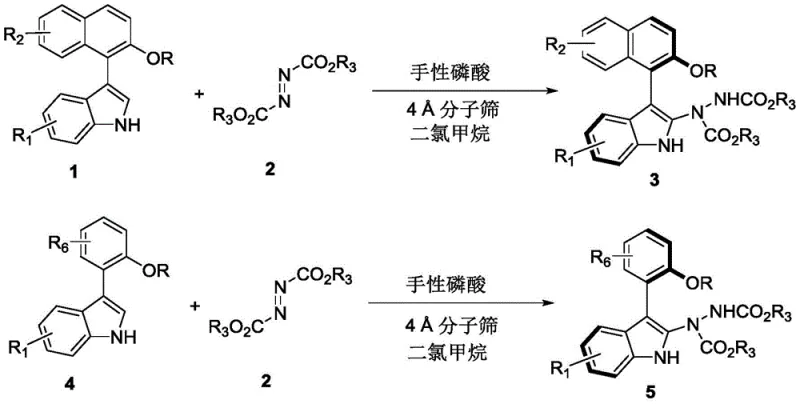
Advanced Catalytic Strategy for Commercial Scale-up of Complex Axially Chiral Indole Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access structurally complex chiral scaffolds, particularly those exhibiting axial chirality which are pivotal in modern drug discovery. A significant breakthrough in this domain is documented in Chinese Patent CN110467555B, which discloses a highly efficient synthetic route for axially chiral aryl indole compounds. This technology addresses a critical gap in organic synthesis by providing a practical method to construct indole-naphthalene and indole-benzene skeletons with exceptional optical purity. For R&D directors and procurement specialists alike, this patent represents a transformative approach to generating high-value intermediates that serve as potential glucocorticoid receptor antagonists and HCV NS5B polymerase inhibitors. The methodology leverages dynamic kinetic resolution strategies, a field previously underexplored for this specific class of molecules, thereby opening new avenues for the development of potent bioactive agents.
Historically, the construction of axially chiral indole frameworks has been fraught with challenges, primarily relying on coupling reactions between indole rings and naphthalene or benzene rings which often suffer from limited substrate scope and harsh reaction conditions. Prior art, such as methods reported in Angewandte Chemie and Nature Chemistry, utilized chiral phosphoric acids to catalyze couplings involving 2-indolecarbinols or azonaphthalenes, yet these approaches remained restrictive in terms of structural diversity and operational simplicity. The conventional pathways frequently necessitated specialized precursors that are difficult to source commercially, leading to supply chain bottlenecks and inflated costs for research-grade materials. Furthermore, achieving high enantioselectivity in these traditional coupling protocols often required rigorous temperature control and extended reaction times, complicating the scale-up process for industrial applications.
In stark contrast, the novel approach detailed in the patent utilizes a direct asymmetric addition reaction between readily available indole derivatives and azo compounds, catalyzed by a specific chiral phosphoric acid. This strategy dramatically simplifies the synthetic workflow by enabling the formation of the axially chiral center in a single step from racemic starting materials. The reaction proceeds under remarkably mild conditions, typically at room temperature (25°C) in dichloromethane, eliminating the need for energy-intensive heating or cryogenic cooling systems. By employing 4A molecular sieves as an additive to manage water content, the process ensures consistent high yields and superior stereocontrol. This shift from complex coupling to direct addition not only enhances the atom economy but also significantly broadens the range of accessible derivatives, allowing for the rapid generation of diverse libraries for biological screening.
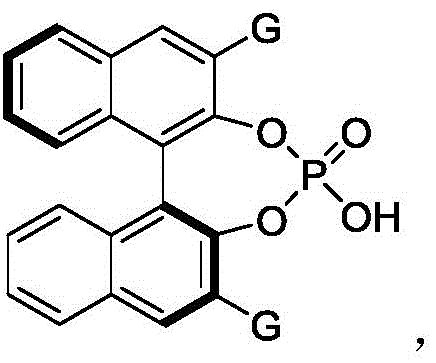
The mechanistic elegance of this transformation lies in the precise activation mode facilitated by the chiral phosphoric acid catalyst, specifically the BINOL-derived derivative known as Formula 61. This catalyst functions through a dual hydrogen-bonding activation mechanism, where the acidic proton of the phosphoric acid moiety activates the electrophilic azo compound while the basic phosphoryl oxygen interacts with the nucleophilic indole species. This bifunctional activation creates a highly organized chiral environment within the transition state, effectively discriminating between the enantiomers of the racemic substrate during the dynamic kinetic resolution process. The bulky 2,4,6-triisopropylphenyl groups at the 3,3'-positions of the BINOL backbone play a crucial steric role, shielding one face of the reacting species and forcing the approach of the nucleophile from the opposite direction, thus dictating the absolute configuration of the newly formed axial chirality.
