Revolutionizing Quinolin-2-one Synthesis: A Scalable Pd-Catalyzed Aminocarbonylation Strategy for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to access privileged heterocyclic scaffolds, particularly quinoline-2(1H)-one derivatives, which serve as critical cores in numerous bioactive molecules. As illustrated in the structural diversity of known bioactive compounds, these scaffolds are integral to MAP Kinase inhibitors, long-acting β2-adrenoceptor agonists, and HBV inhibitors, highlighting their immense therapeutic value. 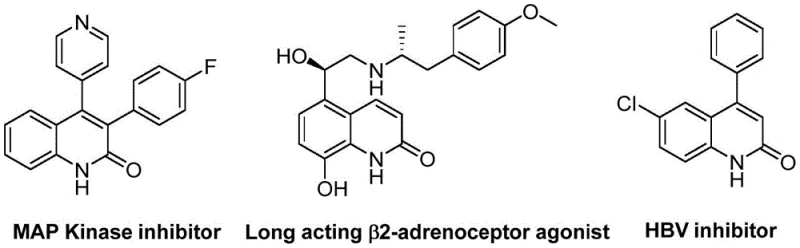 Patent CN113045489A introduces a groundbreaking preparation method that addresses long-standing synthetic challenges by utilizing a palladium-catalyzed aminocarbonylation strategy. This innovation leverages benzisoxazole not merely as a reactant but as a sophisticated dual-source reagent, providing both the nitrogen atom and the formyl group necessary for ring construction. By shifting away from traditional, often hazardous carbonylation protocols, this technology offers a streamlined pathway that enhances safety profiles while maintaining high reaction efficiency, making it an attractive candidate for industrial scale-up and reliable pharmaceutical intermediate supplier partnerships.
Patent CN113045489A introduces a groundbreaking preparation method that addresses long-standing synthetic challenges by utilizing a palladium-catalyzed aminocarbonylation strategy. This innovation leverages benzisoxazole not merely as a reactant but as a sophisticated dual-source reagent, providing both the nitrogen atom and the formyl group necessary for ring construction. By shifting away from traditional, often hazardous carbonylation protocols, this technology offers a streamlined pathway that enhances safety profiles while maintaining high reaction efficiency, making it an attractive candidate for industrial scale-up and reliable pharmaceutical intermediate supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)-one derivatives has relied heavily on classical methodologies such as the Vilsmeier-Haack, Knorr, and Friedlander reactions, which, while effective, often suffer from significant operational drawbacks in a modern manufacturing context. These traditional routes frequently necessitate harsh reaction conditions, including the use of strong acids or bases, elevated temperatures that can degrade sensitive functional groups, and multi-step sequences that erode overall yield and increase waste generation. Furthermore, transition metal-catalyzed modifications to these processes have sometimes required expensive ligands or toxic carbon monoxide gas handled under high pressure, posing substantial safety risks and regulatory hurdles for large-scale production facilities. The complexity of post-treatment in these conventional methods often involves tedious purification steps to remove heavy metal residues or byproducts, leading to increased production costs and extended lead times that strain supply chain reliability for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a mild and highly efficient palladium-catalyzed system that fundamentally reimagines the construction of the quinoline core. By employing benzisoxazole and benzyl chloride compounds as starting materials, the method bypasses the need for external carbon monoxide gas, instead generating the carbonyl moiety in situ from molybdenum hexacarbonyl and the benzisoxazole scaffold itself. This strategic design allows the reaction to proceed under relatively mild thermal conditions, typically around 100°C, which preserves the integrity of sensitive substituents and minimizes energy consumption. The use of readily available and inexpensive reagents, combined with a robust catalytic system involving Pd(OAc)2 and (S)-BINAP, ensures that the process is not only chemically elegant but also economically viable for cost reduction in API intermediate manufacturing. This shift represents a paradigm change towards greener, safer, and more atom-economical synthesis strategies that align with modern sustainability goals.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The core of this technological breakthrough lies in the intricate palladium-catalyzed aminocarbonylation mechanism, which orchestrates the coupling of the benzyl chloride and benzisoxazole fragments with remarkable precision. The catalytic cycle likely initiates with the oxidative addition of the benzyl chloride to the active Pd(0) species, forming a benzyl-palladium(II) intermediate that is primed for subsequent transformations. Following this, the coordination and insertion of carbon monoxide, derived from the decomposition of Mo(CO)6 under the reaction conditions, generates an acyl-palladium complex. This electrophilic species then undergoes nucleophilic attack by the nitrogen atom of the benzisoxazole, facilitating the critical C-N bond formation that closes the heterocyclic ring. 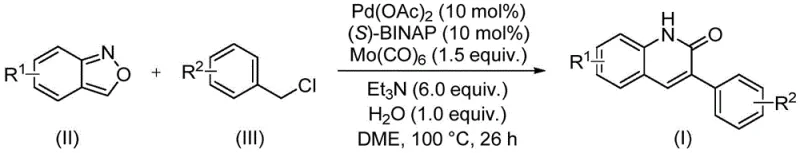 The final step involves reductive elimination or protonolysis to release the 3-arylquinoline-2(1H)-one product and regenerate the active catalyst, completing the cycle with high turnover numbers.
