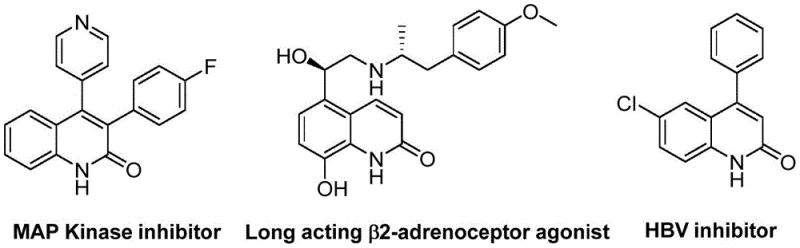
Advanced Palladium-Catalyzed Synthesis of 3-Arylquinoline-2(1H)-one Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic methodologies for constructing privileged heterocyclic scaffolds, particularly quinoline derivatives which serve as critical cores in numerous bioactive molecules. Patent CN113045489A introduces a groundbreaking preparation method for 3-arylquinoline-2(1H)-one derivatives, leveraging a sophisticated palladium-catalyzed aminocarbonylation strategy. This technology addresses long-standing challenges in heterocycle synthesis by utilizing benzisoxazole as a unique dual-function reagent, acting simultaneously as the nitrogen source and the formyl source. The significance of this chemical class cannot be overstated, as quinoline-2(1H)-one motifs are pervasive in drugs exhibiting antibiotic, antiplatelet, antitumor, and receptor antagonist activities. As illustrated in the structural diversity of bioactive compounds, these scaffolds are essential for modern drug discovery pipelines targeting complex diseases.
For procurement managers and R&D directors, the implications of this patent extend beyond mere academic interest; it represents a tangible opportunity for cost reduction in pharmaceutical intermediate manufacturing. By replacing hazardous carbon monoxide gas sources with stable solid reagents and employing a highly efficient catalytic system, the process offers a safer, more scalable alternative to traditional methods. The ability to tolerate a wide range of functional groups on both the benzisoxazole and benzyl chloride components ensures that this methodology can be adapted for the synthesis of diverse libraries of analogs, accelerating the lead optimization phase in drug development while maintaining stringent purity specifications required for clinical candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoline-2(1H)-one derivatives has relied heavily on classical named reactions such as the Vilsmeier-Haack, Knorr, and Friedlander condensations. While these methods are well-established, they suffer from significant drawbacks that hinder their application in modern, green chemistry-focused manufacturing environments. The Vilsmeier-Haack reaction, for instance, typically requires the use of phosphorus oxychloride and dimethylformamide, generating substantial amounts of corrosive phosphorus-containing waste that necessitates complex and costly disposal procedures. Furthermore, these traditional routes often demand harsh reaction conditions, including high temperatures and strongly acidic or basic media, which can lead to the degradation of sensitive functional groups and limit the substrate scope. Transition metal-catalyzed modifications have been attempted, but many still rely on external carbon monoxide sources, posing severe safety risks and requiring specialized high-pressure equipment that increases capital expenditure for production facilities.
The Novel Approach
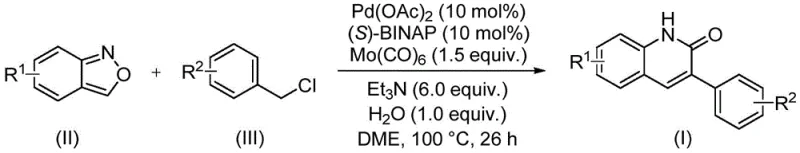
In stark contrast, the novel approach detailed in the patent utilizes a palladium-catalyzed aminocarbonylation reaction that fundamentally reimagines the construction of the quinolinone core. By employing benzisoxazole derivatives as the starting material, the process ingeniously bypasses the need for external CO gas, as the benzisoxazole ring itself provides the necessary carbonyl carbon upon ring opening. This innovation drastically simplifies the operational setup, allowing the reaction to proceed in standard sealed tubes or reactors at moderate temperatures around 100 °C. The use of a chiral ligand system, specifically (S)-BINAP, alongside palladium acetate and molybdenum carbonyl, ensures high catalytic turnover and excellent regioselectivity. This method not only enhances the safety profile by eliminating toxic gases but also improves the overall atom economy, making it an ideal candidate for sustainable chemical production.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic chemistry, involving a intricate catalytic cycle driven by the palladium center. The reaction initiates with the oxidative addition of the benzyl chloride derivative to the active palladium(0) species generated in situ from palladium acetate and the phosphine ligand. Subsequently, the benzisoxazole coordinates to the metal center, facilitating the insertion of the carbonyl moiety derived from the molybdenum carbonyl co-catalyst and the benzisoxazole ring opening. This step is critical as it forms the new carbon-carbon and carbon-nitrogen bonds that define the quinoline skeleton. The presence of water and triethylamine plays a pivotal role in proton transfer steps and neutralizing the hydrochloric acid byproduct, driving the equilibrium towards the formation of the final lactam structure. Understanding this mechanism allows chemists to fine-tune reaction parameters, such as ligand sterics and electronic properties, to maximize yields for specific substrates.
From an impurity control perspective, this catalytic system offers distinct advantages over non-catalytic thermal cyclizations. The high specificity of the palladium catalyst minimizes the formation of regioisomers and polymeric byproducts that often plague traditional condensation reactions. The mild reaction conditions prevent the decomposition of thermally labile substituents, ensuring that the final crude product contains a cleaner profile of impurities. This inherent selectivity reduces the burden on downstream purification processes, such as column chromatography or recrystallization, which are often the most resource-intensive steps in API intermediate production. Consequently, the overall process mass intensity (PMI) is improved, aligning with the industry's push towards greener and more efficient manufacturing protocols.
