Advanced Pd-Catalyzed Synthesis of 3-Arylquinolin-2(1H)ones for Scalable Pharmaceutical Manufacturing
Advanced Pd-Catalyzed Synthesis of 3-Arylquinolin-2(1H)ones for Scalable Pharmaceutical Manufacturing
The development of efficient synthetic routes for bioactive heterocyclic scaffolds remains a cornerstone of modern medicinal chemistry and process development. Patent CN113045489B introduces a groundbreaking methodology for the preparation of 3-arylquinolin-2(1H)one derivatives, a privileged structural motif found in numerous therapeutic agents ranging from MAP kinase inhibitors to long-acting β2-adrenoceptor agonists. This innovation leverages a palladium-catalyzed aminocarbonylation strategy that fundamentally alters the retrosynthetic disconnection of these valuable intermediates. By utilizing benzisoxazole as a dual-purpose reagent acting simultaneously as a nitrogen source and a formyl donor, the process achieves remarkable simplicity and efficiency. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant leap forward in process intensification, offering a robust alternative to classical condensation reactions that often suffer from harsh conditions and poor atom economy.
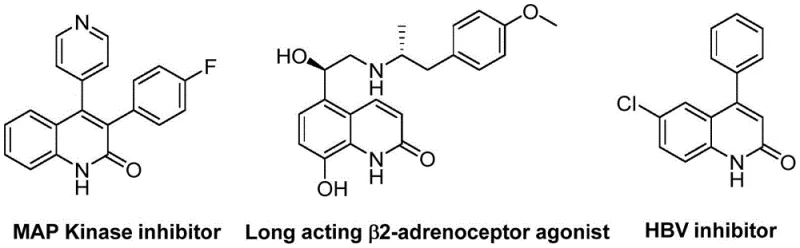
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolin-2(1H)one core has relied heavily on classical methodologies such as the Vilsmeier-Haack reaction, Knorr synthesis, or Friedlander condensation. While these methods are well-documented in academic literature, they frequently present substantial challenges when translated to industrial scale manufacturing. Traditional routes often require the use of corrosive reagents like phosphorus oxychloride or strong Lewis acids, which generate significant volumes of hazardous waste and necessitate complex downstream purification protocols. Furthermore, many conventional approaches involve multi-step sequences to install the necessary functionality at the 3-position, leading to cumulative yield losses and increased production costs. The reliance on pre-functionalized aniline derivatives also limits the scope of accessible chemical space, as sensitive functional groups may not survive the rigorous reaction conditions typically employed in these legacy processes.
The Novel Approach
In stark contrast, the methodology disclosed in the patent data utilizes a transition metal-catalyzed cascade that streamlines the synthesis into a single operational step. The core innovation lies in the strategic use of benzisoxazole derivatives, which undergo ring-opening under the catalytic conditions to provide both the nitrogen atom and the carbonyl carbon required for the lactam formation. This approach eliminates the need for external carbon monoxide gas sources or separate formylation steps, significantly enhancing the safety profile and operational simplicity of the reaction. The compatibility with a wide array of benzyl chloride electrophiles allows for the rapid generation of diverse libraries of 3-arylquinolin-2(1H)ones, facilitating the exploration of structure-activity relationships (SAR) during drug discovery phases. This modular design ensures that the process is not only scientifically elegant but also commercially viable for the production of high-purity OLED material precursors or specialty chemical intermediates.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The reaction mechanism proceeds through a sophisticated palladium catalytic cycle initiated by the oxidative addition of the benzyl chloride substrate to the active Pd(0) species generated in situ from palladium acetate and the chiral ligand (S)-BINAP. Following oxidative addition, the insertion of carbon monoxide, released thermally from the molybdenum hexacarbonyl additive, forms an acyl-palladium intermediate. Concurrently, the benzisoxazole component interacts with the metal center, undergoing N-O bond cleavage to release the nitrogen nucleophile. This nucleophilic attack on the acyl-palladium species, followed by reductive elimination and subsequent cyclization, constructs the quinolinone ring system with high regioselectivity. The inclusion of water as a stoichiometric additive plays a critical role in facilitating the hydrolysis steps necessary for the final aromatization and product release, ensuring that the catalytic turnover is maintained throughout the extended reaction period.
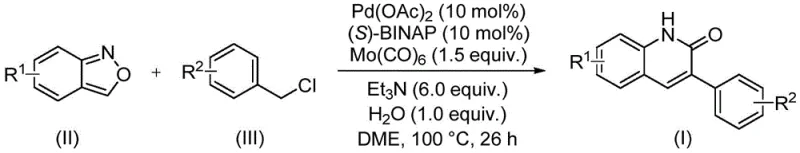
From an impurity control perspective, this catalytic system offers distinct advantages over non-catalytic thermal cyclizations. The mild reaction temperature of 100°C prevents the formation of polymeric byproducts often seen in high-temperature condensations. Moreover, the use of (S)-BINAP as a ligand, although primarily selected for electronic tuning in this context, helps stabilize the palladium center against aggregation into inactive black palladium precipitates, a common failure mode in carbonylation reactions. The high functional group tolerance observed across the substrate scope, including electron-withdrawing groups like cyano and trifluoromethyl as well as electron-donating alkoxy groups, indicates that the catalytic cycle is robust against competitive side reactions such as homocoupling of the benzyl chloride. This robustness translates directly to cleaner crude reaction profiles, reducing the burden on purification units and minimizing solvent consumption during the isolation of the final active pharmaceutical ingredient intermediates.
