Advanced Tolvaptan Manufacturing: Technical Upgrades and Commercial Scalability Analysis
The pharmaceutical landscape for vasopressin V2 receptor antagonists has been significantly reshaped by the introduction of Tolvaptan, a critical active pharmaceutical ingredient used in the management of hyponatremia and heart failure. Recent intellectual property developments, specifically patent CN102260213B, have unveiled a transformative preparation method that addresses long-standing inefficiencies in the synthetic pathway. This novel approach utilizes 7-chloro-1,2,3,4,5-tetrahydro-1H-1-benzo-aza-5-one as a foundational starting material, subjecting it to a meticulously engineered four-step sequence involving reduction, silylating agent protection, amino acylation, and final deprotection. For R&D directors and procurement specialists seeking a reliable tolvaptan supplier, understanding the technical nuances of this patent is essential. The methodology not only promises a substantial increase in overall yield but also ensures a purity profile that meets the stringent requirements of global regulatory bodies. By shifting away from precious metal catalysis and complex purification protocols, this technology represents a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing, offering a robust solution for commercial scale-up of complex pharmaceutical intermediates.
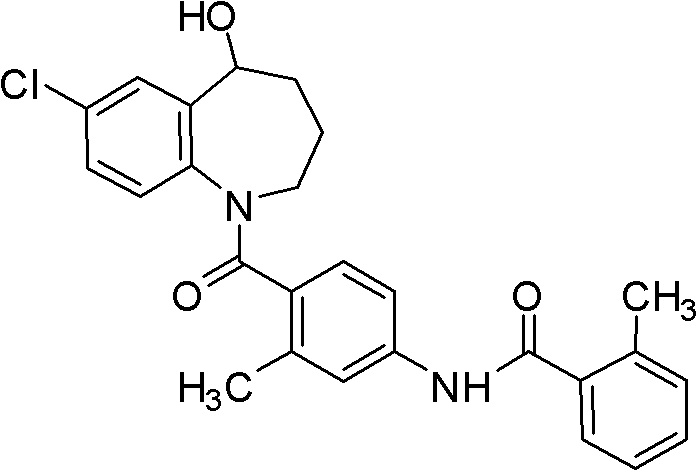
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industrial synthesis of Tolvaptan was plagued by significant technical and economic bottlenecks that hindered efficient production. Historical routes, such as those reported by Kazumi Kondo, relied heavily on the use of platinum dioxide for catalytic reduction, a process that introduces exorbitant costs due to the price of the precious metal and the rigorous safety protocols required for hydrogenation. Furthermore, these legacy methods often necessitated multiple column chromatography purification steps to isolate the target molecule from complex reaction mixtures, a technique that is notoriously difficult to translate from laboratory benchtop to multi-ton manufacturing scales. The reliance on expensive starting materials like 7-chloro-1,2,3,4-tetrahydro benzo azepine-5-ketone in conjunction with low-yielding condensation reactions resulted in overall yields ranging merely from 30% to 50%. Such inefficiencies not only inflate the cost of goods sold but also create supply chain vulnerabilities, making it challenging to ensure the continuous availability of high-purity OLED material or pharmaceutical grades required by downstream partners.
