Advanced Pd-Catalyzed Synthesis of 3-Arylquinolin-2(1H)one Derivatives for Commercial API Production
Introduction to Next-Generation Quinolinone Synthesis
The development of efficient synthetic routes for heterocyclic compounds remains a cornerstone of modern pharmaceutical research, particularly for scaffolds exhibiting potent biological activity. Patent CN113045489B introduces a groundbreaking preparation method for 3-arylquinolin-2(1H)one derivatives, a class of molecules renowned for their presence in various natural products and therapeutic agents ranging from antibiotics to antitumor drugs. This innovation addresses critical bottlenecks in traditional organic synthesis by leveraging a palladium-catalyzed aminocarbonylation strategy that utilizes benzisoxazole as a unique dual-purpose reagent. By acting simultaneously as the nitrogen source and the formyl donor, benzisoxazole streamlines the atomic economy of the reaction, significantly reducing the complexity of reagent handling compared to conventional gas-phase carbonylation techniques. The versatility of this approach is underscored by its ability to generate a broad spectrum of derivatives, including known bioactive structures such as MAP Kinase inhibitors and HBV inhibitors, demonstrating its immediate relevance to drug discovery pipelines.
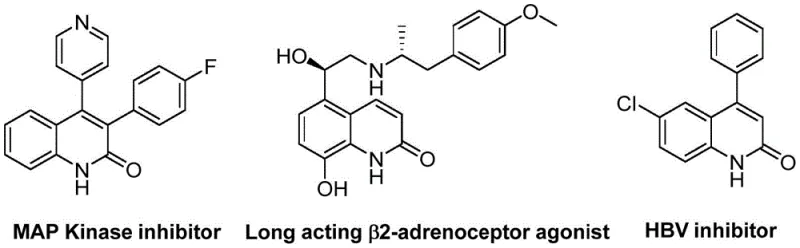
For R&D directors and process chemists, the implications of this technology extend beyond mere academic interest; it represents a tangible pathway to accessing high-value chemical space with improved operational simplicity. The method circumvents the need for hazardous carbon monoxide cylinders or unstable formylating agents, replacing them with a stable solid precursor that releases the necessary carbonyl moiety in situ. This shift not only enhances laboratory safety but also facilitates easier regulatory compliance during the scale-up phases of API manufacturing. Furthermore, the reaction conditions are optimized to tolerate a wide array of functional groups, allowing medicinal chemists to explore diverse chemical libraries without the fear of protecting group incompatibility or substrate decomposition. As the industry moves towards more sustainable and cost-effective manufacturing paradigms, the adoption of such robust catalytic systems becomes a strategic imperative for maintaining competitiveness in the global supply of pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)one derivatives has relied heavily on classical methodologies such as the Vilsmeier-Haack, Knorr, and Friedlander reactions, which, while effective, often suffer from significant drawbacks in a modern industrial context. These traditional routes frequently necessitate the use of harsh reagents, such as phosphorus oxychloride or strong acids, which generate substantial amounts of corrosive waste and require rigorous neutralization protocols prior to disposal. Moreover, many conventional strategies involve multi-step sequences where the introduction of the carbonyl functionality requires separate, distinct operations, leading to cumulative yield losses and increased processing time. Transition metal-catalyzed improvements have been attempted, yet many still rely on external carbon monoxide sources, posing severe safety risks and logistical challenges regarding gas storage and pressure management in large-scale reactors. Additionally, the substrate scope in older methods is often limited by poor functional group tolerance, forcing chemists to employ tedious protecting group strategies that further inflate the cost of goods and extend the overall production timeline.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data revolutionizes this landscape by integrating the nitrogen and carbonyl sources into a single, stable molecular entity: benzisoxazole. This innovative strategy employs a palladium catalyst system, specifically utilizing palladium acetate in conjunction with (S)-BINAP and molybdenum hexacarbonyl, to facilitate a seamless coupling with benzyl chloride derivatives. The reaction proceeds under relatively mild thermal conditions, typically around 100°C in a dimethoxyethane (DME) solvent system, which is far more manageable than the extreme temperatures or pressures required by legacy technologies. By eliminating the need for gaseous CO and toxic formylating agents, this method drastically simplifies the reactor setup and reduces the environmental footprint associated with the synthesis. The result is a highly efficient, one-pot transformation that delivers 3-arylquinolin-2(1H)one derivatives with exceptional yields, often exceeding 90%, while maintaining a clean impurity profile that minimizes downstream purification burdens.