How to Synthesize 3-Arylquinoline-2(1H)-one Efficiently
The practical execution of this synthesis is designed for reproducibility and scalability, making it accessible for both laboratory research and pilot plant operations. The protocol involves mixing the key reagents—benzisoxazole, benzyl chloride, palladium catalyst, ligand, and additives—in a polar aprotic solvent like DME. The reaction is then heated to 100 °C for a defined period, typically around 26 hours, to ensure complete conversion. Detailed standardized synthetic steps for specific derivatives are outlined below, providing a clear roadmap for technical teams to replicate the high yields reported in the patent data.
- Combine palladium acetate, (S)-BINAP ligand, molybdenum carbonyl, triethylamine, water, benzisoxazole, and the specific benzyl chloride derivative in a sealed reaction vessel.
- Add ethylene glycol dimethyl ether (DME) as the solvent and heat the mixture to 100 °C for approximately 26 hours under stirring conditions.
- Upon completion, filter the reaction mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 3-arylquinoline-2(1H)-one.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this synthetic route presents compelling economic and logistical benefits that directly impact the bottom line. The primary advantage lies in the accessibility and cost-effectiveness of the raw materials. Benzisoxazoles and substituted benzyl chlorides are commodity chemicals available from multiple global suppliers, reducing the risk of supply chain bottlenecks associated with proprietary or exotic reagents. Furthermore, the elimination of high-pressure carbon monoxide cylinders removes a significant safety hazard and regulatory burden, lowering insurance and compliance costs for manufacturing sites. The robustness of the reaction conditions means that the process can be transferred to standard glass-lined or stainless steel reactors without requiring specialized high-pressure autoclaves, facilitating rapid technology transfer and scale-up.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom efficiency and the use of inexpensive, commercially available starting materials. By utilizing benzisoxazole as a combined nitrogen and carbon source, the need for additional formylating agents is eliminated, reducing the total number of raw material inputs. Additionally, the high catalytic efficiency allows for lower catalyst loading relative to the substrate, minimizing the cost associated with precious metal recovery and recycling. The simplified workup procedure, which involves filtration and standard chromatography, further reduces labor and solvent consumption compared to multi-step traditional syntheses, leading to substantial overall cost savings in the production of these valuable intermediates.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for uninterrupted API production. This method relies on a modular approach where various substituted benzyl chlorides can be easily swapped to generate different analogs without changing the core reaction conditions. This flexibility allows manufacturers to respond quickly to changes in demand for specific drug candidates. Moreover, the stability of the reagents ensures long shelf-life and ease of storage, mitigating the risks of raw material degradation during transit or warehousing. The use of common solvents like DME and triethylamine further ensures that the supply chain remains resilient against fluctuations in the availability of specialized chemicals.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often reveals hidden inefficiencies, but this palladium-catalyzed route is inherently scalable due to its homogeneous nature and mild thermal requirements. The reaction generates minimal hazardous waste, primarily consisting of benign salts and spent catalyst residues that can be managed through standard waste treatment protocols. This aligns with increasingly stringent environmental regulations regarding volatile organic compounds (VOCs) and heavy metal discharge. The ability to run the reaction at atmospheric pressure (in sealed vessels) or low pressure simplifies reactor design and maintenance, ensuring that the process remains economically viable even at very large production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this synthesis method for industrial applications.
Q: What are the primary advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual function as both the nitrogen source and the formyl (carbonyl) source, eliminating the need for separate carbon monoxide gas or toxic formylating agents, thereby simplifying the operational safety profile and reducing raw material costs.
Q: How does this method compare to traditional Vilsmeier-Haack reactions?
A: Unlike the Vilsmeier-Haack reaction which often requires harsh conditions and generates significant phosphorus waste, this palladium-catalyzed approach operates under milder temperatures with high functional group tolerance, resulting in cleaner reaction profiles and easier downstream purification.
Q: Is this process suitable for large-scale manufacturing of API intermediates?
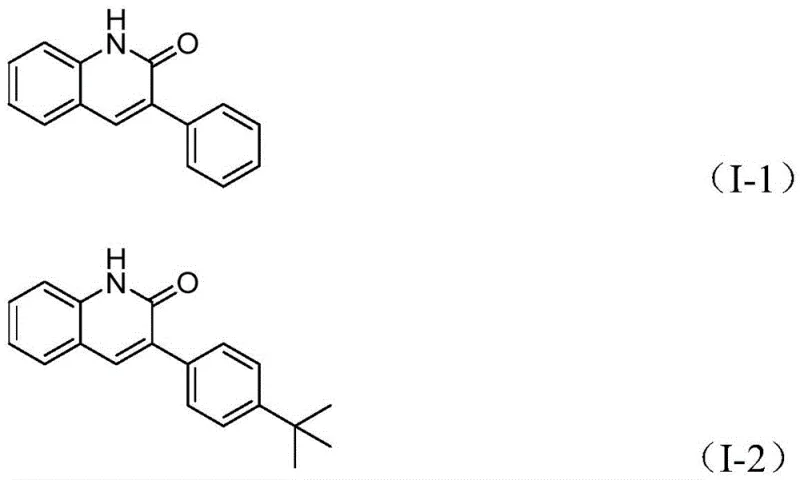
A: Yes, the process utilizes commercially available and inexpensive starting materials like benzyl chlorides and benzisoxazoles, and the catalytic system demonstrates high efficiency with yields up to 97%, making it highly viable for commercial scale-up in pharmaceutical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinoline-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN113045489A for accelerating drug development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. We are committed to delivering high-purity 3-arylquinoline-2(1H)-one derivatives that meet rigorous QC labs standards, supporting your efforts to bring life-saving medications to market faster and more efficiently.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce overall production costs.
