Advanced Asymmetric Synthesis of Axially Chiral Aryl Indoles for High-Value Pharmaceutical Intermediates
Advanced Asymmetric Synthesis of Axially Chiral Aryl Indoles for High-Value Pharmaceutical Intermediates
The pharmaceutical industry is constantly seeking robust methodologies to construct complex chiral scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN110467555B introduces a groundbreaking approach to synthesizing axially chiral aryl indole compounds, specifically targeting the indole-naphthalene and indole-benzene structural motifs. These molecules are not merely academic curiosities; they represent a class of potent bioactive agents with demonstrated efficacy against MCF-7 breast cancer cells. The innovation lies in the utilization of a dynamic kinetic resolution strategy mediated by chiral phosphoric acid organocatalysts. This technology allows for the direct conversion of readily available racemic precursors into high-value, optically pure intermediates with exceptional efficiency. By leveraging this patented methodology, manufacturers can access a versatile library of compounds defined by Formula 3 and Formula 5, opening new avenues for drug discovery programs focused on oncology and other therapeutic areas.
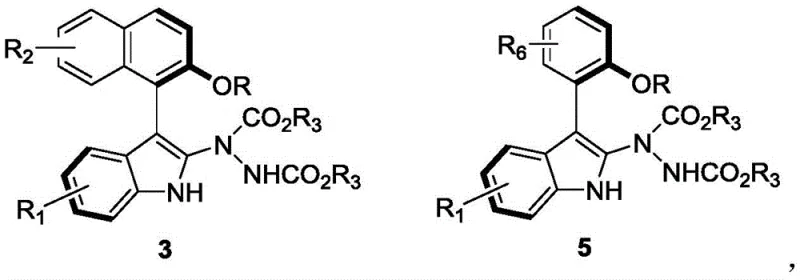
For procurement specialists and supply chain managers, the implications of this technology are profound. The reliance on organocatalysis rather than precious metal complexes fundamentally alters the cost structure and regulatory burden associated with API intermediate manufacturing. The process eliminates the need for expensive palladium or rhodium catalysts and the subsequent rigorous purification steps required to remove trace heavy metals to ppm levels. Furthermore, the reaction proceeds under ambient conditions, drastically reducing energy consumption compared to traditional cryogenic asymmetric syntheses. This combination of operational simplicity, high atom economy, and superior stereocontrol positions this synthetic route as a highly attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios while maintaining stringent quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral biaryl systems containing indole moieties has been fraught with significant synthetic challenges. Traditional approaches often rely on cross-coupling reactions, such as Suzuki-Miyaura or Ullmann couplings, which typically require harsh reaction conditions, including elevated temperatures and strong bases. These conditions can be detrimental to sensitive functional groups often present in complex drug candidates, leading to decomposition or side reactions that complicate downstream purification. Moreover, achieving high axial chirality through these conventional means usually necessitates the use of chiral auxiliaries or resolution of racemates, both of which suffer from inherent inefficiencies. Resolution methods, in particular, are limited by a maximum theoretical yield of 50%, resulting in substantial material waste and increased production costs. Additionally, the use of transition metal catalysts introduces the risk of metal contamination, a critical quality attribute that is heavily regulated in pharmaceutical manufacturing, thereby necessitating costly scavenging processes and extensive analytical testing.
The Novel Approach
In stark contrast, the methodology disclosed in CN110467555B offers a paradigm shift by employing a chiral phosphoric acid-catalyzed nucleophilic addition to azo compounds. This novel approach enables the direct assembly of the axially chiral skeleton from racemic 3-substituted indoles and azodicarboxylates in a single operational step. The reaction is characterized by its remarkable mildness, proceeding efficiently at temperatures ranging from 20 to 30°C, with 25°C being the optimal setpoint. This ambient temperature operation not only enhances safety by eliminating thermal hazards but also preserves the integrity of sensitive substrates. The use of dichloromethane as a solvent ensures excellent solubility for the organic components while facilitating easy workup procedures. Most critically, this dynamic kinetic resolution process theoretically allows for yields approaching 100%, effectively doubling the material efficiency compared to classical resolution techniques. The result is a streamlined, high-yielding process that delivers products with outstanding optical purity, as evidenced by enantiomeric ratios reaching up to 98:2 in optimized examples.