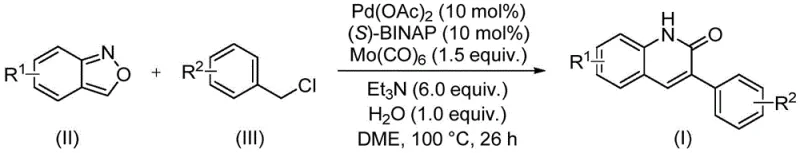
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The core of this technological advancement lies in the sophisticated interplay between the palladium catalyst and the unique reactivity of the benzisoxazole ring. Mechanistically, the cycle initiates with the oxidative addition of the benzyl chloride to the active Pd(0) species, generating an organopalladium intermediate. Subsequently, the molybdenum hexacarbonyl serves as a solid-state carbon monoxide surrogate, releasing CO in situ which then inserts into the palladium-carbon bond to form an acyl-palladium complex. The critical step involves the nucleophilic attack by the nitrogen of the benzisoxazole, followed by the ring-opening of the isoxazole moiety. This ring-opening event is pivotal as it simultaneously provides the nitrogen atom for the quinolinone core and the oxygen atom that eventually forms the lactam carbonyl, effectively acting as an internal formyl source. This intricate cascade is facilitated by the specific ligand environment provided by (S)-BINAP, which stabilizes the palladium center and promotes the necessary reductive elimination to close the catalytic cycle and release the final product.
From an impurity control perspective, this mechanism offers distinct advantages over random radical pathways or uncontrolled thermal decompositions often seen in harsher conditions. The specificity of the palladium cycle ensures that side reactions, such as homocoupling of the benzyl chloride or polymerization of the intermediates, are minimized. The use of water as an additive, as specified in the patent claims, plays a subtle yet crucial role in facilitating the hydrolysis steps required for the final aromatization and tautomerization of the quinolinone ring. This controlled environment allows for the accommodation of sensitive functional groups like cyano, trifluoromethyl, and various halogens without degradation. For quality assurance teams, this translates to a much cleaner crude reaction mixture, reducing the load on chromatographic columns and crystallization steps, ultimately leading to a higher purity final API intermediate that meets stringent regulatory specifications with less effort.
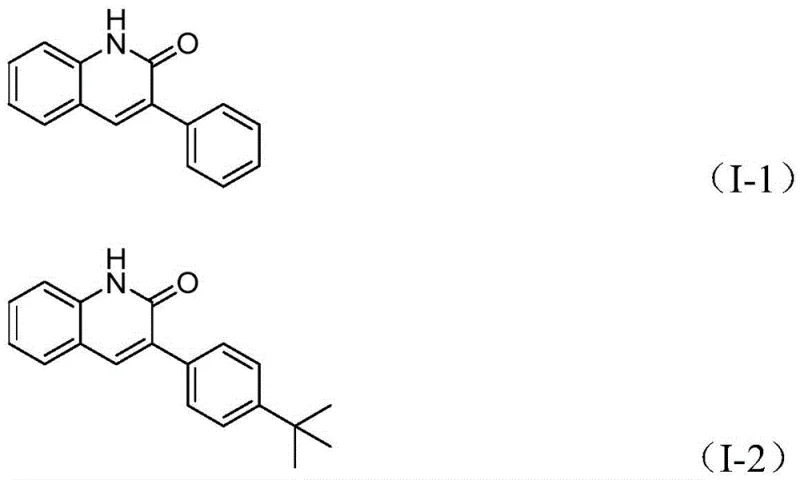
How to Synthesize 3-Arylquinolin-2(1H)one Efficiently
Implementing this synthesis in a practical setting requires precise adherence to the optimized parameters established in the patent to ensure reproducibility and maximum yield. The process is designed to be operationally simple, requiring standard laboratory equipment such as sealed tubes or pressure-rated reactors capable of maintaining 100°C. The stoichiometry is carefully balanced, typically employing a slight excess of the benzyl chloride relative to the benzisoxazole to drive the reaction to completion, while the catalyst loading is kept at an industrially viable level of 10 mol%. The reaction time is optimized at approximately 26 hours, a duration that balances complete conversion with throughput efficiency. Following the reaction, the workup procedure is straightforward, involving filtration to remove insoluble catalyst residues and metal salts, followed by standard silica gel chromatography for final purification. Detailed standardized synthesis steps see the guide below.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in a sealed vessel with DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion of starting materials.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 3-arylquinolin-2(1H)one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The primary driver of cost reduction lies in the simplification of the raw material portfolio; benzisoxazoles and benzyl chlorides are commodity chemicals that are widely available from multiple global suppliers, ensuring a robust and competitive sourcing landscape. Unlike processes that require specialized gas handling infrastructure for carbon monoxide or the procurement of expensive, moisture-sensitive formylating agents, this method utilizes stable solids and liquids that can be stored and handled with standard safety protocols. This reduction in hazard classification lowers insurance premiums and warehousing costs, while the elimination of complex gas delivery systems reduces capital expenditure on reactor modifications. Furthermore, the high atom economy and yield efficiency mean that less raw material is wasted per kilogram of product produced, directly lowering the variable cost of manufacturing.