Advanced Biphenyl Phosphine Ligands: Technical Upgrade and Commercial Scalability for Global Supply Chains
The chemical industry is constantly evolving, driven by the need for more efficient and selective catalytic processes that can meet the rigorous demands of modern pharmaceutical and fine chemical manufacturing. Patent CN102516300A introduces a groundbreaking class of phosphine ligands characterized by a novel biphenyl skeleton that fundamentally alters the landscape of asymmetric catalysis. These ligands are not merely incremental improvements but represent a strategic shift in how chiral information is transferred during synthesis, moving from traditional resolution methods to a more elegant desymmetry approach. By achieving complete transfer from face chirality to axial chirality, this technology offers a robust platform for generating high-value intermediates with exceptional stereochemical control. For global supply chain leaders and R&D directors, understanding the implications of this patent is crucial for maintaining competitiveness in the production of complex active pharmaceutical ingredients. The ability to access such high-performance ligands through a streamlined synthetic route positions this technology as a cornerstone for next-generation catalytic applications.
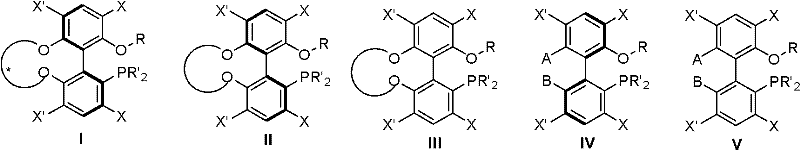
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral monodentate phosphine ligands has relied heavily on binaphthyl skeletons, such as the well-known MOP ligands, which often suffer from significant structural rigidity and limited electronic tunability. These conventional ligands typically possess a fixed dihedral angle that may not be optimal for a diverse range of substrates, leading to suboptimal enantioselectivity in complex transformation reactions. Furthermore, the traditional preparation of such chiral ligands frequently necessitates cumbersome chiral resolution processes, which involve separating enantiomers from racemic mixtures using expensive chiral resolving agents or chromatography. This reliance on resolution not only drastically increases the material cost due to the theoretical maximum yield of 50% for the desired enantiomer but also introduces substantial waste generation and processing time. In an industrial setting, the need for multiple recrystallization steps or preparative HPLC purification creates bottlenecks that hinder the scalability and economic viability of producing these critical catalytic components. Consequently, procurement managers often face inflated costs and extended lead times when sourcing high-purity chiral ligands produced via these legacy methodologies.
The Novel Approach
In stark contrast, the methodology disclosed in CN102516300A utilizes a sophisticated desymmetry reaction strategy that elegantly bypasses the need for traditional chiral resolution entirely. By starting with achiral or pro-chiral biphenyl precursors and introducing a chiral side chain through a ring-forming reaction, the synthesis effectively locks the axial chirality of the biphenyl backbone through the steric constraints imposed by the newly formed cyclic structure. This approach ensures that the chirality is induced directly during the bond-forming steps, theoretically allowing for 100% atom economy regarding the chiral information transfer without the loss of half the material. The synthetic route is designed to be simple and economical, utilizing readily available chiral diols or dicarboxylic acids as the source of chirality, which are often cheaper and more accessible than specialized resolving agents. This paradigm shift from separation to induction means that manufacturers can achieve high optical purity directly from the reaction vessel, significantly simplifying the downstream purification workflow. For supply chain heads, this translates to a more predictable and reliable production schedule, as the variability associated with resolution efficiency is completely eliminated from the process.