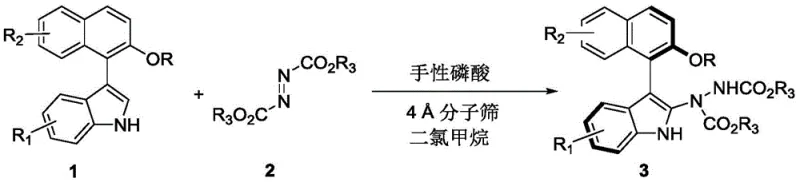
Mechanistic Insights into Chiral Phosphoric Acid-Catalyzed Asymmetric Addition
The success of this synthetic transformation hinges on the precise molecular recognition and activation capabilities of the chiral phosphoric acid catalyst, specifically the BINOL-derived derivative designated as Formula 61. This catalyst functions through a dual hydrogen-bonding activation mechanism. The acidic proton of the phosphoric acid moiety activates the electrophilic azo compound by forming a hydrogen bond with one of the nitrogen atoms, thereby lowering the LUMO energy of the acceptor. Simultaneously, the phosphoryl oxygen acts as a Lewis base, coordinating with the nucleophilic indole substrate via hydrogen bonding interactions. This bifunctional activation brings the reactants into close proximity within a well-defined chiral pocket created by the bulky 2,4,6-triisopropylphenyl groups at the 3,3'-positions of the BINOL backbone. This steric environment strictly controls the trajectory of the nucleophilic attack, ensuring that the reaction proceeds exclusively through one enantiofacial pathway. The result is the highly stereoselective formation of the new C-N bond and the concomitant establishment of the stable axial chirality between the indole and the naphthalene or benzene ring.
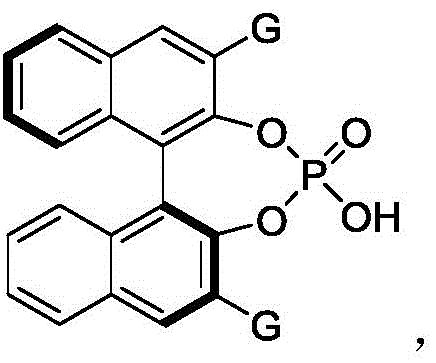
From an impurity control perspective, this organocatalytic mechanism offers distinct advantages over metal-catalyzed alternatives. Since the reaction does not involve radical intermediates or oxidative addition/reductive elimination cycles typical of transition metals, the formation of homocoupling byproducts or metal-induced degradation products is minimized. The primary impurities observed are typically unreacted starting materials or diastereomers arising from minor competing pathways, both of which are readily removed via standard silica gel column chromatography using petroleum ether and ethyl acetate mixtures. The high selectivity of the catalyst means that the crude reaction mixture is already enriched in the desired enantiomer, reducing the burden on final purification steps. This clean reaction profile is essential for maintaining a robust impurity fingerprint, a key requirement for regulatory filings and ensuring the consistency of the final active pharmaceutical ingredient. The stability of the catalyst under the reaction conditions further contributes to process reliability, preventing catalyst decomposition that could lead to colored impurities or difficult-to-remove residues.
How to Synthesize Axially Chiral Aryl Indoles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction monitoring to ensure reproducibility. The protocol is designed to be user-friendly, utilizing standard glassware and common laboratory techniques. The key to success lies in the activation of the molecular sieves to ensure effective water scavenging, as moisture can deactivate the chiral phosphoric acid catalyst and hydrolyze the azo reagent. The reaction progress should be closely monitored using thin-layer chromatography (TLC) to determine the optimal endpoint, preventing over-reaction or decomposition. Once the reaction is deemed complete, the workup procedure is straightforward, involving simple filtration to remove the solid additives followed by solvent evaporation. The resulting crude product can then be purified to pharmaceutical grade using flash chromatography. For detailed operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining the racemic indole substrate (Formula 1 or 4) and the azo compound (Formula 2) in dichloromethane solvent.
- Add 4 Angstrom molecular sieves as an additive and introduce the chiral phosphoric acid catalyst (Formula 61) at a loading of 10 mol%.