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the use of palladium acetate, which, while a precious metal, is used at low loading levels and can potentially be recovered from the filtration waste streams, further mitigating material costs. The avoidance of cryogenic conditions or high-pressure autoclaves significantly reduces energy consumption, as the reaction proceeds at a moderate 100°C using standard heating mantles or oil baths. Additionally, the simplified workup procedure reduces the consumption of solvents and silica gel during purification, which are often hidden cost drivers in fine chemical production. By consolidating the nitrogen and carbon sources into one reagent, the inventory management becomes less complex, reducing the administrative overhead associated with tracking multiple hazardous precursors.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved due to the reliance on bench-stable reagents that do not suffer from the logistical fragility of compressed gases or short-shelf-life intermediates. Benzisoxazole derivatives are synthesized via well-established industrial processes, meaning that supply disruptions are rare and lead times are predictable. This stability allows for better production planning and inventory buffering, ensuring that API manufacturing schedules are not derailed by raw material shortages. The robustness of the reaction conditions also means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without the need for highly specialized equipment, providing flexibility in case of regional supply chain disruptions or capacity constraints.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the homogeneous nature of the reaction and the absence of exothermic gas evolution events that are difficult to manage in large vessels. The waste profile is significantly greener, as the primary byproducts are benign organic fragments rather than heavy metal sludges or acidic aqueous wastes typical of Vilsmeier-type reactions. This aligns perfectly with increasingly stringent environmental regulations, reducing the cost and complexity of waste treatment and disposal. The ability to run the reaction in common solvents like DME, which can be recycled, further enhances the sustainability credentials of the process, making it an attractive option for companies aiming to reduce their carbon footprint and meet corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is critical for R&D teams evaluating the feasibility of this route for their specific target molecules and for procurement teams assessing the risk profile of the supply chain. The answers reflect the balance between high-performance chemistry and practical manufacturability that defines this innovation.
Q: What are the primary advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl (carbonyl) source, eliminating the need for separate carbon monoxide gas or toxic formylating agents, thereby simplifying the reaction setup and improving safety profiles.
Q: Does this method tolerate diverse functional groups on the substrate?
A: Yes, the patented methodology demonstrates excellent functional group tolerance, accommodating substituents such as halogens, alkoxy groups, cyano, and trifluoromethyl groups on both the benzisoxazole and benzyl chloride components without significant yield loss.
Q: Is this process suitable for large-scale manufacturing?
A: The process utilizes commercially available catalysts and reagents under relatively mild thermal conditions (100°C), making it highly amenable to scale-up from laboratory benchtop to multi-ton commercial production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN113045489B for accelerating drug development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with the highest safety standards, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 3-arylquinolin-2(1H)one intermediate meets your exact requirements. We understand that consistency is key in pharmaceutical manufacturing, and our process engineering team is dedicated to optimizing every parameter to maximize yield and minimize impurities.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us be your partner in turning complex chemical challenges into commercial successes.
