Advanced Flavonoid Manufacturing: A Non-Phosphorus Vilsmeier-Haack Cyclization Strategy
The pharmaceutical and fine chemical industries continuously seek robust synthetic routes for bioactive scaffolds, particularly flavonoids, which possess significant therapeutic potential ranging from anti-inflammatory to antiviral activities. Patent CN101186600B introduces a transformative preparation method for flavonoid compounds of formula (II), utilizing a modified Vilsmeier-Haack cyclization strategy. Unlike conventional approaches that rely on harsh acidic conditions or phosphorus-based reagents, this innovation employs bis(trichloromethyl) carbonate (BTC) and N,N-dimethylformamide (DMF) to generate the active chlorinating species in situ. This technical breakthrough addresses critical pain points in process chemistry, specifically regarding environmental compliance and operational safety, while maintaining high reaction efficiency. The method operates under remarkably mild thermal conditions, typically between 0°C and 30°C, which significantly reduces energy consumption compared to traditional reflux protocols. For R&D directors and process engineers, this patent represents a viable pathway to scale up flavonoid production with improved impurity profiles and reduced waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
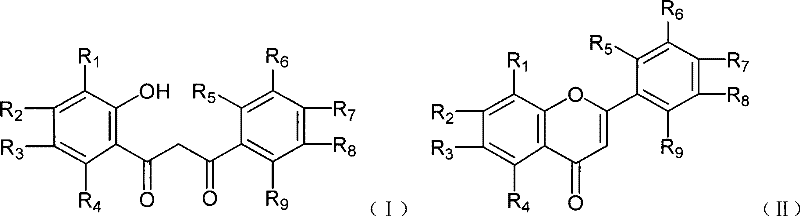
Historically, the cyclization of β-diketone intermediates, such as 1-(2'-hydroxyphenyl)-3-phenyl-1,3-propanedione, into flavonoids has been plagued by severe operational challenges. Traditional methodologies predominantly utilize strong mineral acids like sulfuric acid or acetic acid mixtures, often requiring prolonged heating at reflux temperatures to drive the dehydration and ring-closure steps. These acidic environments are highly corrosive to standard stainless steel reactors, necessitating expensive glass-lined equipment and increasing maintenance costs. Furthermore, the use of phosphorus oxychloride (POCl3) in classical Vilsmeier reactions generates substantial amounts of phosphorus-containing wastewater, creating a heavy burden on effluent treatment facilities and complicating regulatory compliance. The harsh conditions also pose risks to substrate integrity, potentially leading to decomposition or the formation of complex byproduct mixtures that are difficult to purify, thereby lowering the overall isolated yield and purity of the final active pharmaceutical ingredient.
The Novel Approach
The methodology disclosed in CN101186600B fundamentally reengineers the cyclization step by replacing hazardous liquid reagents with solid BTC. By reacting BTC with DMF at low temperatures (0-5°C), a highly reactive Vilsmeier-type chloroiminium salt is generated without the release of toxic phosphorus byproducts. This in situ generation allows for precise control over the reaction kinetics, preventing runaway exotherms common in traditional acid additions. The subsequent addition of the diketone substrate and gradual warming to room temperature (25-30°C) facilitates a smooth cyclization. This approach not only eliminates the need for corrosive strong acids but also drastically simplifies the workup procedure. The reaction mixture can be quenched directly into ice water, allowing for straightforward phase separation and crystallization. This shift from high-energy, high-corrosion processes to a mild, ambient-temperature protocol exemplifies a significant advancement in green chemistry principles applied to flavonoid manufacturing.
Mechanistic Insights into BTC-Mediated Vilsmeier-Haack Cyclization
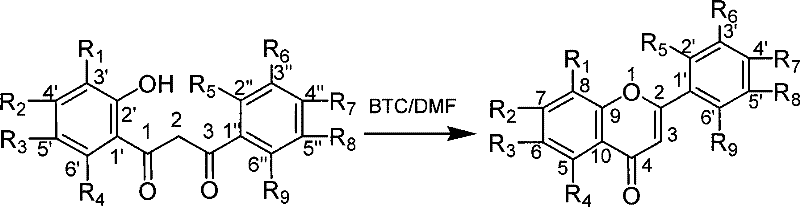
The core of this synthetic innovation lies in the unique activation mechanism facilitated by the BTC-DMF system. Upon mixing bis(trichloromethyl) carbonate with DMF, phosgene equivalents are released which react with the amide to form the electrophilic chloroiminium ion, the active Vilsmeier reagent. This species attacks the enol form of the 1-(2'-hydroxyphenyl)-3-phenyl-1,3-propanedione substrate at the alpha-position relative to the carbonyl group. The proximity of the ortho-hydroxyl group then enables an intramolecular nucleophilic attack on the activated carbonyl carbon, initiating the ring closure. Unlike acid-catalyzed dehydrations that rely on protonation of the hydroxyl group, this mechanism proceeds via a chlorinated intermediate that is a superior leaving group, thereby lowering the activation energy required for cyclization. This mechanistic pathway explains the ability of the reaction to proceed efficiently at near-ambient temperatures, avoiding the thermal stress that often degrades sensitive functional groups on the aromatic rings.
