Advanced Copper-Catalyzed Synthesis of Quinoline Derivatives for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously demand efficient routes to bioactive heterocycles, with quinoline scaffolds representing a cornerstone of modern drug discovery due to their prevalence in antibacterial agents and cholinesterase inhibitors. A pivotal advancement in this domain is detailed in Chinese Patent CN110204487B, which discloses a novel, environmentally benign synthesis method for quinoline derivatives. This technology leverages readily available 2,1-benzisoxazole derivatives and beta-diketone precursors to construct the quinoline core through a copper-catalyzed cascade reaction. For R&D directors and procurement specialists, this patent represents a strategic shift away from hazardous reagents like sodium azide towards a greener, more economically viable protocol that maintains high yields and operational simplicity. The ability to access diverse 2,3-disubstituted quinolines efficiently addresses critical bottlenecks in the supply of high-purity pharmaceutical intermediates.
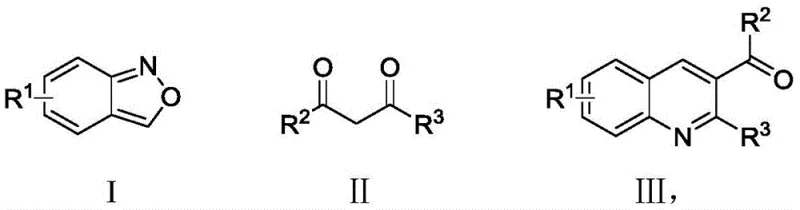
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinoline skeleton has relied on methodologies that pose significant safety and economic challenges for large-scale manufacturing. Traditional routes often utilize o-bromobenzaldehyde coupled with dibenzoyl methane in the presence of sodium azide, a highly toxic and explosive substance that necessitates rigorous safety protocols and specialized waste treatment facilities. Furthermore, alternative pathways employing o-aminobenzaldehyde and omega-benzylacetophenone suffer from the prohibitive cost of starting materials, particularly the latter, which renders the process uneconomical for bulk production. These legacy methods not only inflate the cost of goods sold (COGS) but also introduce substantial regulatory hurdles regarding halogenated waste and heavy metal contamination, complicating the path to commercial approval for new drug candidates.
The Novel Approach
In stark contrast, the methodology described in CN110204487B introduces a transformative approach that circumvents these historical deficiencies by utilizing inexpensive 2,1-benzisoxazoles and beta-diketones as primary building blocks. This innovative route operates under an oxygen atmosphere using a廉价 copper catalyst, effectively eliminating the need for toxic azides or precious metals like palladium. The reaction proceeds with remarkable efficiency, tolerating a broad spectrum of functional groups including halogens and trifluoromethyl moieties, which are essential for tuning the pharmacokinetic properties of final drug products. By shifting to this copper-catalyzed system, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing while simultaneously adhering to stricter environmental, health, and safety (EHS) standards required by global regulatory bodies.
Mechanistic Insights into Copper-Catalyzed Cyclization
The mechanistic elegance of this transformation lies in the synergistic interaction between the copper catalyst and the beta-diketone substrate, which initiates a cascade leading to ring construction. Initially, the divalent copper species coordinates with the beta-diketone to form a stable copper-enolate complex, activating the methylene position for nucleophilic attack. This activated intermediate then engages with the 2,1-benzisoxazole ring, triggering a ring-opening event that is crucial for exposing the reactive nitrogen and oxygen atoms necessary for cyclization. The presence of a strong base, such as cesium carbonate, facilitates the deprotonation steps required to drive the equilibrium forward, ensuring high conversion rates even with sterically hindered substrates.
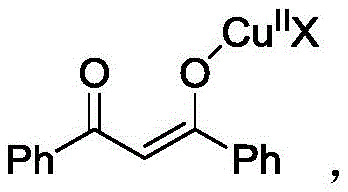
Following the initial coupling, the reaction pathway involves an intramolecular dehydration step that aromatizes the newly formed ring system, yielding the stable quinoline derivative. This mechanism is particularly robust because it avoids the formation of unstable intermediates that often plague other cyclization strategies. The use of hexafluoroisopropanol (HFIP) as a solvent further enhances the reaction kinetics by stabilizing charged intermediates through hydrogen bonding, thereby improving overall yield and selectivity. For process chemists, understanding this mechanism is vital for optimizing reaction parameters such as temperature (110-150°C) and stoichiometry to minimize impurity profiles and maximize the purity of the final API intermediate.