The final step involves reductive elimination or protonolysis to release the 3-arylquinoline-2(1H)-one product and regenerate the active catalyst, completing the cycle with high turnover numbers.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions that typically plague carbonylation processes. The specific choice of the (S)-BINAP ligand creates a steric environment that favors the desired cross-coupling over homocoupling of the benzyl chloride or decomposition of the benzisoxazole. Additionally, the presence of water and triethylamine in the reaction mixture plays a crucial role in buffering the system and facilitating the hydrolysis or rearrangement steps necessary to finalize the quinoline structure without generating excessive acidic byproducts. This controlled environment ensures that the resulting crude mixture is cleaner, reducing the burden on downstream purification units and allowing for the consistent production of high-purity OLED material or pharmaceutical precursors with minimal batch-to-batch variability.
How to Synthesize 3-Arylquinoline-2(1H)-one Efficiently
The practical execution of this synthesis is designed for operational simplicity, requiring standard laboratory or pilot plant equipment without the need for specialized high-pressure reactors. The protocol involves charging a sealed vessel with the requisite amounts of palladium acetate, (S)-BINAP, molybdenum carbonyl, triethylamine, water, benzisoxazole, and the specific benzyl chloride derivative in a solvent such as ethylene glycol dimethyl ether (DME). The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Combine palladium acetate, (S)-BINAP, molybdenum carbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in DME solvent within a sealed vessel.
- Heat the reaction mixture to 100°C and maintain stirring for 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify using column chromatography to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere chemical yield. The reliance on benzisoxazole and benzyl chloride compounds, which are commodity chemicals available from multiple global vendors, drastically reduces the risk of raw material shortages and price volatility associated with exotic or proprietary reagents. This accessibility ensures a stable supply base, allowing manufacturers to negotiate better pricing and secure long-term contracts that protect against market fluctuations. Furthermore, the elimination of high-pressure carbon monoxide cylinders removes a significant safety liability and regulatory compliance burden, simplifying facility permitting and insurance costs while enhancing overall operational continuity.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by its exceptional atom economy and the dual functionality of the benzisoxazole reactant, which eliminates the need for separate formylating agents and reduces the total number of synthetic steps. By consolidating the nitrogen and carbon sources into a single molecule, the method significantly lowers the mass intensity of the process, resulting in reduced solvent usage and waste disposal costs. The high reaction efficiency observed across a broad range of substrates means that less starting material is wasted on side products, directly improving the cost-per-kilogram metric for the final active pharmaceutical ingredient. Additionally, the simplified post-treatment procedure, which often requires only filtration and standard chromatography, minimizes labor hours and equipment occupancy time, further driving down the overall manufacturing overhead.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system against varying substrate electronic properties ensures that supply chains remain resilient even when switching between different analogues or derivatives. Because the reaction tolerates a wide array of functional groups—including halogens, alkoxy, and cyano groups—manufacturers can produce a diverse library of intermediates using a single, standardized platform technology. This flexibility allows for rapid response to changing R&D demands without the need for extensive process re-validation or retooling. The use of common solvents like DME and standard catalysts like Pd(OAc)2 ensures that logistics are straightforward, with no requirement for cryogenic shipping or specialized hazmat handling that could delay deliveries.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram quantities is facilitated by the mild thermal profile and the absence of gaseous reagents that are difficult to manage in large batches. The process operates effectively at 100°C, a temperature easily achievable with standard steam or oil heating systems found in most multipurpose chemical plants. From an environmental standpoint, the reduction in hazardous reagents and the potential for catalyst recycling contribute to a lower E-factor, aligning with increasingly stringent global environmental regulations. This green chemistry profile not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a sustainable partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route, derived directly from the experimental data and beneficial effects reported in the patent documentation. Understanding these nuances is critical for R&D teams evaluating the feasibility of integrating this technology into their existing pipelines.
Q: What is the unique role of benzisoxazole in this synthetic route?
A: In this patented methodology, benzisoxazole serves a dual function as both the nitrogen source and the formyl (carbonyl) source, which significantly simplifies the atom economy and reduces the need for separate carbon monoxide sources or complex formylating agents.
Q: What are the specific reaction conditions required for high yields?
A: The optimal conditions involve reacting the substrates at 100°C for 26 hours in ethylene glycol dimethyl ether (DME) using a catalytic system of Pd(OAc)2 and (S)-BINAP with Mo(CO)6 as the carbonyl source.
Q: Does this method support diverse functional group tolerance?
A: Yes, the process demonstrates excellent substrate compatibility, successfully tolerating various substituents such as halogens, alkoxy groups, cyano groups, and trifluoromethyl groups on both the benzisoxazole and benzyl chloride rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinoline-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed aminocarbonylation technology for the next generation of therapeutic agents. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless and efficient. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-arylquinoline-2(1H)-one derivative meets the highest international standards for quality and consistency.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this methodology can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, and let us help you accelerate your development timeline with confidence.
