Advanced Electrochemical Synthesis of 9-Azacyclo-Substituted Xanthene Compounds for Commercial Scale
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards sustainable and atom-economical processes, driven by the urgent need for greener supply chains in the pharmaceutical and agrochemical sectors. A pivotal advancement in this domain is documented in patent CN109761963B, which discloses a novel organic electrosynthesis method for preparing 9-azacyclo-substituted xanthene compounds. This technology leverages selective electrochemical oxidation of Csp3-H bonds to construct C-N bonds directly, bypassing the need for pre-functionalized substrates or harsh chemical oxidants. For R&D Directors and Procurement Managers seeking reliable fine chemical intermediate supplier partnerships, this patent represents a significant opportunity to optimize synthetic routes. By utilizing electrons as traceless reagents, the process not only enhances environmental compliance but also streamlines the purification workflow, thereby addressing critical pain points related to cost reduction in pharmaceutical intermediates manufacturing. The following analysis details the technical merits and commercial viability of this electrochemical strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing 9-azacyclo-substituted xanthene scaffolds often rely on classical nucleophilic substitution reactions or transition-metal catalyzed cross-couplings. These conventional methodologies frequently necessitate the use of stoichiometric amounts of strong chemical oxidants or expensive noble metal catalysts, which introduce significant impurities into the final product matrix. The presence of residual heavy metals poses a severe challenge for downstream applications, particularly in the synthesis of high-purity API intermediates, where regulatory limits are stringent. Furthermore, traditional methods often require elevated temperatures and prolonged reaction times, leading to higher energy consumption and potential thermal degradation of sensitive functional groups. The generation of substantial chemical waste from oxidants and catalyst ligands also complicates waste treatment protocols, increasing the overall environmental footprint and operational costs associated with commercial scale-up of complex polymer additives or pharmaceutical building blocks.
The Novel Approach
In stark contrast, the electrochemical method described in the patent utilizes electric current to drive the oxidative coupling, effectively replacing chemical oxidants with electrons. This approach operates under remarkably mild conditions, typically at room temperature, which preserves the integrity of sensitive substituents on the xanthene and heterocyclic rings. The use of inexpensive and readily available electrode materials, such as graphite rods and platinum plates, eliminates the dependency on scarce transition metals, thereby enhancing supply chain security. This methodology offers a robust pathway for reducing lead time for high-purity pharmaceutical intermediates by simplifying the work-up procedure; without metal catalysts to remove, the purification process becomes more efficient. The broad substrate tolerance demonstrated in the patent suggests that this platform technology can be adapted for the synthesis of diverse libraries of bioactive molecules, providing a versatile tool for modern organic synthesis.
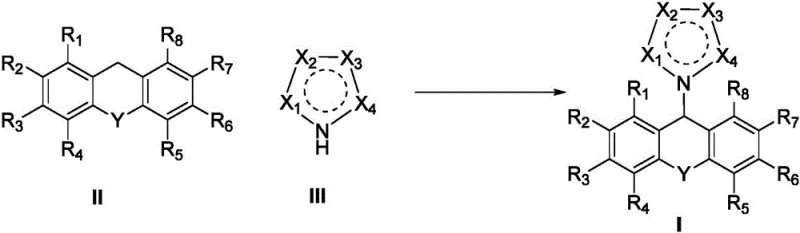
Mechanistic Insights into Electrochemical Csp3-H Amination
The core innovation of this technology lies in the selective activation of the inert Csp3-H bond at the 9-position of the xanthene ring system. Under anodic oxidation conditions, the xanthene substrate undergoes a single electron transfer (SET) to generate a radical cation intermediate. This highly reactive species is then deprotonated to form a carbon-centered radical, which subsequently couples with the nitrogen nucleophile from the heterocyclic partner. The precise control of potential and current density allows for the modulation of reaction selectivity, minimizing over-oxidation or side reactions that typically plague radical chemistry. For technical teams evaluating cost reduction in electronic chemical manufacturing or pharma synthesis, understanding this mechanism is crucial as it highlights the tunability of the process. The use of methanesulfonic acid as an additive further facilitates the proton transfer steps, ensuring smooth turnover of the catalytic cycle without the accumulation of acidic byproducts that could corrode equipment or degrade the product.
Impurity control is inherently superior in this electrochemical system due to the absence of exogenous oxidizing agents that often generate unpredictable side products. The reaction pathway is dictated by the redox potentials of the substrates and the electrode material, offering a level of chemoselectivity that is difficult to achieve with traditional reagents. This results in a cleaner crude reaction mixture, which translates to higher isolated yields and reduced solvent usage during chromatography. For supply chain stakeholders, this efficiency means less raw material waste and lower disposal costs. The ability to synthesize high-purity OLED material precursors or agrochemical intermediates with minimal impurity profiles is a distinct competitive advantage. The mechanistic elegance of using electricity to forge C-N bonds underscores the potential for this technology to become a standard operation in green chemistry manufacturing facilities.