- Stir the reaction at 25°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chiral phosphoric acid catalyzed route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The shift from metal-based catalysis to organocatalysis represents a strategic move towards greener and more sustainable manufacturing practices, which aligns with the increasing environmental, social, and governance (ESG) mandates of major pharmaceutical companies. By eliminating the need for precious metals, the process removes a significant variable cost driver and mitigates the supply risk associated with fluctuating prices of commodities like palladium or platinum. Furthermore, the simplified downstream processing reduces the consumption of specialized scavenging resins and the generation of hazardous metal-containing waste streams, leading to substantial cost savings in waste disposal and environmental compliance. The mild reaction conditions also translate to lower energy overheads, as there is no requirement for energy-intensive heating or cryogenic cooling systems, making the process inherently more scalable and energy-efficient.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy and the use of commercially available, inexpensive starting materials. The dynamic kinetic resolution nature of the reaction ensures that the theoretical yield is not capped at 50%, effectively maximizing the output per batch and reducing the cost of goods sold (COGS). Additionally, the catalyst loading is relatively low (10 mol%), and the catalyst itself can potentially be recovered or recycled in optimized continuous flow setups, further driving down unit costs. The avoidance of expensive chiral ligands and metal salts simplifies the raw material inventory, allowing procurement teams to source reagents from multiple qualified vendors, thereby enhancing supply security and negotiating leverage.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain resilience. Operating at ambient temperature (25°C) minimizes the risk of thermal runaways and equipment failures that can disrupt production schedules. The tolerance of the catalyst to various functional groups on the indole and naphthalene rings allows for a flexible substrate scope, meaning the same core process can be adapted to produce a wide range of analogues without extensive re-optimization. This flexibility enables manufacturers to respond quickly to changing demands from R&D departments for diverse compound libraries. Moreover, the use of standard solvents like dichloromethane ensures that solvent supply chains remain stable and unaffected by niche chemical shortages, guaranteeing consistent production throughput.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or tonne scale is facilitated by the absence of exothermic hazards and the simplicity of the workup. The filtration of molecular sieves and silica gel purification are unit operations that are easily translated to large-scale equipment. From an environmental standpoint, the process generates significantly less hazardous waste compared to traditional methods. The elimination of heavy metals simplifies the environmental impact assessment and reduces the regulatory burden associated with discharge permits. This "green" profile not only lowers compliance costs but also enhances the marketability of the final intermediates to environmentally conscious clients who prioritize sustainable sourcing in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this axially chiral synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of incorporating these intermediates into their drug discovery pipelines and for quality assurance teams establishing specification limits.
Q: What is the primary advantage of using chiral phosphoric acid catalysts for this synthesis?
A: The primary advantage is the ability to achieve high enantioselectivity (er values up to 98:2) under extremely mild conditions (25°C) without the need for toxic transition metals, ensuring a cleaner impurity profile suitable for pharmaceutical applications.
Q: Can this synthetic method be scaled for industrial production?
A: Yes, the method utilizes common solvents like dichloromethane and operates at room temperature, which significantly simplifies thermal management and safety protocols required for large-scale commercial manufacturing compared to cryogenic processes.
Q: What biological activity do these axially chiral compounds exhibit?
A: Biological testing indicates that these compounds possess cytotoxic activity against MCF-7 breast cancer cells, with specific derivatives showing IC50 values as low as 4.44 μg/mL, highlighting their potential as oncology drug leads.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Aryl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in accelerating the development of life-saving medicines. Our technical team has thoroughly analyzed the synthetic route described in CN110467555B and is fully prepared to support your projects with custom manufacturing services. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including advanced chiral HPLC analysis to guarantee the enantiomeric excess required for your clinical trials. We understand that every project is unique, and our engineers are ready to optimize the chiral phosphoric acid catalysis parameters to suit your specific volume and timeline requirements.
We invite you to collaborate with us to leverage this innovative technology for your next drug candidate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are eager to provide you with specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise in asymmetric organocatalysis can drive value and efficiency in your supply chain. Let us be your partner in transforming complex chemical challenges into commercial successes.
