Revolutionizing 1,2-Disubstituted-4-Quinolone Production: A Metal-Free, High-Yield Synthetic Route for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for synthesizing critical heterocyclic scaffolds. Patent CN106892866B introduces a groundbreaking methodology for the preparation of 1,2-disubstituted-4-quinolones, a class of compounds renowned for their broad-spectrum antibacterial activity and utility in material science applications such as luminescent materials. Unlike conventional approaches that rely heavily on transition metal catalysis, this invention utilizes a direct condensation strategy between substituted acyl chlorides and N-substituted o-aminoacetophenones. This shift represents a significant technological leap, offering a robust alternative that eliminates the need for expensive noble metal catalysts like palladium or copper, thereby streamlining the production workflow and enhancing the environmental profile of the manufacturing process.
Historically, the synthesis of 1,2-disubstituted-4-quinolones has been plagued by significant operational challenges and economic inefficiencies. Prior art methods, such as those reported by Bernini et al. and Xu Bin, often necessitate the use of complex catalytic systems involving PdCl2(PPh3)2/CuI or palladium acetate, which not only inflate raw material costs but also introduce stringent regulatory hurdles regarding heavy metal residue limits in final API intermediates. Furthermore, alternative routes utilizing n-BuLi require cryogenic conditions down to -78°C, posing severe safety risks due to the pyrophoric nature of the reagent and demanding specialized, energy-intensive equipment. These conventional methodologies are characterized by cumbersome multi-step sequences, low atom economy, and harsh reaction environments that limit their feasibility for large-scale commercial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on precious metal catalysts in traditional quinolone synthesis creates a bottleneck for both cost reduction and supply chain reliability. The removal of trace palladium or copper from the final product often requires additional purification steps, such as scavenging resins or extensive chromatography, which drastically reduces overall throughput and increases waste generation. Additionally, the use of unstable intermediates like alkynones synthesized under cryogenic conditions introduces significant variability and safety hazards into the production line. The high energy consumption associated with maintaining sub-zero temperatures and the volatility of reagents like phenylacetylene further exacerbate the operational expenditures, making these legacy processes increasingly uncompetitive in a market that demands greener and leaner manufacturing solutions.
The Novel Approach
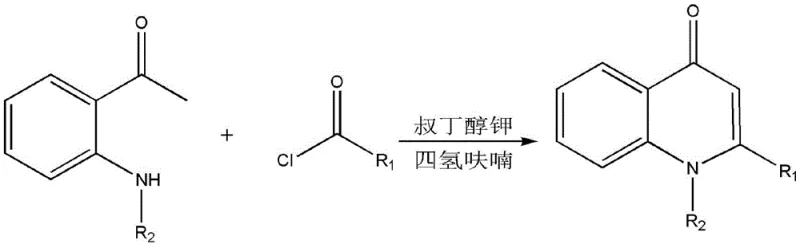
The methodology disclosed in CN106892866B fundamentally reimagines the construction of the quinolone core by leveraging a base-mediated intramolecular Claisen condensation. By reacting readily available substituted acyl chlorides with N-substituted o-aminoacetophenones in the presence of a strong base like potassium tert-butoxide (BuOK), the process achieves cyclization under remarkably mild conditions ranging from 25°C to 60°C. This approach completely bypasses the need for transition metals and cryogenic cooling, utilizing stable, commodity-grade starting materials instead. The reaction proceeds through a nucleophilic acyl substitution followed by ring closure, delivering high-purity products with exceptional yields, as evidenced by the 92% yield obtained for p-chlorophenyl derivatives. This streamlined protocol not only simplifies the operational workflow but also aligns perfectly with modern principles of green chemistry.
Mechanistic Insights into Base-Mediated Cyclization
The mechanistic pathway of this novel synthesis is elegantly straightforward yet highly effective, driven by the inherent reactivity of the amine and ketone functionalities within the precursors. Initially, the lone pair of electrons on the nitrogen atom of the N-substituted o-aminoacetophenone performs a nucleophilic attack on the carbonyl carbon of the substituted acyl chloride. This step forms a tetrahedral intermediate which subsequently collapses to eliminate a molecule of hydrogen chloride, yielding a stable beta-keto amide intermediate. This acylation step is rapid and exothermic, setting the stage for the crucial ring-closing event. The choice of solvent, specifically anhydrous tetrahydrofuran (THF), plays a pivotal role in stabilizing the ionic species involved and ensuring the solubility of both organic reactants and the inorganic base.
Upon the addition of the strong base, typically BuOK, the mechanism shifts to an intramolecular Claisen condensation. The base abstracts the acidic alpha-proton adjacent to the ketone carbonyl group, generating a resonance-stabilized enolate carbanion. This nucleophilic center then attacks the amide carbonyl carbon within the same molecule, forming a new carbon-carbon bond and closing the six-membered quinolone ring. The subsequent elimination of a water molecule aromatizes the system, driving the equilibrium towards the formation of the final 1,2-disubstituted-4-quinolone product. This cascade occurs efficiently at moderate temperatures (25-60°C), minimizing thermal degradation and side reactions such as polymerization or hydrolysis, which are common pitfalls in high-temperature reflux protocols. The result is a clean reaction profile that facilitates easy downstream processing.
