Advanced Photocatalytic Synthesis of Trifluoromethyl Imidazo[1,2-a]pyridines for Commercial Drug Development
Advanced Photocatalytic Synthesis of Trifluoromethyl Imidazo[1,2-a]pyridines for Commercial Drug Development
The introduction of trifluoromethyl groups into heterocyclic scaffolds is a critical strategy in modern medicinal chemistry, significantly enhancing the metabolic stability, lipophilicity, and bioavailability of drug candidates. Patent CN110590774B presents a groundbreaking approach to synthesizing trifluoromethyl imidazole fused-ring compounds, specifically targeting the C3 position of imidazo[1,2-a]pyridines and related heterocycles. This technology leverages visible light photocatalysis to achieve direct C-H functionalization, bypassing the need for pre-functionalized starting materials. The general structural framework of these high-value intermediates is depicted below, showcasing the versatility of the A and B ring systems which can be tuned for specific biological targets.
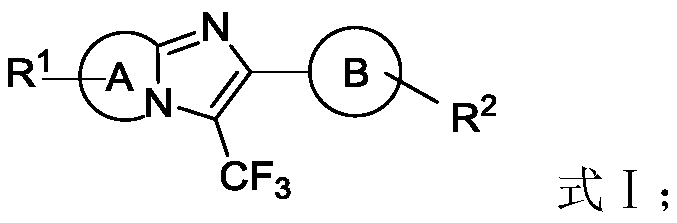
For R&D directors and process chemists, the ability to install a trifluoromethyl group under mild conditions represents a significant advancement over classical methodologies. Traditional routes often suffer from poor atom economy, the generation of hazardous waste, and the necessity for rigorous purification to remove toxic metal residues. By utilizing an organic photocatalyst system driven by visible light, this invention offers a greener, more sustainable pathway to complex nitrogen-containing heterocycles. This report analyzes the technical merits of this process and its implications for cost-effective manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct trifluoromethylation of imidazo[1,2-a]pyridines has been fraught with challenges that hinder efficient scale-up and commercial viability. Conventional synthetic strategies typically rely on the use of stoichiometric amounts of expensive transition metal catalysts, such as copper or palladium complexes, which not only drive up raw material costs but also introduce significant downstream processing burdens. The removal of trace heavy metals to meet stringent pharmaceutical specifications often requires additional chelation steps or specialized chromatography, extending production lead times. Furthermore, many existing protocols necessitate the use of strong oxidants or harsh acidic/basic conditions, which can degrade sensitive functional groups on the heterocyclic core, leading to complex impurity profiles and reduced overall yields.
The Novel Approach
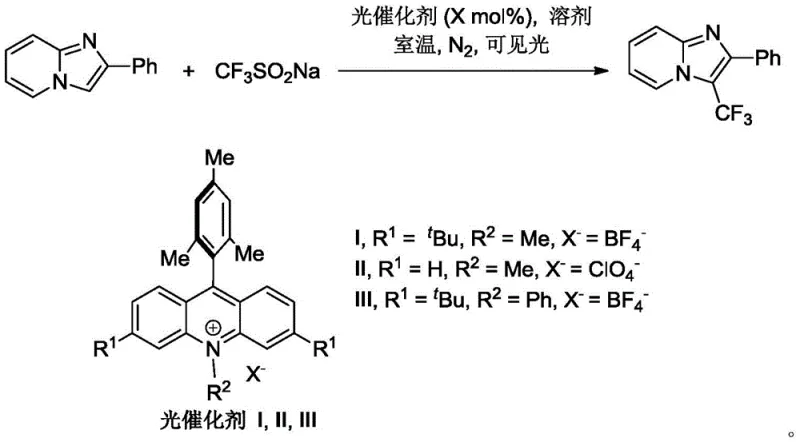
In stark contrast, the methodology disclosed in the patent utilizes a visible-light-mediated radical mechanism that operates efficiently at room temperature. By employing sodium trifluoromethanesulfinate as a safe and stable trifluoromethyl source, combined with an organic acridinium photocatalyst, the reaction proceeds without the need for external oxidants or transition metals. As illustrated in the specific reaction scheme for the synthesis of 2-phenyl-3-trifluoromethylimidazo[1,2-a]pyridine, the process is remarkably straightforward, mixing the substrate, the sulfinate salt, and the catalyst in a common solvent like 1,2-dichloroethane under LED irradiation. This shift from thermal/metal-catalyzed processes to photochemical organocatalysis drastically simplifies the workflow and minimizes the environmental footprint of the synthesis.
![Specific reaction scheme for the synthesis of 2-phenyl-3-trifluoromethylimidazo[1,2-a]pyridine using visible light](/insights/img/trifluoromethyl-imidazopyridine-photocatalytic-synthesis-pharma-supplier-20260306101349-04.webp)
Mechanistic Insights into Visible Light Photocatalytic Trifluoromethylation
The core of this innovation lies in the photoredox catalytic cycle initiated by the excitation of the acridinium salt. Upon absorption of visible light photons, the photocatalyst enters an excited state with sufficient redox potential to oxidize the trifluoromethanesulfinate anion. This single-electron transfer (SET) event generates a trifluoromethyl radical and sulfur dioxide, while reducing the photocatalyst. The highly reactive trifluoromethyl radical then adds selectively to the C3 position of the imidazo[1,2-a]pyridine ring, a site known for its nucleophilic character in radical reactions. Subsequent oxidation and deprotonation steps restore aromaticity and regenerate the ground-state photocatalyst, completing the cycle. This mechanism ensures high regioselectivity and avoids the formation of isomeric by-products that often plague electrophilic substitution methods.
