Advanced Photocatalytic Synthesis of Trifluoromethyl Imidazo[1,2-a]pyridines for Pharmaceutical Applications
Advanced Photocatalytic Synthesis of Trifluoromethyl Imidazo[1,2-a]pyridines for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust methodologies to introduce fluorine-containing motifs into drug candidates, driven by the profound impact of the trifluoromethyl group on metabolic stability, lipophilicity, and bioavailability. Patent CN110590774B presents a groundbreaking advancement in this domain by disclosing a novel preparation method for trifluoromethyl imidazole fused-ring compounds. This technology leverages visible light photocatalysis to achieve direct C3-trifluoromethylation of imidazo[1,2-a]pyridine scaffolds, a structural backbone found in numerous bioactive molecules such as Zolpidem and Olprinone. Unlike traditional approaches that rely on harsh conditions, this invention utilizes environmentally benign organic acridinium salts as photocatalysts, enabling the reaction to proceed efficiently at room temperature. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this patent offers a pathway to high-purity compounds with significantly reduced environmental footprints and operational costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct functionalization of imidazopyridines at the C3 position has been fraught with significant chemical and operational challenges. Conventional trifluoromethylation strategies often necessitate the use of expensive transition metal catalysts, such as palladium or copper complexes, which introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). Furthermore, these traditional routes frequently require stoichiometric amounts of strong oxidants or aggressive acidic and basic conditions to drive the reaction forward. Such harsh environments not only limit the substrate scope, causing decomposition of sensitive functional groups, but also generate substantial quantities of hazardous waste. The downstream processing becomes equally burdensome, requiring complex purification steps to remove metal residues and by-products, thereby inflating the overall cost of goods and extending the lead time for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN110590774B revolutionizes the synthesis landscape by employing a metal-free, visible-light-driven protocol. This innovative approach utilizes sodium trifluoromethanesulfinate as a stable and inexpensive trifluoromethyl radical source, coupled with organic acridinium photocatalysts that absorb visible light to initiate the reaction cycle. The process operates under exceptionally mild conditions, typically at room temperature and under a neutral atmosphere, which preserves the integrity of diverse functional groups on the substrate. As illustrated in the reaction scheme below, the transformation is direct and efficient, converting readily available imidazo[1,2-a]pyridines into valuable trifluoromethylated derivatives with high selectivity.
![Reaction scheme showing visible light photocatalytic trifluoromethylation of 2-phenylimidazo[1,2-a]pyridine](/insights/img/trifluoromethyl-imidazole-photocatalysis-pharma-supplier-20260306101256-04.webp)
This shift from thermal, metal-catalyzed processes to photochemical, organocatalytic ones represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing. By eliminating the need for transition metals and harsh reagents, the novel approach simplifies the workflow, reduces safety hazards associated with strong oxidants, and facilitates a greener production cycle that aligns with modern regulatory standards for environmental compliance.
Mechanistic Insights into Visible Light Photocatalytic C-H Activation
The core of this technological breakthrough lies in the sophisticated interplay between the organic photocatalyst and the visible light source. The reaction mechanism initiates when the acridinium salt photocatalyst absorbs photons from the visible light spectrum, transitioning to an excited state with high oxidative potential. This excited species acts as a potent single-electron oxidant, capable of abstracting an electron from the trifluoromethanesulfinate anion. This single-electron transfer (SET) event triggers the fragmentation of the sulfinate salt, releasing sulfur dioxide and generating the highly reactive trifluoromethyl radical. This radical species then selectively attacks the electron-rich C3 position of the imidazo[1,2-a]pyridine ring system. The resulting radical intermediate undergoes subsequent oxidation and deprotonation steps to restore aromaticity, yielding the final trifluoromethylated product while regenerating the ground-state photocatalyst to continue the cycle.
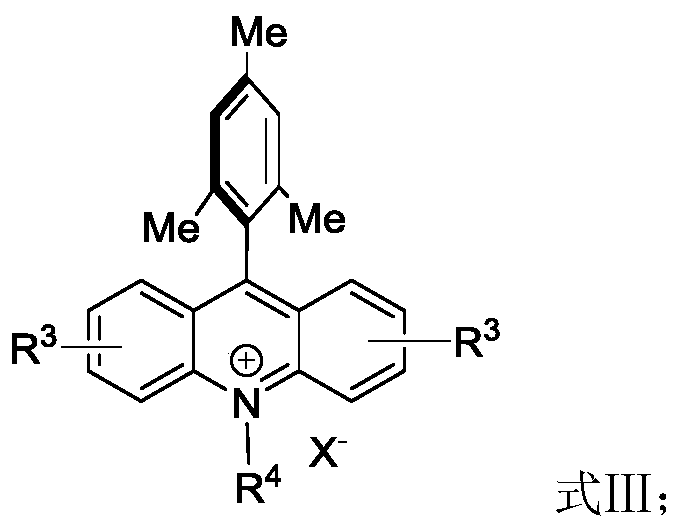
From an impurity control perspective, this radical mechanism offers distinct advantages over ionic pathways. The mildness of the room temperature conditions minimizes thermal degradation and prevents the formation of polymeric by-products often seen in high-temperature reactions. Furthermore, the specificity of the radical attack at the C3 position ensures high regioselectivity, reducing the burden on downstream purification teams. The use of organic acridinium salts, whose structure is depicted above, avoids the introduction of inorganic metal ions, thereby simplifying the impurity profile and ensuring that the final product meets stringent quality specifications required for clinical applications without the need for specialized metal scavenging resins.
