Revolutionizing 10-Hydroxyphenanthrene Production via Green Electrochemical Oxidative Coupling
Introduction to Advanced Electrochemical Synthesis Technologies
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift towards sustainable manufacturing processes, driven by the urgent need to reduce environmental footprints and operational costs. A groundbreaking development in this sector is detailed in patent CN112126941B, which discloses a novel preparation method for polysubstituted 10-hydroxyphenanthrene derivatives. These structural motifs are critical building blocks found in numerous natural products with significant biological activities and are increasingly valuable in material science and organic synthesis ligand development. The core innovation lies in the utilization of electrochemical C-H/C-H oxidative dehydrogenation coupling, a technique that fundamentally reimagines how complex polycyclic aromatic hydrocarbons are constructed. By leveraging electrical energy to drive the reaction, this technology bypasses the traditional reliance on stoichiometric chemical oxidants, offering a cleaner, safer, and more atom-economical pathway for producing high-purity pharmaceutical intermediates.

This patent represents a significant leap forward for reliable pharmaceutical intermediate suppliers seeking to modernize their production capabilities. The method achieves the intramolecular oxidative coupling of substituted acetyl biphenyl derivatives under mild conditions, specifically utilizing a constant current in an undivided cell. This approach not only streamlines the synthetic route by reducing the number of steps but also ensures that the final products are free from heavy metal residues, a common pain point in conventional catalytic methods. For research and development teams focused on drug discovery, access to such diverse and cleanly synthesized scaffolds opens new avenues for exploring structure-activity relationships without the burden of complex purification protocols associated with transition metal removal.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 10-hydroxyphenanthrene skeleton has relied heavily on transition metal-catalyzed intramolecular or intermolecular ring closure reactions. These traditional pathways typically necessitate the use of expensive noble metals such as iridium, ruthenium, rhodium, and palladium, which pose significant economic and environmental challenges. Beyond the high cost of the catalysts themselves, these methods invariably require the addition of equivalent amounts of chemical oxidants like copper acetate, silver acetate, or tert-butyl hydroperoxide to regenerate the active catalytic species. The introduction of these stoichiometric oxidants inevitably leads to the generation of substantial quantities of toxic reaction by-products, complicating the downstream separation and purification processes. Furthermore, the use of oxygen gas as an oxidant in combination with flammable organic solvents at elevated temperatures introduces severe safety hazards, including the risk of fire and explosion, which are unacceptable in large-scale industrial settings.
The Novel Approach
In stark contrast, the electrochemical methodology described in the patent offers a transformative solution by replacing chemical oxidants with electrons, effectively using electricity as a traceless reagent. This novel approach avoids the use of noble metal catalysts entirely, thereby eliminating the risk of heavy metal contamination in the final product, which is a critical quality attribute for pharmaceutical applications. The reaction system is remarkably simple, employing a reticulated vitreous carbon (RVC) anode and a platinum cathode in an undivided cell, which significantly reduces equipment complexity and capital expenditure. By generating hydrogen gas as the sole by-product, the process aligns perfectly with the principles of green chemistry, minimizing waste treatment costs and environmental impact. This shift from chemical to electrochemical oxidation not only enhances the safety profile of the manufacturing process but also drastically simplifies the workup procedure, allowing for easier isolation of the target 10-hydroxyphenanthrene derivatives.
Mechanistic Insights into Electrochemical C-H/C-H Oxidative Coupling
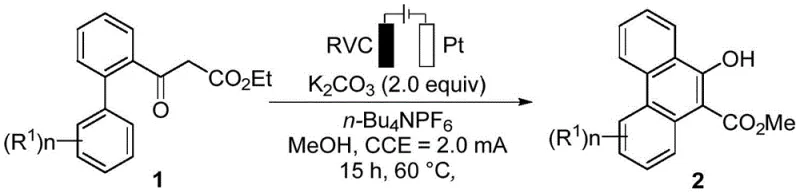
The mechanistic foundation of this synthesis relies on the direct functionalization of carbon-hydrogen bonds through anodic oxidation, a process that exhibits extremely high atom economy. In this electrochemical cell, the substituted acetyl biphenyl derivative undergoes oxidation at the anode surface, likely forming a radical cation intermediate that facilitates the subsequent intramolecular cyclization. The use of a supporting electrolyte, such as tetrabutylammonium hexafluorophosphate (n-Bu4NPF6), ensures sufficient conductivity within the solvent medium, while the base, typically potassium carbonate, plays a crucial role in deprotonation steps necessary for aromatization. The choice of solvent, often methanol or ethanol, is not merely for dissolution but can participate in the reaction dynamics, stabilizing intermediates and influencing the selectivity of the oxidative coupling. This precise control over the reaction environment allows for the efficient construction of the phenanthrene core without the need for pre-functionalized substrates, representing a significant advancement in C-H activation chemistry.