From an impurity control perspective, the mildness of the BTC-DMF system offers distinct advantages over traditional acid reflux methods. Harsh acidic conditions often promote side reactions such as polymerization, over-chlorination, or hydrolysis of sensitive substituents like esters or ethers. By maintaining the reaction temperature below 30°C, the kinetic window for these degradation pathways is significantly narrowed. The patent data indicates consistent yields across a diverse range of substrates, including those with electron-withdrawing nitro groups and electron-donating methoxy groups, suggesting that the mechanism is robust against electronic variations in the substrate. This uniformity is crucial for GMP manufacturing, where batch-to-batch consistency is paramount. The absence of phosphorus residues also simplifies the purification train, reducing the risk of metal or heavy atom contamination in the final API intermediate.
How to Synthesize Flavonoids Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the careful preparation of the Vilsmeier reagent solution, followed by the controlled addition of the diketone precursor. The reaction is allowed to stir for an extended period (5-15 hours) at mild temperatures to ensure complete conversion. Following the reaction, a simple aqueous quench and extraction sequence isolates the crude product, which is then purified via recrystallization. This streamlined workflow minimizes unit operations and solvent usage.
- Preparation of the Vilsmeier Reagent: Dissolve bis(trichloromethyl) carbonate (BTC) in an organic solvent such as 1,2-dichloroethane and add it dropwise to N,N-dimethylformamide (DMF) at 0-5°C.
- Cyclization Reaction: Add the 1-(2'-hydroxyphenyl)-3-phenyl-1,3-propanedione substrate to the reaction mixture at 0-5°C, then allow the temperature to rise to 25-30°C for 5-15 hours.
- Workup and Purification: Quench the reaction in ice water, separate the organic layer, wash with brine, dry over sodium sulfate, and recrystallize to obtain the pure flavonoid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this BTC-mediated synthesis route offers compelling economic and logistical benefits beyond mere chemical yield. The primary advantage lies in the substitution of hazardous, regulated reagents with safer, solid alternatives. Bis(trichloromethyl) carbonate is a stable solid that is easier to transport, store, and handle than liquid phosphorus oxychloride or concentrated sulfuric acid, reducing logistics costs and insurance premiums associated with hazardous material transport. Furthermore, the elimination of strong acids extends the lifespan of reactor vessels and piping, significantly lowering capital expenditure on equipment maintenance and replacement. The mild reaction conditions also translate to reduced utility costs, as there is no need for energy-intensive heating systems or specialized cooling infrastructure beyond standard chillers.
- Cost Reduction in Manufacturing: The transition to this novel method drives cost efficiency through multiple channels. By eliminating the need for expensive glass-lined reactors resistant to strong acid corrosion, manufacturers can utilize standard stainless steel equipment, representing a substantial reduction in capital investment. Additionally, the removal of phosphorus reagents negates the costly downstream processes required for phosphorus waste treatment and disposal. The high selectivity of the reaction minimizes the formation of byproducts, which reduces the volume of solvents and adsorbents needed for purification, further driving down the cost of goods sold (COGS) for high-purity flavonoid intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available commodity chemicals. BTC and DMF are bulk industrial chemicals with stable global supply chains, unlike specialized catalysts or reagents that may face sourcing bottlenecks. The robustness of the reaction across various substituted substrates ensures that production schedules are not disrupted by the need to re-optimize conditions for different flavonoid analogs. This flexibility allows manufacturers to respond rapidly to market demands for diverse flavonoid derivatives without significant process validation delays, ensuring a steady flow of materials to downstream formulation partners.
- Scalability and Environmental Compliance: Scaling this process from kilogram to multi-ton production is facilitated by the inherent safety of the reagents and the exothermic control offered by the low-temperature initiation. The absence of toxic phosphorus waste aligns perfectly with increasingly stringent environmental regulations, mitigating the risk of regulatory fines or shutdowns due to effluent violations. This environmental stewardship enhances the corporate sustainability profile, a key factor for multinational clients who prioritize green supply chains. The simplified workup also reduces the generation of organic waste streams, contributing to a lower overall environmental footprint for the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method. They are derived from the specific experimental data and comparative advantages highlighted in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: Why is Bis(trichloromethyl) Carbonate (BTC) preferred over POCl3 in this synthesis?
A: BTC serves as a solid, safer alternative to liquid POCl3 for generating the Vilsmeier reagent. It eliminates organophosphorus pollution, simplifies handling, and avoids the severe equipment corrosion associated with traditional phosphorus oxychloride methods.
Q: What are the typical reaction conditions for this flavonoid cyclization?
A: The reaction initiates at low temperatures (0-5°C) to control the formation of the reactive intermediate, followed by a mild warming phase to 25-30°C. This contrasts sharply with traditional methods requiring harsh reflux conditions.
Q: Does this method support substrates with diverse substituents?
A: Yes, the patent demonstrates high yields (85-94%) across a wide range of substrates, including those with electron-withdrawing groups like nitro and chloro, as well as electron-donating groups like methoxy and methyl, ensuring broad applicability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flavonoid Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic methodologies like the BTC-mediated cyclization described in CN101186600B. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this mild, non-phosphorus route are fully realized in large-scale manufacturing. Our rigorous QC labs and stringent purity specifications guarantee that every batch of flavonoid intermediate meets the exacting standards required for pharmaceutical applications, providing our partners with a secure and compliant supply source.
We invite global pharmaceutical and agrochemical companies to leverage our technical expertise for their flavonoid requirements. By collaborating with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume needs. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing you to validate the superior quality and economic advantages of our manufacturing capabilities firsthand.