How to Synthesize 3-Arylquinolin-2(1H)one Efficiently
The practical execution of this synthesis is designed for ease of adoption in standard process development laboratories. The protocol involves charging a sealed reaction vessel with the requisite amounts of palladium catalyst, ligand, carbonyl source, base, and substrates in a glyme solvent system. The reaction is then heated to a moderate temperature, allowing the slow release of CO and the progression of the coupling cycle. Detailed standard operating procedures regarding specific molar ratios, solvent volumes, and work-up techniques are essential for reproducibility. For a comprehensive guide on the precise experimental parameters validated in the patent examples, please refer to the standardized synthesis steps outlined below.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride compound in a sealed vessel.
- Heat the reaction mixture to 100°C in DME solvent and maintain stirring for approximately 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this novel synthetic route are profound. The shift from multi-step, reagent-intensive classical methods to this streamlined catalytic process directly addresses the industry's demand for cost reduction in pharmaceutical intermediate manufacturing. By consolidating multiple synthetic transformations into a single pot, the process drastically reduces the total processing time and the associated utility costs for heating, cooling, and agitation. Furthermore, the elimination of hazardous gaseous carbon monoxide cylinders from the supply chain removes a significant logistical bottleneck and safety compliance burden, allowing for more flexible facility siting and reduced insurance premiums related to toxic gas storage.
- Cost Reduction in Manufacturing: The economic benefits are driven primarily by the use of commodity starting materials. Benzisoxazoles and substituted benzyl chlorides are widely available from bulk chemical suppliers at competitive price points, avoiding the need for expensive, custom-synthesized building blocks. Additionally, the catalytic loading of palladium is kept at industrially feasible levels (10 mol%), and the use of molybdenum hexacarbonyl as a solid CO source avoids the capital expenditure required for high-pressure autoclaves rated for gas handling. These factors combine to lower the overall cost of goods sold (COGS), enabling substantial cost savings that can be passed down the value chain or reinvested into further R&D initiatives.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the raw material base. Unlike specialized reagents that may have single-source suppliers or long lead times, the key inputs for this reaction are produced on a multi-ton scale globally. The reaction's high yield, consistently exceeding 90% for many substrates as demonstrated in the patent data, ensures that less starting material is required to produce a fixed amount of product, effectively stretching the purchasing budget. This efficiency minimizes the risk of production delays caused by raw material shortages and provides a buffer against market volatility in chemical pricing, ensuring a steady flow of critical intermediates to downstream formulation sites.
- Scalability and Environmental Compliance: Scaling this process from gram-scale discovery to multi-kilogram commercial production is facilitated by the homogeneous nature of the reaction mixture and the absence of exothermic hazards associated with gas sparging. The simplified work-up procedure, involving filtration and standard chromatography or crystallization, generates less aqueous waste compared to acid-base extraction heavy traditional methods. This aligns with modern green chemistry principles, reducing the environmental footprint of the manufacturing process and simplifying regulatory filings related to waste discharge and solvent recovery, which is increasingly critical for maintaining social license to operate in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and mechanistic understanding presented in the patent documentation, providing clarity for technical teams evaluating this route for potential licensing or contract manufacturing opportunities. Understanding these nuances is vital for assessing the feasibility of integrating this chemistry into existing production pipelines.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual function as both the nitrogen source and the formyl (C1) source, simplifying the reagent list and improving atom economy compared to traditional multi-component condensations.
Q: Is high-pressure carbon monoxide gas required for this reaction?
A: No, the process utilizes molybdenum hexacarbonyl (Mo(CO)6) as a solid CO surrogate, which releases carbon monoxide in situ under heating, thereby eliminating the safety hazards associated with handling high-pressure CO gas cylinders.
Q: What is the functional group tolerance of this catalytic system?
A: The method demonstrates excellent tolerance for various substituents including halogens (Cl, F), alkoxy groups (OMe), cyano groups, and bulky alkyl groups (t-Bu), allowing for the synthesis of diverse derivatives without protecting group strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced catalytic technologies in securing a competitive edge in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay. Our expertise in palladium chemistry allows us to optimize this specific aminocarbonylation process for maximum yield and minimal metal residue, addressing the critical quality attributes required for GMP manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this route for your specific target molecule. We encourage you to contact us to obtain specific COA data for our catalog compounds and to request detailed route feasibility assessments for your proprietary candidates, ensuring that your supply chain is built on the most efficient and reliable chemical foundations available today.