How to Synthesize Quinoline Derivatives Efficiently
Implementing this synthesis protocol requires precise control over reaction conditions to ensure reproducibility and safety on a larger scale. The process begins with the charging of 2,1-benzisoxazole and the selected beta-diketone derivative into a reactor along with the copper bromide catalyst and cesium carbonate base. The system must be purged with oxygen to maintain the oxidative environment required for the catalytic cycle, followed by the addition of the organic solvent and heating to the specified temperature range. Detailed standardized synthetic steps, including specific molar ratios and workup procedures, are outlined below to guide process development teams in adapting this chemistry for pilot plant operations.
- Charge 2,1-benzisoxazole, beta-diketone derivative, copper bromide catalyst, and cesium carbonate base into a Schlenk tube under reduced pressure.
- Replace the atmosphere with oxygen three times and add hexafluoroisopropanol solvent, then stir at 130°C for 24 hours.
- Purify the crude product via silica gel column chromatography using petroleum ether and ethyl acetate to isolate the target quinoline compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this copper-catalyzed methodology offers profound advantages in terms of raw material security and logistical stability. The primary substrates, 2,1-benzisoxazoles and beta-diketones, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency that often plagues projects relying on exotic custom synthons. Furthermore, the elimination of sodium azide removes a major hazardous material from the supply chain, simplifying transportation logistics and reducing insurance premiums associated with handling explosive substances. This shift not only streamlines procurement processes but also enhances the overall resilience of the manufacturing supply chain against regulatory disruptions.
- Cost Reduction in Manufacturing: The economic impact of switching to this method is driven primarily by the replacement of expensive precursors and catalysts with commodity-grade alternatives. Copper bromide is significantly cheaper than palladium or rhodium catalysts often used in cross-coupling reactions, and the avoidance of toxic azides reduces waste disposal costs substantially. Additionally, the high yields reported across various substrates mean less raw material is wasted, directly lowering the variable cost per kilogram of the produced intermediate. These factors combine to create a leaner cost structure that improves margin potential for downstream pharmaceutical products.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the robustness of the reaction conditions, which tolerate a wide range of substituents without requiring bespoke optimization for each new analog. This flexibility allows manufacturers to produce a diverse library of quinoline derivatives using a unified platform, reducing the need for multiple dedicated production lines. The use of stable, non-hygroscopic reagents ensures consistent quality batch-to-batch, minimizing the risk of production delays caused by原料 degradation or variability. Consequently, lead times for high-purity pharmaceutical intermediates can be significantly shortened, enabling faster time-to-market for new drug candidates.
- Scalability and Environmental Compliance: Scaling this process is facilitated by the absence of gas-evolving reagents or highly exothermic steps that typically complicate reactor design. The reaction operates at moderate temperatures and pressures compatible with standard glass-lined steel reactors found in most multipurpose chemical plants. Moreover, the green nature of the process, characterized by atom economy and the use of less toxic reagents, aligns perfectly with modern sustainability goals. This compliance reduces the burden of environmental permitting and wastewater treatment, making it an attractive option for companies aiming to reduce their carbon footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinoline synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for decision-makers evaluating this route for their pipeline. Understanding these nuances is critical for assessing the feasibility of integrating this chemistry into existing manufacturing workflows.
Q: What are the advantages of this copper-catalyzed method over traditional quinoline synthesis?
A: Unlike traditional methods requiring toxic sodium azide or expensive o-bromobenzaldehyde, this patent utilizes cheap 2,1-benzisoxazoles and beta-diketones with an inexpensive copper catalyst, significantly reducing raw material costs and environmental hazards.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method employs robust reaction conditions (110-150°C) and common solvents like HFIP or DMSO, avoiding sensitive reagents, which facilitates straightforward scale-up from laboratory to commercial tonnage production.
Q: What is the substrate scope for this quinoline synthesis?
A: The process demonstrates high tolerance for various substituents including electron-withdrawing groups (F, Cl, Br, CF3) and electron-donating groups (Me, OMe) on both the benzisoxazole and beta-diketone rings, allowing for diverse library generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of robust synthetic routes like the one described in CN110204487B for securing the supply of critical pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of quinoline derivative meets the exacting standards required by global regulatory agencies, providing our partners with the confidence needed to advance their clinical programs.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this advanced copper-catalyzed technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of adopting this route for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next-generation therapeutics are built on a foundation of chemical excellence and supply chain security.
