Advanced Electrochemical C-H Activation for Scalable 9-Azacyclo Xanthene Production
Advanced Electrochemical C-H Activation for Scalable 9-Azacyclo Xanthene Production
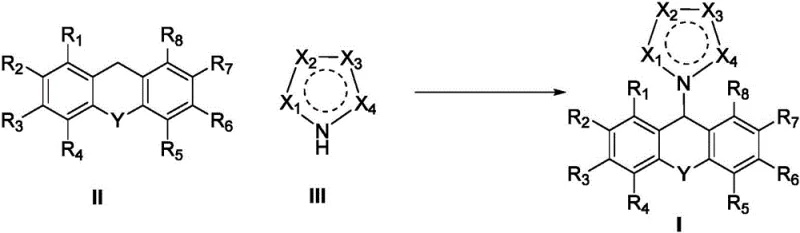
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards sustainable and efficient synthetic methodologies, driven by the urgent need for greener processes in the pharmaceutical and agrochemical sectors. A groundbreaking development in this arena is detailed in patent CN109761963B, which introduces a novel organic electrosynthesis method for preparing 9-azacyclo-substituted xanthene compounds. This technology leverages selective electrochemical oxidation of Csp3-H bonds to construct critical C-N bonds, bypassing the limitations of traditional thermal catalysis. By utilizing electricity as a traceless reagent, this approach offers a compelling alternative for producing high-value heterocyclic intermediates, addressing key pain points regarding environmental compliance and process safety that plague conventional oxidation strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 9-position functionalized xanthenes has relied heavily on harsh chemical oxidants or transition metal catalysts, which introduce significant logistical and environmental burdens. Traditional routes often require stoichiometric amounts of toxic heavy metals or aggressive oxidizing agents, leading to complex downstream purification processes to remove metal residues that are strictly regulated in pharmaceutical applications. Furthermore, these conventional methods frequently suffer from limited substrate scope, failing to tolerate sensitive functional groups such as halogens or nitro groups without side reactions. The reliance on high temperatures and pressure in some prior art methods also escalates energy consumption and safety risks, making the commercial scale-up of complex polymer additives or drug intermediates economically challenging and operationally hazardous for supply chain managers.
The Novel Approach
In stark contrast, the electrochemical strategy disclosed in the patent utilizes a constant current electrolysis system to drive the C-H functionalization under remarkably mild conditions. The core innovation lies in the anodic oxidation of the xanthene substrate at the C9 position, generating a reactive intermediate that couples efficiently with nitrogen-containing heterocycles. This method operates at room temperature in common solvents like acetonitrile, eliminating the need for external heating or cooling infrastructure. The use of inexpensive graphite rods as anodes and platinum plates as cathodes ensures that the process remains cost-effective while maintaining high selectivity. As illustrated in the general reaction scheme below, this direct coupling avoids the multi-step sequences typical of older methodologies, thereby streamlining the production workflow.
Mechanistic Insights into Electrochemical C-H Activation
The mechanistic elegance of this process stems from the precise control of electron transfer at the electrode-solution interface. Upon application of a constant current, typically optimized at 10 mA for laboratory scales, the xanthene derivative undergoes a single-electron oxidation at the anode to form a radical cation. This species rapidly loses a proton to generate a stabilized benzylic radical or carbocation at the C9 position, which is highly susceptible to nucleophilic attack. The nitrogen heterocycle, acting as the nucleophile, attacks this electrophilic center to form the new C-N bond, completing the functionalization. The presence of methanesulfonic acid as an auxiliary agent plays a crucial role in facilitating proton transfer and stabilizing the reaction medium, ensuring that the electrochemical cycle proceeds with high efficiency and minimal side product formation.
From an impurity control perspective, this electrochemical pathway offers superior selectivity compared to free-radical initiators used in thermal processes. The potential applied across the electrodes can be tuned to selectively oxidize the specific Csp3-H bond of the xanthene without affecting other sensitive moieties on the aromatic rings. This chemoselectivity is vital for R&D directors aiming to minimize the formation of regio-isomers or over-oxidized byproducts, which are difficult to separate and can compromise the purity profile of the final API intermediate. The use of nBu4NBF4 as a supporting electrolyte further enhances conductivity and stability, allowing the reaction to proceed smoothly even with substrates bearing electron-withdrawing groups, thus broadening the utility of this method for synthesizing diverse chemical libraries.
