Advanced Palladium-Catalyzed Synthesis of 3-Arylquinoline-2(1H) Ketones for Commercial Scale-up
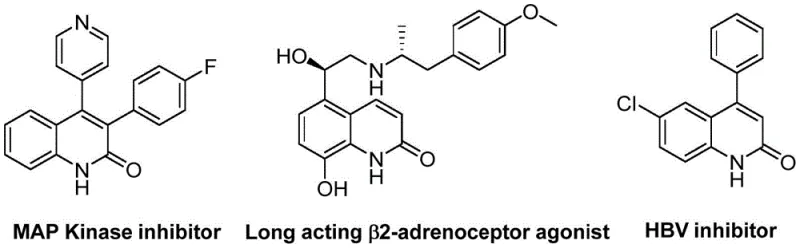
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic compounds due to their prevalence in bioactive molecules. Patent CN113045489B discloses a groundbreaking preparation method for 3-arylquinoline-2(1H) ketone derivatives, a scaffold found in numerous therapeutic agents ranging from antibiotics to antitumor drugs. This technology leverages a palladium-catalyzed aminocarbonylation strategy that fundamentally shifts the paradigm of quinolinone synthesis by utilizing benzisoxazole as a dual-purpose reagent. The significance of this chemical class is underscored by its presence in potent MAP Kinase inhibitors and HBV inhibitors, highlighting the critical need for efficient manufacturing processes. By addressing the limitations of prior art, this innovation provides a reliable pharmaceutical intermediate supplier pathway that ensures high purity and structural integrity for downstream drug development.
Historically, the construction of the quinolin-2(1H)one core has relied heavily on classical condensation reactions such as the Vilsmeier-Haack, Knorr, or Friedlander methodologies. These conventional methods often impose severe constraints on the synthetic chemist, including the requirement for harsh acidic or basic conditions that can degrade sensitive functional groups on the substrate. Furthermore, traditional approaches frequently struggle with regioselectivity issues and often necessitate multi-step sequences to install the carbonyl functionality, leading to accumulated yield losses and increased waste generation. The reliance on pre-functionalized aniline derivatives in older protocols also limits the diversity of accessible analogs, creating bottlenecks in the rapid exploration of structure-activity relationships during early-stage drug discovery campaigns.
In stark contrast, the novel approach detailed in the patent data introduces a streamlined catalytic cycle that merges carbonylation and cyclization into a single operational step. This method employs a sophisticated catalyst system comprising palladium acetate and (S)-1,1'-binaphthyl-2,2'-bis-diphenylphosphine, activated by carbonyl molybdenum as a safe carbon monoxide surrogate. The reaction proceeds smoothly at 100°C in ethylene glycol dimethyl ether, demonstrating exceptional tolerance towards various substituents such as halogens, alkoxy groups, and cyano moieties. This transition metal-catalyzed improvement not only simplifies the operational workflow but also significantly enhances the atom economy of the transformation, representing a major leap forward in cost reduction in API manufacturing.
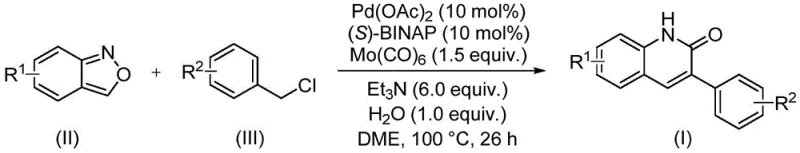
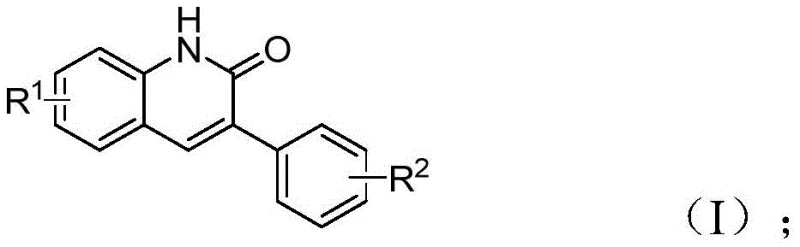
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation reveal a complex interplay between the transition metal center and the unique reactivity of benzisoxazole. The catalytic cycle initiates with the oxidative addition of the benzyl chloride to the active palladium species, followed by the insertion of carbon monoxide generated in situ from the molybdenum complex. Crucially, the benzisoxazole ring undergoes cleavage to release the necessary nitrogen atom and the formyl group simultaneously, a feature that eliminates the need for external formylating agents. This intricate mechanism ensures that the carbonyl group is installed precisely at the C-2 position while the aryl group from the benzyl chloride occupies the C-3 position, yielding the target 3-arylquinoline-2(1H) ketone structure with high regiocontrol.