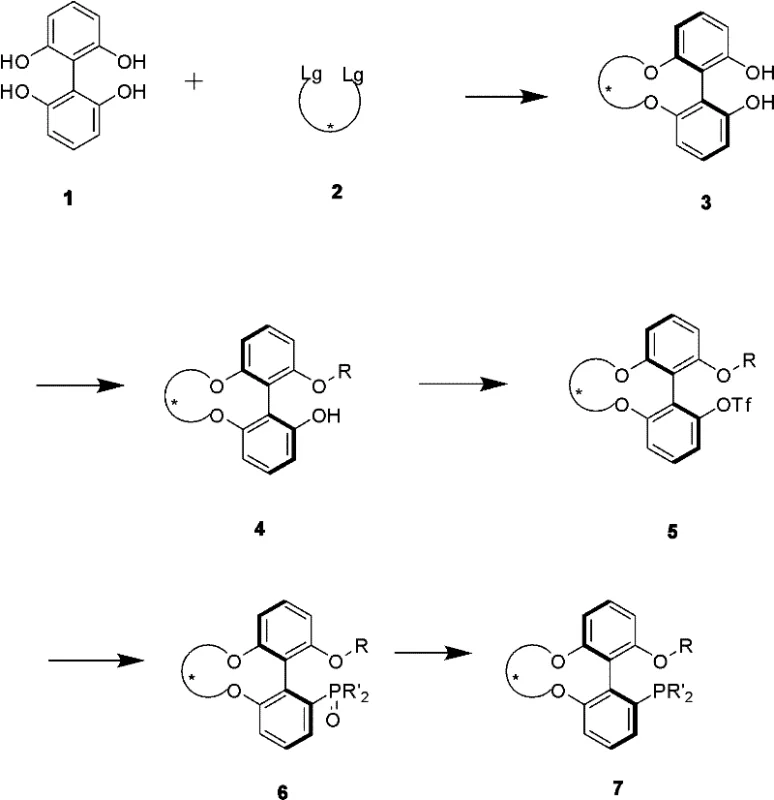
Mechanistic Insights into Desymmetry-Driven Axial Chirality Transfer
The core mechanistic innovation of this technology lies in the precise control of the dihedral angle of the biphenyl system through the introduction of a chiral bridging unit. In asymmetric catalysis, the spatial arrangement of the ligand around the metal center is paramount, as it dictates the trajectory of the substrate approach and the energy difference between competing transition states. The novel ligands described in the patent utilize a chiral diol-derived bridge that connects the two phenyl rings, effectively freezing the rotation around the biaryl axis and establishing a stable atropisomeric configuration. This structural rigidity ensures that the chiral environment presented to the catalytic metal center is consistent and well-defined, which is critical for achieving high enantiomeric excess in the final product. The transfer of chirality from the central chirality of the diol bridge to the axial chirality of the biphenyl backbone is driven by thermodynamic stability and steric hindrance during the cyclization step. This mechanism allows for fine-tuning of the ligand's steric bulk and electronic properties by simply varying the substituents on the chiral bridge or the phosphine moiety, offering a modular platform for catalyst optimization. R&D directors can leverage this modularity to tailor ligands for specific challenging transformations, such as asymmetric allylation or Suzuki couplings, where standard ligands often fail to provide adequate selectivity.
Furthermore, the impurity profile of the resulting ligands is inherently cleaner due to the stereospecific nature of the desymmetry reaction. In traditional resolution processes, trace amounts of the opposite enantiomer can persist even after multiple purification steps, potentially acting as a poison or a competing catalyst in sensitive asymmetric reactions. However, since the chirality in this new system is generated de novo from a single enantiomer of the starting diol, the risk of contamination by the wrong atropisomer is minimized provided the starting material is pure. The patent details specific reaction conditions, such as the use of inorganic bases like cesium carbonate in polar aprotic solvents, which facilitate the nucleophilic substitution required for ring closure without epimerizing the chiral centers. The subsequent steps, including selective etherification and triflation, are controlled to maintain the integrity of the axial chirality throughout the synthesis. This high level of stereochemical fidelity ensures that the final phosphine ligand performs consistently in catalytic cycles, reducing the risk of batch-to-batch variability in the production of downstream pharmaceutical intermediates. Such consistency is a key metric for quality assurance teams evaluating new suppliers for critical raw materials.
How to Synthesize Biphenyl Phosphine Ligands Efficiently
The synthesis of these advanced ligands follows a logical sequence of transformations that are amenable to scale-up in a standard chemical manufacturing facility. The process begins with the activation of the biphenyl core followed by the critical desymmetry step, which sets the stereochemistry for the entire molecule. Subsequent functionalization involves protecting group strategies and cross-coupling reactions that are well-understood in organic synthesis, reducing the technical risk associated with technology transfer. Detailed standardized synthesis steps see the guide below, which outlines the specific reagents, temperatures, and molar ratios required to replicate the high yields reported in the patent examples. Adhering to these parameters is essential for maintaining the high enantiomeric excess and chemical purity that define the commercial value of these ligands.
- Perform a desymmetry ring-closing reaction between 2,2',6,6'-tetrahydroxybiphenyl and a chiral diol derivative to establish axial chirality.
- Execute selective mono-etherification followed by triflation of the remaining hydroxyl group to activate the position for phosphination.
