Advanced Manufacturing of 5-Trifluoromethyl-1,2,4-Triazoles: A Metal-Free Synthetic Breakthrough
Advanced Manufacturing of 5-Trifluoromethyl-1,2,4-Triazoles: A Metal-Free Synthetic Breakthrough
The integration of trifluoromethyl groups into nitrogen-containing heterocycles represents a cornerstone strategy in modern medicinal chemistry, profoundly influencing the pharmacokinetic profiles of drug candidates. As illustrated in the structural diversity of bioactive molecules such as deferasirox and fluconazole shown below, the 1,2,4-triazole scaffold is ubiquitous in high-value pharmaceuticals and functional materials. Patent CN110467579B introduces a transformative preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds that addresses long-standing synthetic challenges. This technology leverages a non-metallic iodine-promoted cyclization strategy, offering a robust alternative to traditional methods that often rely on hazardous reagents or expensive transition metal catalysts. For R&D directors and procurement specialists alike, this innovation signals a shift towards more sustainable and economically viable manufacturing pathways for complex heterocyclic intermediates.
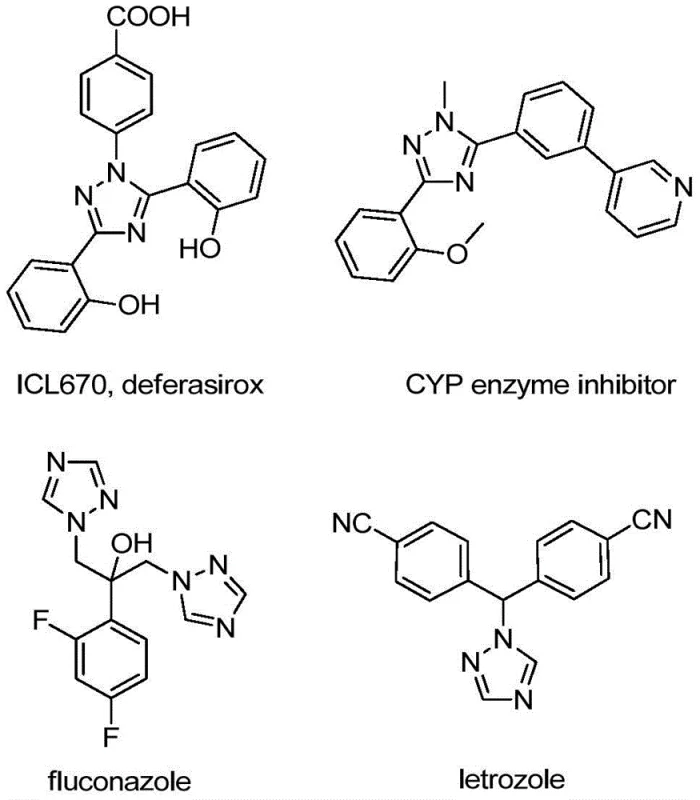
The significance of this patent extends beyond mere academic interest; it provides a practical blueprint for the commercial scale-up of complex pharmaceutical intermediates. By utilizing readily available starting materials like hydrazones and trifluoroacetimidoyl chlorides, the process eliminates the need for specialized, moisture-sensitive environments typically required for organometallic transformations. This operational simplicity translates directly into reduced capital expenditure for reactor conditioning and lower operational overheads. Furthermore, the broad substrate tolerance described in the patent suggests that this methodology can be adapted for a wide array of derivatives, making it a versatile tool for process chemists aiming to optimize supply chains for API precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a trifluoromethyl group onto a triazole ring has been fraught with synthetic difficulties and safety concerns. Conventional literature reports primarily describe two approaches: the direct trifluoromethylation of pre-synthesized nitrogen heterocycles or the cycloaddition of trifluoromethyl synthons. The former often necessitates the use of specialized trifluoromethylating reagents, such as Togni or Umemoto reagents, which are prohibitively expensive for large-scale manufacturing and generate significant stoichiometric waste. Alternatively, methods employing trifluorodiazoethane pose severe safety risks due to the explosive nature of diazo compounds, requiring specialized equipment and rigorous safety protocols that inflate production costs. Additionally, many traditional routes rely heavily on transition metal catalysis, which introduces the persistent challenge of residual metal removal to meet stringent ICH Q3D guidelines for pharmaceutical products.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN110467579B offers a streamlined, metal-free alternative that fundamentally simplifies the synthetic workflow. The core innovation lies in the reaction between trifluoroacetimidoyl chloride and hydrazones, promoted by elemental iodine and sodium acetate. As depicted in the general reaction scheme below, this approach bypasses the need for unstable diazo species or costly fluorinating agents. The reaction proceeds efficiently in common organic solvents like dichloroethane (DCE) at moderate temperatures of 80°C. This shift not only enhances the safety profile of the manufacturing process but also drastically reduces the complexity of the downstream purification steps. By avoiding heavy metal catalysts, manufacturers can eliminate expensive scavenging steps, thereby achieving substantial cost reduction in pharmaceutical intermediate manufacturing while ensuring higher purity profiles.