Impurity control is inherently managed through the high selectivity of the palladium catalyst system, which minimizes the formation of side products commonly associated with radical pathways or uncontrolled polymerization. The use of water as a co-reagent in specific molar ratios further aids in stabilizing the catalytic species and facilitating the hydrolysis steps required for the final aromatization of the quinolinone ring. By maintaining strict control over reaction parameters such as temperature and stoichiometry, the process ensures that the final crude mixture contains minimal byproducts, thereby simplifying the downstream purification burden. This level of mechanistic precision is vital for producing high-purity OLED material or pharmaceutical intermediates where trace impurities can compromise biological efficacy or material performance.
How to Synthesize 3-Arylquinoline-2(1H) Ketone Efficiently
Executing this synthesis requires careful attention to the stoichiometric balance of the catalyst system and the quality of the starting materials to ensure reproducible results. The standardized protocol involves charging a sealed tube with the palladium catalyst, chiral ligand, carbonyl source, base, and substrates before heating to the specified temperature range. Detailed standard operating procedures regarding the exact addition order and workup techniques are critical for maximizing yield and safety in a production environment. The following guide outlines the essential steps derived from the patent examples to assist technical teams in replicating this efficient transformation.
- Combine palladium acetate, (S)-BINAP, carbonyl molybdenum, triethylamine, water, benzisoxazole, and benzyl chloride in DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Perform post-processing including filtration and silica gel mixing, followed by column chromatography purification to isolate the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial cost savings by eliminating the need for expensive, specialized formylating reagents and reducing the number of unit operations required. The starting materials, specifically benzisoxazole and substituted benzyl chlorides, are commodity chemicals that are widely available from global chemical suppliers, ensuring a stable and resilient supply chain. The operational simplicity of the reaction, which avoids extreme pressures or cryogenic temperatures, translates directly into lower energy consumption and reduced capital expenditure on specialized reactor equipment. These factors collectively contribute to a more economical manufacturing process that enhances the overall competitiveness of the final active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The elimination of transition metal removal steps typically associated with other catalytic processes is not required here due to the efficient nature of the reaction, but more importantly, the use of benzisoxazole as a dual source drastically cuts raw material costs. By combining the nitrogen and carbon sources into a single molecule, the process reduces the total mass of reagents required per kilogram of product, leading to significant waste reduction. This streamlined material usage directly impacts the bottom line, allowing for more competitive pricing strategies in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing risks are minimized because the key reactants are not proprietary or restricted substances, allowing procurement managers to qualify multiple vendors for raw material supply. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by equipment failures or environmental constraints, ensuring consistent delivery timelines. This reliability is crucial for maintaining continuous manufacturing flows for critical medicines, reducing the lead time for high-purity pharmaceutical intermediates and preventing stockouts in the downstream supply chain.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential as it does not rely on hazardous gases like pressurized carbon monoxide, using a solid surrogate instead which is safer to handle on a multi-ton scale. The solvent system and reaction byproducts are manageable within standard waste treatment protocols, facilitating compliance with increasingly stringent environmental regulations. This ease of scale-up from laboratory to commercial production ensures that the technology can meet growing market demand efficiently while maintaining a low environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity on process capabilities. Understanding these details helps stakeholders make informed decisions regarding technology adoption and partnership opportunities.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual function as both the nitrogen source and the formyl source, which simplifies the reaction stoichiometry and reduces the need for multiple expensive reagents, thereby lowering overall material costs.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the reaction utilizes commercially available starting materials and operates at moderate temperatures (100°C), making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates without requiring extreme pressure or cryogenic conditions.
Q: How does this method compare to traditional Friedlander reactions?
A: Unlike traditional methods that often suffer from harsh conditions and limited functional group tolerance, this palladium-catalyzed approach offers wider substrate compatibility and higher reaction efficiency, significantly reducing lead time for high-purity intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinoline-2(1H) Ketone Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced synthetic technology to support your drug development pipeline with high-quality intermediates. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from clinical trials to market launch. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-arylquinoline-2(1H) ketone derivative meets the highest industry standards for safety and efficacy.
We invite you to engage with our technical procurement team to discuss how this innovative route can optimize your specific manufacturing requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this method for your portfolio. Please contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, and let us help you accelerate your path to commercial success.
