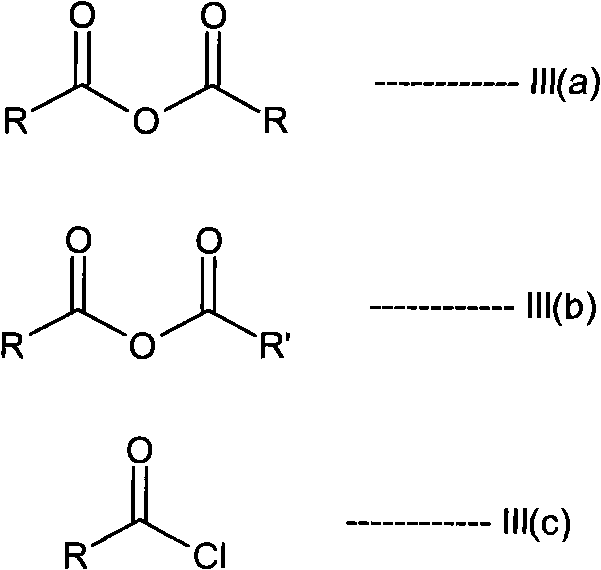
The core of this technology lies in the reversible acylation of the 4-amino group on the quinazoline ring. In the first stage, the primary amine of the alfuzosin free base acts as a nucleophile, attacking the carbonyl carbon of an acid anhydride or acid chloride. This reaction is typically catalyzed or facilitated by coupling agents like N,N-carbonyldiimidazole (CDI) during the initial amidation with tetrahydro-2-furoic acid. The resulting N-acyl intermediate (Formula II) is chemically stable enough to withstand purification procedures such as filtration and washing with anti-solvents like hexane or acetone. This stability is crucial because it allows manufacturers to physically separate the desired intermediate from unreacted starting materials, polymeric byproducts, and colored impurities that often plague direct synthesis routes.
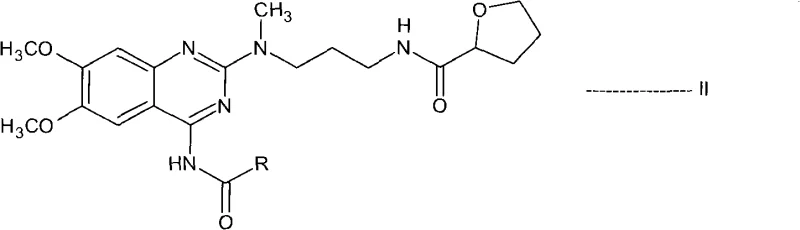
Following purification, the second mechanistic phase involves the hydrolytic removal of the acyl protecting group. This is achieved under acidic or basic conditions, preferably using methanolic hydrochloric acid, which cleaves the amide bond to regenerate the free 4-amino group. The choice of hydrolysis conditions is critical; the patent specifies temperatures ranging from 25°C to reflux, ensuring complete deprotection without damaging the sensitive tetrahydrofuran amide side chain. The final step involves the crystallization of the alfuzosin salt, typically the hydrochloride. Because the precursor intermediate was already purified to a high standard, the final crystallization serves primarily to establish the correct polymorphic form and remove residual solvents, rather than to remove gross chemical impurities. This two-stage mechanism ensures that the final API meets the rigorous purity specifications demanded by regulatory bodies.
How to Synthesize N-Acetyl Alfuzosin Efficiently
The synthesis of these high-value intermediates requires precise control over reaction stoichiometry and solvent systems to maximize yield and minimize waste. The patent outlines a robust protocol where the molar ratio of activating reagent to substrate is carefully optimized, typically ranging from 1 to 10 moles, with a preferred range of 2 to 5 moles to drive the reaction to completion. Solvent selection is equally critical, with dichloromethane and toluene emerging as preferred media due to their ability to dissolve both the polar intermediates and the non-polar impurities, facilitating effective separation.