From a quality control perspective, the absence of transition metals is a decisive advantage for impurity management. The reaction profile is clean, with the primary by-products being benign sulfur dioxide gas and the spent organic catalyst, both of which are easily separated during workup. The patent data highlights that various substituents on the phenyl ring (B ring) and the fused pyridine ring (A ring) are well-tolerated. Whether the substrate bears electron-donating groups like methoxy or electron-withdrawing halogens, the radical addition proceeds efficiently. This robustness suggests that the electronic properties of the substrate do not significantly inhibit the radical generation or addition steps, making the process highly reliable for generating diverse libraries of analogs for SAR studies.
How to Synthesize 2-Phenyl-3-trifluoromethylimidazo[1,2-a]pyridine Efficiently
The practical implementation of this synthesis is designed for ease of operation in standard laboratory or pilot plant settings. The protocol does not require specialized high-pressure equipment or cryogenic cooling, as the reaction thrives at ambient temperature under simple LED illumination. The standard procedure involves charging the imidazo[1,2-a]pyridine substrate and sodium trifluoromethanesulfinate into a reaction vessel with the photocatalyst and solvent, followed by degassing to establish an inert nitrogen atmosphere. Detailed standardized synthesis steps for replicating this high-yielding transformation are provided in the guide below.
- Combine 2-phenylimidazo[1,2-a]pyridine (0.3 mmol), sodium trifluoromethanesulfinate (0.2 mmol), and an acridinium photocatalyst (0.01 mmol) in 1,2-dichloroethane (2.0 mL) under nitrogen.
- Irradiate the reaction mixture with a blue LED light source (3W) at room temperature for approximately 24 hours to facilitate the radical trifluoromethylation.
- Filter the reaction mixture through celite, concentrate the filtrate under reduced pressure, and purify the crude residue via column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible operational efficiencies and risk mitigation. The elimination of precious metal catalysts removes a major cost driver and supply chain bottleneck, as the prices of metals like palladium and iridium are subject to high volatility and geopolitical constraints. Furthermore, the use of sodium trifluoromethanesulfinate, a commodity chemical, ensures a stable and cost-effective supply of the trifluoromethylating agent. The simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials and shortens the batch cycle time, thereby increasing overall plant throughput.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the substitution of expensive transition metal catalysts with inexpensive organic dyes and the reduction of energy consumption due to room temperature operation. By avoiding the need for high-temperature heating or cryogenic cooling, utility costs are significantly lowered. Additionally, the streamlined purification process reduces solvent usage and waste disposal costs, contributing to a leaner manufacturing cost structure without compromising on the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: Relying on readily available organic photocatalysts and stable sulfinate salts mitigates the risk of raw material shortages that often accompany specialized organometallic reagents. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in operating parameters, leading to more consistent batch-to-batch quality. This reliability is crucial for maintaining continuous supply lines to downstream drug manufacturers, ensuring that production schedules are met without unexpected delays caused by reagent instability or complex handling requirements.
- Scalability and Environmental Compliance: The photochemical nature of the reaction is inherently scalable using modern flow chemistry techniques or large-scale LED reactor arrays, allowing for seamless transition from gram-scale R&D to ton-scale commercial production. From an environmental standpoint, the process aligns with green chemistry principles by minimizing hazardous waste generation and avoiding toxic heavy metals. This facilitates easier regulatory approval and compliance with increasingly strict environmental regulations regarding pharmaceutical manufacturing effluents and residual metal limits in drug substances.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethylation technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction scope, safety, and scalability for potential partners and licensees.
Q: What are the primary advantages of this photocatalytic method over traditional trifluoromethylation?
A: Unlike conventional methods that often require expensive transition metal catalysts, harsh oxidants, or extreme temperatures, this patented process utilizes visible light and organic acridinium salts. This results in milder reaction conditions (room temperature), reduced environmental impact, and easier post-treatment without heavy metal contamination.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method demonstrates excellent scalability potential. It uses commercially available reagents like sodium trifluoromethanesulfinate and common solvents like 1,2-dichloroethane. The reliance on LED light sources rather than high-pressure reactors simplifies the engineering requirements for scaling up to multi-kilogram batches.
Q: What is the substrate scope for this trifluoromethylation reaction?
A: The patent data indicates a broad substrate scope. It successfully accommodates various substituents on the imidazo[1,2-a]pyridine core, including electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups like halogens (chloro, bromo, fluoro) and trifluoromethyl groups, maintaining good to excellent yields across diverse derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Imidazo[1,2-a]pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible-light photocatalysis in the synthesis of complex heterocyclic intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN110590774B can be successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle the specific analytical requirements of fluorinated compounds, guaranteeing that every batch meets the highest standards for pharmaceutical applications.
We invite global pharmaceutical companies and research institutions to collaborate with us on the development and supply of these high-value trifluoromethylated intermediates. By leveraging our expertise in process optimization and scale-up, we can help you reduce time-to-market for your drug candidates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis for your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your supply chain goals.