Understanding the impurity profile is essential for regulatory compliance, and this catalytic system offers inherent advantages in杂质 control. The high enantioselectivity, with enantiomeric ratios (er) reaching up to 98:2 as demonstrated in the experimental data, means that the formation of the undesired enantiomer is minimized at the source rather than requiring costly downstream separation. The use of mild conditions also suppresses side reactions such as polymerization or decomposition of the sensitive azo intermediates, which are common pitfalls in more aggressive synthetic routes. Consequently, the crude reaction mixtures are cleaner, reducing the burden on purification steps and ensuring that the final active pharmaceutical ingredient (API) intermediates meet stringent purity specifications required by global health authorities.
How to Synthesize Axially Chiral Aryl Indole Efficiently
The synthesis protocol outlined in the patent provides a standardized procedure that balances efficiency with ease of execution, making it ideal for both laboratory optimization and pilot plant operations. The process begins with the preparation of the reaction vessel containing the indole substrate and the azo coupling partner in anhydrous dichloromethane, followed by the addition of activated 4A molecular sieves to maintain anhydrous conditions critical for catalyst performance.
- Prepare the reaction mixture by combining the indole substrate (Formula 1 or 4) and the azo compound (Formula 2) in dichloromethane solvent with 4A molecular sieves.
- Add the chiral phosphoric acid catalyst (specifically Formula 61) to the mixture at a loading of approximately 10 mol%.
- Stir the reaction at room temperature (25°C) until completion as monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The reliance on commercially available starting materials, such as substituted indoles and azo dicarboxylates, mitigates the risk of raw material shortages and price volatility often associated with exotic custom synthons. Since the reaction operates efficiently at ambient temperature, the energy consumption profile is drastically reduced compared to processes requiring reflux or sub-zero cooling, translating directly into lower utility costs per kilogram of product. Furthermore, the simplified work-up procedure involving filtration and standard silica gel chromatography eliminates the need for specialized equipment or hazardous quenching steps, streamlining the manufacturing timeline and enhancing overall throughput.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of organocatalysis significantly lowers the raw material cost base, while the high yields reduce the amount of starting material required per unit of output. The avoidance of heavy metals also removes the necessity for costly metal scavenging and residual metal testing, which are mandatory and expensive steps in API production. Additionally, the high selectivity minimizes waste generation, aligning with green chemistry principles and reducing waste disposal fees.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining steady supply lines to downstream drug manufacturers. The use of common solvents like dichloromethane and standard reagents means that sourcing is not dependent on single-source suppliers, thereby diversifying the supply base and reducing vulnerability to geopolitical or logistical disruptions. This reliability is paramount for long-term contracts where delivery continuity is a key performance indicator.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential due to its exothermic neutrality and lack of hazardous gas evolution, allowing for safe scale-up from gram to multi-ton quantities without significant engineering redesigns. The environmental footprint is minimized through the use of recyclable molecular sieves and the absence of toxic metal waste, facilitating easier compliance with increasingly stringent environmental regulations in major chemical manufacturing hubs. This sustainability profile enhances the marketability of the final product to eco-conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this axially chiral indole synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this technology for integration into their existing pipelines.
Q: What is the key advantage of the chiral phosphoric acid catalyst in this synthesis?
A: The chiral phosphoric acid catalyst (Formula 61) enables high enantioselectivity (er up to 98:2) under mild conditions (25°C), avoiding the need for harsh reagents or extreme temperatures often required in traditional coupling methods.
Q: What are the biological applications of these axially chiral indole compounds?
A: These compounds exhibit significant cytotoxic activity against MCF-7 breast cancer cells, with lead compound 3aa showing an IC50 value of 4.44 μg/mL, indicating strong potential as anticancer drug leads.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes readily available raw materials, common solvents like dichloromethane, and operates at room temperature, making it highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Aryl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of axially chiral aryl indoles as next-generation pharmaceutical intermediates, particularly for oncology and antiviral applications. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including advanced chiral HPLC analysis to guarantee the high enantiomeric excess demanded by modern drug development programs.
We invite you to collaborate with our technical team to explore how this innovative synthetic route can be tailored to your specific project needs. By engaging with us, you can request a Customized Cost-Saving Analysis that evaluates the economic impact of switching to this organocatalytic method for your specific target molecules. Please contact our technical procurement team today to obtain specific COA data for our reference standards and comprehensive route feasibility assessments that will accelerate your path to clinical trials.