How to Synthesize 9-Azacyclo-Substituted Xanthene Efficiently
Implementing this electrochemical protocol requires careful attention to cell configuration and reaction parameters to ensure reproducibility and safety. The standard procedure involves dissolving the xanthene derivative and the nitrogen heterocycle in anhydrous acetonitrile, followed by the addition of a supporting electrolyte such as tetrabutylammonium tetrafluoroborate. The reaction is conducted in an undivided cell equipped with a graphite anode and a platinum cathode, applying a constant current of approximately 10 mA. Detailed standardized synthesis steps see the guide below.
- Prepare the electrolytic cell by adding xanthene derivative, nitrogen heterocycle, electrolyte (nBu4NBF4), and methanesulfonic acid in acetonitrile solvent.
- Insert a graphite rod anode and a platinum plate cathode into the dry three-necked flask and ensure secure connections.
- Apply a constant current of 10 mA at room temperature for 2 hours, monitoring progress via TLC until the reaction is complete.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this electrochemical synthesis route offers transformative benefits for procurement strategies and supply chain resilience. The elimination of expensive transition metal catalysts and stoichiometric oxidants directly correlates to a significant reduction in raw material costs. Moreover, the simplified purification process reduces the consumption of silica gel and organic solvents, further driving down the cost of goods sold (COGS). For procurement managers, this translates to a more stable pricing model for reliable agrochemical intermediate supplier contracts, as the process is less susceptible to fluctuations in the market prices of precious metals. The mild reaction conditions also enhance operational safety, reducing the risk of thermal runaways and allowing for more flexible manufacturing scheduling.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the replacement of costly chemical reagents with electricity, which is generally cheaper and more consistent in price. By removing the need for heavy metal catalysts, manufacturers avoid the expensive and time-consuming steps required for metal scavenging and residue testing, which are mandatory in pharmaceutical production. This streamlined workflow allows for faster batch turnover and reduced labor costs associated with complex work-ups. Additionally, the high atom economy of the reaction ensures that a greater proportion of raw materials are converted into the desired product, minimizing waste disposal fees and maximizing resource efficiency.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like acetonitrile and graphite electrodes, rather than specialized catalysts with long lead times, significantly de-risks the supply chain. This accessibility ensures that production can be maintained even during periods of global supply disruption for rare earth metals or complex ligands. The robustness of the electrochemical setup allows for continuous processing possibilities, which can drastically improve throughput and inventory management. For supply chain heads, this means a more predictable delivery schedule for commercial scale-up of complex polymer additives or intermediates, ensuring continuity for downstream clients.
- Scalability and Environmental Compliance: Electrochemical reactors are inherently scalable through the numbering-up of cells or the use of flow chemistry setups, facilitating a smooth transition from laboratory grams to multi-ton production. The green nature of the process aligns perfectly with increasingly strict environmental regulations regarding volatile organic compounds (VOCs) and heavy metal discharge. By adopting this technology, companies can future-proof their manufacturing sites against tightening environmental laws, avoiding potential fines and shutdowns. This sustainability profile also enhances the brand value of the final product, appealing to end-users who prioritize green sourcing in their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing production lines.
Q: What are the primary advantages of using electrochemical synthesis for xanthene derivatives?
A: Electrochemical synthesis eliminates the need for stoichiometric chemical oxidants and transition metal catalysts, significantly reducing heavy metal contamination risks and waste generation while offering milder reaction conditions.
Q: Is this method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes simple electrode materials like graphite and platinum, operates at room temperature, and uses common solvents like acetonitrile, making it highly adaptable for flow chemistry and large-scale manufacturing.
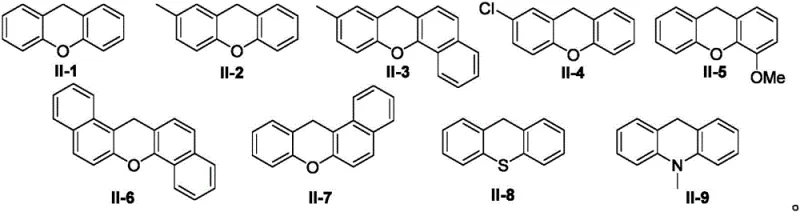
Q: What is the substrate scope for the nitrogen heterocycle component?
A: The method demonstrates broad compatibility with various nitrogen heterocycles including indazoles, benzotriazoles, and pyrazoles, allowing for the synthesis of diverse 9-azacyclo-substituted xanthene libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Azacyclo-Substituted Xanthene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of high-value intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this are successfully translated into robust industrial processes. Our facility is equipped with state-of-the-art electrochemical reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required by global pharmaceutical and agrochemical companies.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this electrochemical route can optimize your budget. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of 9-azacyclo-substituted xanthene derivatives.