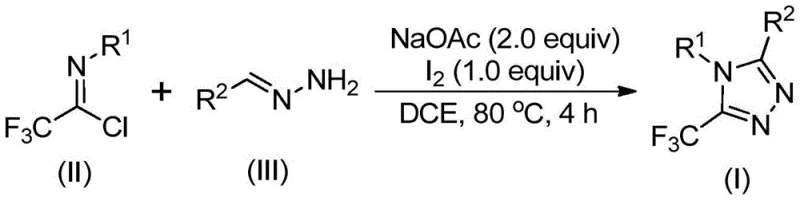
Mechanistic Insights into Iodine-Promoted Oxidative Cyclization
The mechanistic pathway of this transformation is a sophisticated example of base-promoted oxidative cyclization, offering deep insights for process optimization. The reaction likely initiates with a base-mediated intermolecular carbon-nitrogen bond formation between the trifluoroacetimidoyl chloride and the hydrazone, generating a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization to align the reactive centers for cyclization. The critical role of elemental iodine cannot be overstated; it acts as a mild oxidant that facilitates the oxidative iodination of the intermediate. This step generates an electrophilic iodine species that triggers an intramolecular electrophilic substitution, closing the triazole ring. The final aromatization step yields the stable 5-trifluoromethyl-1,2,4-triazole product. Understanding this mechanism allows chemists to fine-tune the stoichiometry of iodine and base to maximize yield and minimize side reactions.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based trifluoromethylation methods. The ionic nature of the cyclization pathway tends to produce cleaner reaction profiles with fewer byproducts derived from radical recombination or hydrogen abstraction. The use of sodium acetate as a mild base ensures that sensitive functional groups on the aryl rings of the hydrazone or imidoyl chloride remain intact, demonstrating excellent functional group tolerance. This selectivity is crucial for maintaining high purity specifications without the need for aggressive chromatographic separations. The ability to tolerate various substituents, including halogens, alkyl groups, and electron-withdrawing nitro groups, as evidenced by the diverse substrate scope in the patent, confirms the robustness of this catalytic system for producing high-purity OLED material precursors and pharmaceutical building blocks.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible route for generating these valuable heterocycles. The process begins with the precise mixing of sodium acetate, trifluoroacetimidoyl chloride, and the corresponding hydrazone in an aprotic organic solvent. The reaction mixture is then heated to promote the initial condensation, followed by the strategic addition of elemental iodine to drive the oxidative cyclization to completion. Detailed standard operating procedures regarding specific molar ratios, solvent volumes, and workup techniques are essential for ensuring consistent quality across batches. For a comprehensive guide on the exact experimental parameters and purification methods validated by the patent data, please refer to the standardized synthesis steps provided below.
- Mix sodium acetate, trifluoroacetimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane (DCE).
- Heat the reaction mixture to 80°C and stir for 2 to 4 hours to facilitate the initial condensation.
- Add elemental iodine to the system and continue heating for an additional 1 to 2 hours to promote oxidative cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted synthesis route offers compelling economic and logistical benefits that extend far beyond the laboratory bench. The elimination of precious metal catalysts removes a significant variable from the cost structure, shielding the supply chain from the volatility of metal markets. Furthermore, the reliance on commodity chemicals like hydrazones and simple imidoyl chlorides ensures a stable and diversified supply base, reducing the risk of single-source bottlenecks. The operational simplicity of the process, which does not demand rigorous anhydrous or anaerobic conditions, allows for utilization of standard manufacturing infrastructure, thereby accelerating the timeline from pilot plant to commercial production.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete avoidance of expensive transition metal catalysts and specialized trifluoromethylating reagents. By utilizing elemental iodine, a low-cost and abundant promoter, the direct material costs are significantly lowered. Moreover, the absence of heavy metals simplifies the downstream processing; there is no need for costly metal scavenger resins or complex filtration systems to meet regulatory limits for metal residues. This streamlining of the purification train results in higher overall throughput and reduced waste disposal costs, contributing to substantial cost savings in API manufacturing.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, particularly the hydrazones and trifluoroacetimidoyl chlorides, are derived from widely available aldehydes and amines. This reliance on bulk chemical feedstocks ensures a reliable supply chain that is less susceptible to the disruptions often seen with niche, custom-synthesized reagents. The robustness of the reaction conditions means that production can be maintained consistently without the need for specialized inert atmosphere equipment, reducing downtime and maintenance requirements. This reliability is critical for maintaining continuous supply lines for high-purity pharmaceutical intermediates in a global market.
- Scalability and Environmental Compliance: The process has been demonstrated to be easily scalable, with the patent noting successful expansion to gram-level synthesis without loss of efficiency. The use of dichloroethane, while requiring proper handling, is a well-understood solvent in industrial settings with established recovery and recycling protocols. The atom economy of the reaction is favorable compared to multi-step alternatives, generating less chemical waste per kilogram of product. This alignment with green chemistry principles facilitates easier environmental compliance and permitting, smoothing the path for large-scale commercialization of complex polymer additives or agrochemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within patent CN110467579B, providing a factual basis for evaluating the feasibility of this route for your specific applications. Understanding these nuances is vital for making informed decisions about process adoption and vendor selection.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the process described in patent CN110467579B utilizes elemental iodine as a promoter instead of expensive heavy metal catalysts like palladium or copper, significantly reducing purification costs and metal residue risks.
Q: What are the optimal reaction conditions for this triazole formation?
A: The optimal conditions involve heating the reactants in an aprotic solvent like dichloroethane (DCE) at 80°C, using a molar ratio of trifluoroacetimidoyl chloride to hydrazone of approximately 1:2.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method avoids strict anhydrous or anaerobic conditions and uses commercially available raw materials, making it highly scalable for industrial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient, metal-free synthetic routes in the modern pharmaceutical landscape. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN110467579B are fully realized in a GMP-compliant manufacturing environment. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle fluorinated chemistry safely and efficiently positions us as a preferred partner for companies seeking to optimize their supply chains for next-generation therapeutics.
We invite you to collaborate with us to evaluate the feasibility of this iodine-promoted triazole synthesis for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that compares this novel route against your current supply options. Please contact us today to request specific COA data for our existing triazole inventory or to discuss route feasibility assessments for your custom synthesis projects. Let us help you secure a competitive advantage through superior chemical manufacturing.