How to Synthesize 2-Phenyl-3-trifluoromethylimidazo[1,2-a]pyridine Efficiently
Implementing this photocatalytic protocol in a laboratory or pilot plant setting is straightforward and requires standard equipment available in most modern synthetic facilities. The procedure involves dissolving the imidazo[1,2-a]pyridine substrate and sodium trifluoromethanesulfinate in a suitable solvent such as 1,2-dichloroethane, followed by the addition of a catalytic amount of the acridinium salt. The mixture is then subjected to irradiation from a visible light source, such as a blue LED array, under an inert nitrogen atmosphere to prevent oxygen quenching of the radical intermediates. After the reaction reaches completion, typically within 24 hours, the workup involves simple filtration through celite to remove the catalyst, concentration of the filtrate, and purification via flash column chromatography. The detailed standardized synthesis steps are provided in the guide below.
- Combine 2-phenylimidazo[1,2-a]pyridine (0.3 mmol), sodium trifluoromethanesulfinate (0.2 mmol), and an acridinium photocatalyst (0.01 mmol) in 1,2-dichloroethane (2.0 mL).
- Irradiate the reaction mixture with a blue LED light source (3W) at room temperature under a nitrogen atmosphere for approximately 24 hours.
- Filter the reaction mixture through celite, concentrate the filtrate under reduced pressure, and purify the crude product via column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The elimination of precious transition metals removes a significant variable from the raw material cost structure, shielding the supply chain from the volatility of metal markets. Additionally, the use of commodity chemicals like sodium trifluoromethanesulfinate and common organic solvents ensures a stable and reliable supply of inputs, mitigating the risk of production delays caused by sourcing bottlenecks. The operational simplicity of running reactions at room temperature also drastically reduces energy consumption compared to thermal processes requiring heating or cryogenic cooling, contributing to substantial cost savings in utility expenditures over the lifecycle of the product.
- Cost Reduction in Manufacturing: The economic model of this synthesis is heavily favored by the absence of expensive noble metal catalysts and the use of low-cost organic dyes. By removing the necessity for complex metal removal steps, such as charcoal treatment or resin scavenging, the overall processing time is shortened, and yield losses during purification are minimized. This streamlined workflow directly lowers the cost of goods sold (COGS), allowing for more competitive pricing in the global market for fine chemical intermediates while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reliance on bench-stable reagents and ambient reaction conditions enhances the robustness of the manufacturing process. Unlike methods requiring sensitive organometallic reagents that demand strict anhydrous conditions and cold chain logistics, the reagents in this protocol are easy to handle and store. This ease of handling reduces the likelihood of batch failures due to reagent degradation or operator error, ensuring consistent delivery schedules and strengthening the reliability of the supply chain for downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge, but advancements in LED technology and flow chemistry have made this highly feasible. The process generates minimal hazardous waste, as it avoids heavy metals and strong oxidants, simplifying waste disposal and reducing environmental compliance costs. The ability to run the reaction under visible light, potentially even utilizing sunlight in certain configurations, underscores its alignment with green chemistry principles, making it an attractive option for companies aiming to reduce their carbon footprint and meet sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic trifluoromethylation technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on substrate scope, reaction parameters, and product quality. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing production pipelines.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN110590774B utilizes organic acridinium salts as photocatalysts, completely eliminating the need for costly and toxic transition metals like palladium or iridium.
Q: What are the typical reaction conditions for this trifluoromethylation?
A: The reaction proceeds under mild conditions, specifically at room temperature using visible light irradiation (such as blue LEDs or sunlight) in common organic solvents like 1,2-dichloroethane.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is highly scalable due to the use of inexpensive reagents, ambient temperature operation, and simple post-treatment procedures involving filtration and standard chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Imidazo[1,2-a]pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photocatalytic synthesis methods described in CN110590774B for the next generation of pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of trifluoromethylated imidazole derivatives delivered to our clients adheres to the highest international quality standards.
We invite global pharmaceutical and agrochemical companies to collaborate with us to leverage this green and efficient synthetic route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in photocatalytic chemistry can optimize your supply chain and accelerate your drug development timelines.