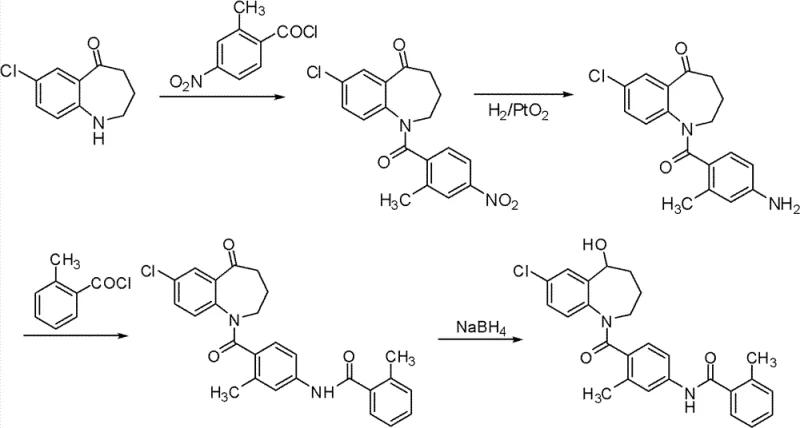
The Novel Approach
In stark contrast to the cumbersome legacy pathways, the novel method disclosed in CN102260213B introduces a streamlined logic that prioritizes atom economy and operational simplicity. By employing a strategic silyl protection group early in the synthesis, the process effectively masks reactive functional groups that would otherwise lead to unwanted by-products during the acylation stage. This clever chemical design allows for the direct use of crude intermediates in subsequent steps without the need for intermediate purification, thereby collapsing the production timeline significantly. The elimination of transition metal catalysts removes the risk of heavy metal contamination, a critical quality attribute for any API intermediate, and simplifies the waste treatment process. Consequently, this approach achieves an impressive overall yield of approximately 78%, representing a dramatic improvement over previous benchmarks. For supply chain heads focused on reducing lead time for high-purity pharmaceutical intermediates, this methodology offers a clear path to more resilient and cost-effective manufacturing operations.
Mechanistic Insights into Silyl-Protection and Mixed Anhydride Coupling
The core innovation of this synthesis lies in the precise manipulation of reactivity through temporary protection strategies. In the second step of the process, the intermediate alcohol derived from the initial reduction is treated with a silylating agent, such as trimethylchlorosilane or hexamethyldisilazane, under controlled alkaline conditions. This reaction converts the hydroxyl group and the secondary amine into their respective silyl ethers and silyl amines. This dual protection is mechanistically crucial because it prevents the nucleophilic attack of the oxygen atom during the subsequent acylation step, ensuring that the acyl group attaches exclusively to the nitrogen atom of the benzazepine ring. Without this protection, the formation of O-acylated impurities would be thermodynamically favorable, complicating the purification process and lowering the final assay. The use of mild bases like triethylamine or sodium methylate facilitates this transformation at low temperatures, typically between -10°C and 0°C, preserving the stereochemical integrity of the molecule while maximizing conversion rates.
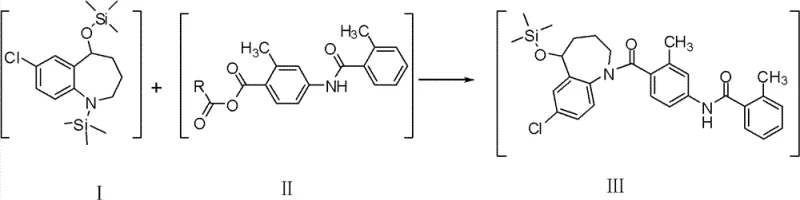
Following the protection phase, the synthesis proceeds through a highly selective coupling reaction utilizing a mixed anhydride activation strategy. The carboxylic acid component, 4-(2-toluylamino)-2-methyl-benzoic acid, is activated in situ using reagents like pivaloyl chloride or trifluoroacetic anhydride to form a reactive mixed anhydride species. This activated electrophile then reacts efficiently with the silyl-protected nucleophile to form the key carbon-nitrogen bond. The choice of activating agent is critical; pivaloyl chloride, for instance, creates a sterically hindered anhydride that minimizes self-condensation of the acid while remaining reactive enough to couple with the amine. The final deprotection step involves acidic hydrolysis, which cleanly removes the silyl groups to reveal the free hydroxyl and amine functionalities of the target Tolvaptan. This sequence ensures that impurity control is built into the chemistry itself, rather than relying solely on downstream purification, resulting in a final product purity exceeding 99.5%.
How to Synthesize Tolvaptan Efficiently
Implementing this synthesis route requires careful attention to reaction conditions and reagent stoichiometry to maximize the benefits of the patented process. The procedure begins with the reduction of the ketone starting material using sodium borohydride in methanol, a safe and scalable reagent combination that avoids the hazards associated with high-pressure hydrogenation. Following the workup, the crude alcohol is immediately subjected to silylation without further purification, leveraging the high purity of the reduction step to drive the protection reaction forward. The subsequent coupling with the mixed anhydride is performed at low temperatures to control exotherms and maintain selectivity. Detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, temperature profiles, and workup procedures necessary to replicate the high yields reported in the patent literature. Adhering to these parameters is essential for achieving the commercial viability and quality consistency expected in modern pharmaceutical manufacturing.