From an impurity control perspective, the electrochemical nature of the reaction provides a distinct advantage by avoiding the formation of metal-complexed side products that are difficult to remove. Traditional methods often suffer from incomplete catalyst turnover or non-selective oxidation by chemical reagents, leading to complex impurity profiles that require extensive chromatographic purification. In this electrochemical protocol, the potential applied across the electrodes can be finely tuned to selectively oxidize the substrate while leaving other sensitive functional groups intact. The absence of exogenous oxidants means there are no reduced forms of metal salts or peroxide decomposition products to contaminate the reaction mixture. Consequently, the crude reaction mixture is much cleaner, facilitating a straightforward purification process via silica gel column chromatography, which is essential for maintaining high throughput in a commercial manufacturing environment.
How to Synthesize 10-Hydroxyphenanthrene Derivatives Efficiently
The practical implementation of this synthesis route is designed for robustness and scalability, making it accessible for both laboratory research and industrial production. The general procedure involves mixing the electron-withdrawing group-substituted acetylbiphenyl derivative with the electrolyte and base in a suitable alcohol solvent, followed by electrolysis under a constant current mode. The reaction is typically conducted at a moderate temperature of 60°C for approximately 15 hours, ensuring complete conversion while maintaining energy efficiency. Detailed standardized synthesis steps, including specific molar ratios and purification parameters, are outlined in the technical guide below to ensure reproducibility and optimal yield for your specific substrate requirements.
- Mix the substituted acetyl biphenyl derivative substrate with electrolyte (n-Bu4NPF6) and base (K2CO3) in a suitable alcohol solvent.
- Install a reticulated vitreous carbon (RVC) anode and platinum (Pt) cathode in an undivided cell under inert gas protection.
- Apply a constant current of 2.0 mA at 60°C for 15 hours, then concentrate and purify the product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology presents a compelling value proposition centered on cost optimization and supply reliability. The elimination of noble metal catalysts removes a major variable cost driver and mitigates the supply chain risks associated with the fluctuating prices and geopolitical scarcity of metals like palladium and rhodium. Furthermore, the simplified purification process resulting from the absence of heavy metal contaminants reduces the consumption of silica gel and solvents during workup, leading to substantial cost savings in raw materials and waste disposal. The ability to produce high-purity intermediates with minimal downstream processing directly translates to shorter manufacturing cycles and improved overall equipment effectiveness, enhancing the agility of the supply chain to respond to market demands.
- Cost Reduction in Manufacturing: The replacement of expensive chemical oxidants and noble metal catalysts with electricity significantly lowers the direct material costs associated with production. By avoiding the need for specialized scavengers to remove trace metals from the final API or intermediate, manufacturers can achieve a leaner cost structure. The process utilizes commercially available and inexpensive reagents such as potassium carbonate and common alcohols, ensuring that the input costs remain stable and predictable. Additionally, the high atom economy of the reaction minimizes the loss of valuable starting materials, further contributing to the economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on electricity as the primary oxidant decouples the production process from the supply constraints of hazardous chemical oxidants, which often face strict transportation and storage regulations. The starting materials, substituted acetyl biphenyl derivatives, are readily accessible and can be synthesized through established pathways, ensuring a continuous and reliable feedstock supply. The robustness of the electrochemical cell setup, which does not require high-pressure equipment or inert gas manifolds beyond standard protection, simplifies facility requirements and reduces the risk of production downtime due to equipment failure or safety incidents.
- Scalability and Environmental Compliance: This method is inherently scalable, as electrochemical reactors can be easily expanded or operated in parallel to meet increasing production volumes without significant changes to the reaction chemistry. The generation of hydrogen as the only by-product simplifies waste management and ensures compliance with increasingly stringent environmental regulations regarding hazardous waste discharge. The absence of toxic heavy metals in the effluent stream reduces the burden on wastewater treatment facilities and lowers the environmental compliance costs, making this a sustainable choice for long-term commercial production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 10-hydroxyphenanthrene derivatives, based on the detailed specifications and experimental data provided in the patent literature. These insights are intended to clarify the operational parameters and benefits for potential partners evaluating this technology for their supply chain integration. Understanding these nuances is critical for assessing the feasibility of adopting this green chemistry approach in your existing manufacturing infrastructure.
Q: What are the primary advantages of this electrochemical method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic noble metal catalysts like palladium or rhodium, and replaces hazardous chemical oxidants with electricity, producing only hydrogen as a byproduct.
Q: Is this synthesis protocol suitable for large-scale industrial production?
A: Yes, the process uses simple starting materials, avoids heavy metal contamination which simplifies purification, and generates minimal waste, making it highly suitable for commercial scale-up.
Q: What types of substituents are tolerated on the biphenyl substrate?
A: The reaction demonstrates a wide substrate scope, tolerating various electron-withdrawing groups such as esters, cyano, halogens, and alkyl groups at multiple positions on the aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 10-Hydroxyphenanthrene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in delivering high-value pharmaceutical intermediates with superior purity and sustainability profiles. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112126941B can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 10-hydroxyphenanthrene derivative meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with our technical team to explore how this cost-effective and environmentally friendly synthesis route can enhance your product portfolio. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your project scale. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in advanced chemical manufacturing and supply chain optimization.