- Conduct transition metal-catalyzed coupling with diphenylphosphine oxide followed by trichlorosilane reduction to yield the final phosphine ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ligand technology offers profound benefits that extend beyond mere technical performance metrics, directly impacting the bottom line and operational resilience of chemical manufacturing enterprises. The elimination of chiral resolution steps represents a fundamental reduction in process complexity, which inherently lowers the capital expenditure required for separation equipment and reduces the consumption of solvents and resolving agents. This simplification of the manufacturing workflow allows for faster batch turnover times, enabling suppliers to respond more agilely to fluctuations in market demand without the need for excessive inventory buffering. For procurement managers, this means a more stable supply of critical catalytic materials with reduced risk of shortage-induced price volatility. Additionally, the high reactivity and selectivity of these ligands in downstream applications mean that pharmaceutical manufacturers can achieve higher yields of their active ingredients, reducing the overall cost of goods sold for the final drug product. The ability to source ligands that enable such efficient downstream processing creates a ripple effect of cost savings throughout the entire value chain, making this technology a strategic asset for long-term partnerships.
- Cost Reduction in Manufacturing: The synthetic route described in the patent avoids the use of expensive transition metal catalysts for the chirality induction step itself, relying instead on stoichiometric organic transformations that are cheaper to execute on a large scale. By removing the need for chiral chromatography or repeated crystallization for resolution, the process significantly reduces the consumption of high-purity solvents and silica gel, which are major cost drivers in fine chemical production. The overall yield of the desired chiral ligand is theoretically doubled compared to resolution-based methods, as no material is discarded as the unwanted enantiomer. This improvement in material efficiency directly translates to substantial cost savings in raw material procurement, allowing for more competitive pricing models for the final ligand product. Furthermore, the use of common reagents like triflic anhydride and diphenylphosphine oxide ensures that the supply chain for synthesis inputs is robust and less susceptible to geopolitical disruptions.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as substituted biphenyls and commodity chiral diols, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, means that production is less likely to be halted by minor deviations in process parameters, ensuring consistent output quality. This reliability is critical for supply chain heads who must guarantee continuous availability of catalysts to prevent downtime in API manufacturing plants. The simplified purification process also reduces the lead time from reactor to shipment, allowing for quicker replenishment of stock and better alignment with just-in-time manufacturing philosophies. By integrating this technology, companies can build a more resilient supply network that is capable of withstanding external shocks while maintaining high service levels for their customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are easily transferred from laboratory glassware to industrial reactors without significant re-engineering. The reduction in waste generation, particularly the avoidance of mother liquors rich in the wrong enantiomer from resolution processes, aligns with increasingly stringent environmental regulations and corporate sustainability goals. Fewer purification steps mean lower energy consumption for heating, cooling, and solvent recovery, contributing to a smaller carbon footprint for the manufacturing operation. The use of less hazardous reagents and the potential for solvent recycling further enhance the environmental profile of the production process. For organizations committed to green chemistry principles, adopting this ligand synthesis route demonstrates a proactive approach to reducing environmental impact while maintaining technical excellence. This alignment with sustainability metrics can also provide competitive advantages in tenders where environmental compliance is a weighted evaluation criterion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel phosphine ligand technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating these ligands into their existing catalytic processes. The responses cover aspects ranging from catalytic performance comparisons to the versatility of the ligand scaffold across different metal centers.
Q: What is the primary advantage of this biphenyl ligand over traditional MOP ligands?
A: The novel biphenyl ligand achieves significantly higher conversion rates and enantiomeric excess (ee) values in asymmetric Suzuki reactions compared to MOP, while avoiding complex chiral resolution steps during synthesis.
Q: Which metal catalysts are compatible with these phosphine ligands?
A: These ligands are versatile and have been demonstrated for use with palladium, rhodium, nickel, copper, iridium, ruthenium, iron, cobalt, gold, and platinum in various asymmetric transformations.
Q: How does the desymmetry reaction impact production costs?
A: By utilizing a desymmetry strategy to transfer face chirality to axial chirality, the synthesis bypasses the need for expensive and time-consuming chiral separation processes, thereby streamlining manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphine Ligands Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in the laboratory can be reliably reproduced on an industrial scale. Our facilities are equipped with state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of phosphine ligand meets the exacting standards required for pharmaceutical grade applications. We understand the critical nature of chiral catalysts in drug synthesis and are committed to delivering products that enable our clients to achieve their own quality and efficiency targets. Our team of chemists is ready to collaborate on process optimization to further enhance the economic and environmental performance of your specific manufacturing路线.
We invite you to engage with our technical procurement team to discuss how these advanced ligands can be integrated into your supply chain to drive value and innovation. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential financial benefits specific to your production volume and current process constraints. We encourage you to contact us to obtain specific COA data and route feasibility assessments that will demonstrate the tangible advantages of partnering with us. Let us help you secure a competitive edge through superior catalytic technology and a supply partnership built on transparency, quality, and shared success.