- Reduce 7-chloro-1,2,3,4,5-tetrahydro-1H-1-benzo-aza-5-one using sodium borohydride in methanol to form the corresponding alcohol intermediate.
- Protect the hydroxyl and amine groups of the alcohol intermediate using trimethylchlorosilane or hexamethyldisilazane under alkaline conditions.
- React the protected intermediate with an activated mixed anhydride derived from 4-(2-toluylamino)-2-methyl-benzoic acid to form the coupled product.
- Perform acidic hydrolysis to remove the silyl protecting groups and crystallize the final Tolvaptan product with purity exceeding 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers profound advantages for procurement managers and supply chain directors tasked with optimizing the cost structure of API production. The primary value driver is the significant reduction in raw material and processing costs achieved by eliminating expensive catalysts and simplifying purification workflows. By removing the dependency on platinum-based catalysts, manufacturers avoid the volatility of precious metal markets and the costly recovery processes associated with heavy metal removal. Furthermore, the ability to telescope multiple steps without isolating intermediates reduces solvent consumption, labor hours, and equipment occupancy time, leading to substantial cost savings in pharmaceutical intermediates manufacturing. These efficiencies translate directly into a more competitive pricing structure for the final active ingredient, allowing partners to maintain healthy margins even in a price-sensitive market environment.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the fundamental simplification of the chemical workflow. By avoiding column chromatography and utilizing inexpensive reagents like sodium borohydride and pivaloyl chloride, the variable cost per kilogram of product is drastically lowered. The high yield of 78% means that less starting material is wasted, improving the overall material balance and reducing the environmental footprint associated with waste disposal. Additionally, the absence of heavy metals simplifies the regulatory filing process, as extensive data on metal clearance is not required, further reducing the indirect costs associated with quality control and compliance.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of readily available starting materials and reagents. Unlike routes that depend on specialized or scarce intermediates, this method utilizes commodity chemicals that can be sourced from multiple suppliers globally. This diversification of the supply base mitigates the risk of shortages and price spikes, ensuring a steady flow of materials for production. The robustness of the chemistry also means that the process is less sensitive to minor variations in raw material quality, reducing the likelihood of batch failures and ensuring consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing unit operations that are standard in the fine chemical industry. The avoidance of hazardous reagents and the reduction in solvent usage align with green chemistry principles, making it easier to obtain environmental permits and maintain compliance with increasingly strict regulations. The simplified workup procedures reduce the volume of organic waste generated, lowering disposal costs and enhancing the sustainability profile of the manufacturing site. This alignment with environmental, social, and governance (ESG) goals is increasingly important for multinational corporations seeking responsible partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Tolvaptan synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in patent CN102260213B, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of adopting this route for their specific production needs and ensures alignment with quality and cost objectives.
Q: How does the silyl protection strategy improve Tolvaptan purity?
A: The silyl protection strategy prevents the direct acylation of the hydroxyl group, which is a common side reaction in conventional methods. By masking both the amine and hydroxyl functionalities temporarily, the reaction selectively targets the desired nitrogen acylation, significantly reducing impurity profiles and achieving purity levels above 99.5% without extensive chromatography.
Q: What are the cost advantages of this novel synthesis route?
A: This route eliminates the need for expensive transition metal catalysts like platinum dioxide and avoids complex column chromatography purification steps. The use of readily available reducing agents like sodium borohydride and the ability to use crude intermediates directly in subsequent steps drastically reduces material costs and processing time.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It utilizes standard reagents and mild reaction conditions (mostly between -10°C and 40°C). The high overall yield of approximately 78% compared to prior art methods (30-50%) and the simplified workup procedures make it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tolvaptan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into reliable commercial supply. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this synthesis route are fully realized in practice. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Tolvaptan meets the highest international standards. We understand that consistency is key in the pharmaceutical industry, and our dedicated technical team works closely with clients to optimize process parameters for maximum efficiency and quality.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our expertise can drive value for your organization. Let us be your partner in delivering high-quality Tolvaptan with the reliability and efficiency your business demands.