- React the crude alfuzosin free base solution with an activating reagent such as acetic anhydride to form the N-acyl protected intermediate (Formula II).
- Isolate the N-acyl intermediate solid through cooling or anti-solvent addition, effectively separating it from unreacted starting materials and side products.
- Hydrolyze the purified intermediate using acidic conditions (e.g., methanolic HCl) to remove the protecting group and crystallize the final alfuzosin hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this N-acyl intermediate strategy offers profound logistical and economic benefits beyond simple chemical yield. The elimination of the solid free base isolation step removes a major bottleneck in the production schedule, reducing the overall cycle time and minimizing the risk of cross-contamination between batches. This streamlined workflow translates directly into enhanced supply chain reliability, as the process is less susceptible to variations in raw material quality or minor fluctuations in reaction parameters. Furthermore, the ability to purify the intermediate before the final step means that expensive downstream processing equipment is not burdened with low-quality feedstock, thereby extending equipment life and reducing maintenance downtime.
- Cost Reduction in Manufacturing: The process utilizes widely available and cost-effective reagents such as acetic anhydride and common organic solvents like dichloromethane and methanol. By avoiding the use of expensive transition metal catalysts like Raney nickel in the final stages and eliminating the need for complex chromatographic purification, the overall cost of goods sold is significantly reduced. The qualitative improvement in process efficiency means that less energy is consumed per kilogram of product, and the reduced solvent load lowers waste disposal costs, contributing to substantial cost savings in API manufacturing without compromising quality.
- Enhanced Supply Chain Reliability: The robustness of the acylation-hydrolysis cycle ensures consistent output even when scaling from pilot plants to multi-ton production facilities. Since the intermediate can be isolated and stored if necessary, it provides a buffer against supply disruptions, allowing manufacturers to build inventory of the purified precursor. This flexibility is essential for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream formulation teams receive their materials on schedule. The use of stable intermediates also simplifies logistics, as they are generally less hygroscopic and more stable during transport than the free base.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns well with green chemistry principles by reducing the total volume of solvents required and avoiding the generation of heavy metal waste. The crystallization steps are designed to be efficient, maximizing recovery and minimizing mother liquor waste. This makes the technology highly scalable, as the unit operations involved (reaction, filtration, distillation) are standard in the fine chemical industry. Consequently, manufacturers can confidently commit to long-term supply agreements, knowing that the process is environmentally compliant and capable of meeting the growing global demand for alfuzosin.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route. They are derived from the specific advantages and operational details outlined in the patent documentation, providing clarity on how this method compares to established industry standards.
Q: How does the N-acyl protection strategy improve alfuzosin purity compared to conventional methods?
A: Conventional methods often struggle with removing impurities from the free base. By converting the amine into an N-acyl derivative (Formula II), the physical properties such as solubility and crystallinity change significantly. This allows for effective purification of the intermediate before the final deprotection step, ensuring the final API exceeds 99.9% purity.
Q: Does this process require the isolation of solid alfuzosin free base?
A: No, a key advantage of this patented process is that it eliminates the need to isolate the solid alfuzosin free base. The free base is generated in situ and immediately reacted with the acylating agent, which simplifies the workflow and reduces potential degradation or contamination associated with handling the unstable free base.
Q: What activating reagents are preferred for industrial scalability?
A: The patent highlights the use of acid anhydrides, specifically acetic anhydride or propionic anhydride, as preferred activating reagents. These are cost-effective, easier to handle on a large scale compared to acid chlorides, and generate manageable byproducts, making them ideal for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alfuzosin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated N-acyl protection chemistry described in CN101687859A is executed with precision. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced HPLC and GC systems to verify that every batch of alfuzosin intermediate meets the >99.9% purity benchmark required for modern pharmaceutical applications.
We invite global pharmaceutical partners to collaborate with us to optimize their supply chains. By leveraging our technical capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of quality, reliability, and scientific excellence.