How to Synthesize 1,2-Disubstituted-4-Quinolone Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, precise control over stoichiometry and reaction conditions is essential to maximize yield and purity. The process begins with the in situ or separate preparation of the acyl chloride from the corresponding carboxylic acid using thionyl chloride, followed by the N-alkylation of o-aminoacetophenone. Once the key precursors are ready, they are combined in anhydrous THF under an inert atmosphere. The addition of the base must be controlled to manage the exotherm, and the reaction mixture is maintained at the optimal temperature range until conversion is complete as monitored by TLC. Detailed standardized operating procedures regarding workup, including extraction with dichloromethane and drying over magnesium sulfate, ensure consistent recovery of the pale yellow solid product.
- Preparation of Substituted Acyl Chloride: React substituted carboxylic acid with excess thionyl chloride (SOCl2) under reflux for 1-3 hours, followed by distillation to remove excess reagent.
- N-Alkylation of o-Aminoacetophenone: React o-aminoacetophenone with potassium carbonate and an alkyl halide (e.g., allyl bromide) in DMF at 50-80°C for 22-26 hours to form the N-substituted precursor.
- Cyclization Reaction: Dissolve the acyl chloride in anhydrous THF, add the N-substituted o-aminoacetophenone, and stir. Add a strong base like potassium tert-butoxide (BuOK) at 25-60°C to induce intramolecular Claisen condensation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthetic route offers transformative benefits that directly impact the bottom line and operational resilience. By eliminating the dependency on volatile precious metal markets for palladium and copper catalysts, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing. The removal of heavy metal catalysts also obviates the need for expensive metal scavenging agents and complex purification protocols, thereby shortening the production cycle time and reducing solvent consumption. Furthermore, the use of stable, non-pyrophoric reagents like acyl chlorides and BuOK enhances workplace safety and reduces the insurance and compliance costs associated with handling hazardous cryogenic materials like n-BuLi.
- Cost Reduction in Manufacturing: The exclusion of noble metal catalysts represents a direct saving on raw material costs, which is particularly impactful given the fluctuating prices of palladium and rhodium. Moreover, the simplified workup procedure, which avoids specialized metal removal steps, reduces labor hours and consumable usage. The high atom utilization rate of this reaction means less waste is generated per kilogram of product, lowering waste disposal fees and improving the overall process mass intensity (PMI). These factors combine to create a substantially more economical production model compared to legacy transition-metal catalyzed routes.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as substituted benzoic acids and o-aminoacetophenones, are commodity chemicals with robust global supply chains, unlike specialized organometallic reagents which may face availability bottlenecks. The mild reaction conditions (25-60°C) allow the process to be run in standard glass-lined or stainless steel reactors without the need for specialized cryogenic cooling infrastructure. This flexibility ensures that production can be scaled up or shifted between facilities with minimal capital investment, guaranteeing continuity of supply even during market disruptions.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns with strict regulatory standards by avoiding toxic heavy metals in the final product, simplifying the regulatory filing process for new drug applications. The absence of explosive or highly flammable reagents reduces the risk profile of the manufacturing site, facilitating easier permitting and inspection outcomes. The ability to operate at near-ambient temperatures also translates to lower energy consumption for heating and cooling, contributing to a reduced carbon footprint and supporting corporate sustainability goals without compromising on output quality or volume.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a clear picture of the method's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating process transfer and for procurement specialists assessing vendor qualifications.
Q: Why is this metal-free synthesis method superior to traditional palladium-catalyzed routes?
A: Traditional methods often require expensive palladium or copper catalysts and harsh conditions like cryogenic temperatures (-78°C) with n-BuLi. This novel route operates at mild temperatures (25-60°C) without noble metals, significantly reducing raw material costs and eliminating the complex purification steps required to remove heavy metal residues, which is critical for pharmaceutical compliance.
Q: What yields can be expected from this synthetic pathway?
A: The patent data demonstrates excellent efficiency across various substrates. For instance, the synthesis of the p-chlorophenyl derivative achieved a yield of 92%, while the 2-iodophenyl variant reached 84%. Overall, the method consistently delivers high atom utilization and yields ranging typically from 72% to over 90% depending on the specific substituents.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable. It avoids hazardous reagents like phenylacetylene and pyrophoric n-BuLi, using stable acyl chlorides and simple bases instead. The mild reaction conditions (25-60°C) and the use of common solvents like THF make it safe and economically viable for commercial scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Disubstituted-4-Quinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the potential of the metal-free cyclization route described in CN106892866B and is fully prepared to leverage this technology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including rigorous moisture control for anhydrous reactions and stringent purity specifications to meet the highest pharmacopeial standards.
We invite you to collaborate with us to optimize your supply chain for quinolone-based intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to this efficient route can improve your margins. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us help you secure a reliable, cost-effective, and compliant supply of high-purity 1,2-disubstituted-4-quinolones for your next generation of therapeutic or material science applications.