How to Synthesize 9-Azacyclo Xanthene Efficiently
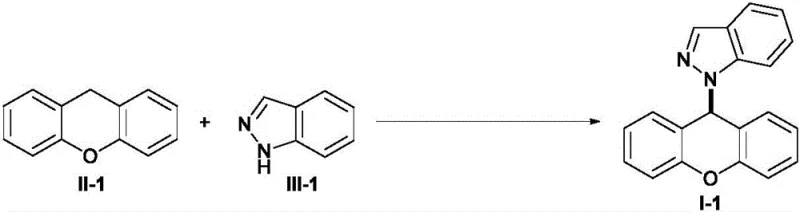
To implement this synthesis effectively, one must adhere to the optimized parameters established in the patent examples, which balance reaction rate with product quality. The process begins with the preparation of a dry reaction vessel equipped with the specified electrode configuration to prevent moisture interference, which can quench the electrochemical active species. A representative example involves the coupling of unsubstituted xanthene with indazole, yielding the target product in excellent isolated yields after simple workup procedures. The reaction progress is conveniently monitored via TLC, allowing operators to stop the electrolysis precisely upon completion to prevent over-reaction. The specific transformation is depicted below, highlighting the simplicity of the setup and the cleanliness of the conversion.
- Load a dry three-necked flask with xanthene derivative, azole compound, electrolyte (nBu4NBF4), auxiliary acid (MsOH), and acetonitrile solvent.
- Insert graphite rod anode and platinum plate cathode, then apply a constant current of 10 mA at room temperature for approximately 2 hours.
- Upon completion, wash the mixture with water, extract with diethyl ether, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology translates into tangible strategic benefits beyond mere technical novelty. The elimination of expensive stoichiometric oxidants and precious metal catalysts drastically simplifies the bill of materials, reducing exposure to volatile commodity prices associated with palladium or rhodium. Furthermore, the benign nature of the reagents—primarily electricity, solvent, and simple salts—mitigates the regulatory hurdles associated with the disposal of heavy metal waste, aligning perfectly with modern ESG (Environmental, Social, and Governance) mandates. This streamlined process not only lowers the direct cost of goods sold but also reduces the lead time for high-purity intermediates by shortening the purification timeline, as there are no metal scavenging steps required.
- Cost Reduction in Manufacturing: The primary economic driver here is the substitution of costly chemical reagents with electricity, which is significantly cheaper and more consistent in price. By removing the need for transition metal catalysts, the process avoids the expensive and time-consuming step of metal removal, which often requires specialized resins or additional chromatography. This reduction in unit operations directly lowers labor and material costs, resulting in substantial cost savings in fine chemical manufacturing without compromising on the quality or yield of the final product.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as xanthene derivatives and various azoles, are commercially available and structurally diverse, reducing the risk of single-source dependency. The robustness of the electrochemical cell setup means that production is less susceptible to the supply disruptions often caused by the scarcity of specialized catalysts. Additionally, the mild reaction conditions reduce equipment wear and tear, ensuring higher uptime and more reliable delivery schedules for critical pharmaceutical intermediates needed for just-in-time manufacturing pipelines.
- Scalability and Environmental Compliance: Electrochemical processes are inherently scalable; increasing production volume often involves adding more electrode pairs or increasing cell size rather than reinventing the chemistry. This modularity supports the commercial scale-up of complex heterocyclic intermediates from pilot plants to multi-ton facilities with minimal re-optimization. Moreover, the absence of toxic heavy metal waste simplifies wastewater treatment and lowers the environmental footprint, ensuring compliance with increasingly stringent global environmental regulations and facilitating smoother audits from international clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis route. These answers are derived directly from the experimental data and scope defined in the patent literature, providing a realistic overview of the technology's capabilities and limitations for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production lines or new product development pipelines.
Q: What are the primary advantages of this electrochemical method over traditional oxidation?
A: This method eliminates the need for stoichiometric chemical oxidants and heavy metal catalysts, utilizing electricity as a clean reagent. It operates under mild room temperature conditions with high atom economy and significantly reduced waste generation.
Q: Does this process support a wide range of substrate substituents?
A: Yes, the patent demonstrates excellent tolerance for various functional groups including halogens, alkyls, alkoxy, and nitro groups on both the xanthene and azole rings, ensuring broad applicability for diverse intermediate synthesis.
Q: Is this electrochemical protocol suitable for industrial scale-up?
A: Absolutely. The use of constant current electrolysis with robust electrode materials like graphite and platinum allows for straightforward scaling from laboratory benchtop to commercial production reactors without complex parameter adjustments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Azacyclo-substituted xanthene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of high-value intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by top-tier pharmaceutical companies. We are committed to delivering high-purity 9-azacyclo-substituted xanthene derivatives that meet the exacting standards of the global market.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in green chemistry can drive efficiency and value for your supply chain.
