Advanced Pd-Catalyzed C-H Alkenylation for Scalable Aryl Vinyl Silane Production
Advanced Pd-Catalyzed C-H Alkenylation for Scalable Aryl Vinyl Silane Production
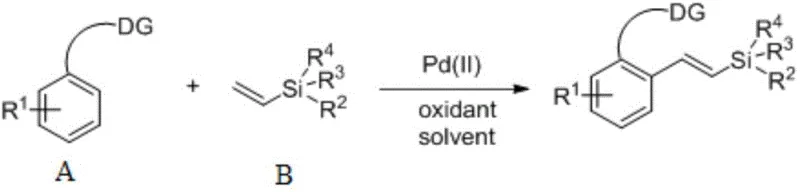
The landscape of organosilicon chemistry is undergoing a significant transformation driven by the demand for more efficient and sustainable synthetic methodologies. Patent CN110698507A introduces a groundbreaking preparation method for aryl vinyl silane compounds that addresses critical bottlenecks in modern organic synthesis. This technology leverages a palladium-catalyzed directed C-H alkenylation strategy, utilizing vinylsilanes as direct alkenylating agents rather than relying on traditional pre-functionalized substrates. By employing commercially available heteroatom-directed arenes and simple vinylsilanes, this process achieves high yields and excellent stereoselectivity under remarkably mild conditions. The elimination of complex ligand systems and the ability to operate without inert gas protection represent a major leap forward in process chemistry, offering a robust platform for the production of high-value intermediates used in pharmaceuticals and advanced materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl vinyl silanes has relied heavily on classical approaches such as hydrosilylation of alkynes or Heck coupling reactions involving halogenated arenes. These conventional pathways suffer from inherent inefficiencies that impact both cost and environmental sustainability. Hydrosilylation often requires expensive hydrosilane reagents and precise control over regioselectivity, while Heck coupling necessitates the prior installation of halogen atoms on the aromatic ring. This pre-functionalization step not only adds synthetic complexity and reduces overall atom economy but also generates stoichiometric amounts of halide waste. Furthermore, many transition-metal catalyzed processes traditionally demand rigorous exclusion of oxygen and moisture, requiring specialized equipment like gloveboxes or Schlenk lines, which complicates scale-up and increases capital expenditure for manufacturing facilities.
The Novel Approach
In stark contrast, the methodology disclosed in CN110698507A circumvents these drawbacks through a direct C-H functionalization strategy. By utilizing a palladium salt as a catalyst precursor in the presence of an oxidant, the system activates the inert C-H bond ortho to a directing group, enabling direct coupling with vinylsilanes. This approach eliminates the need for pre-halogenated substrates, thereby streamlining the synthetic route and improving the overall mass balance of the process. The reaction operates effectively at temperatures ranging from 25°C to 80°C and tolerates a wide variety of functional groups, including alkyl, alkoxy, and halogen substituents. Crucially, the protocol does not require external phosphine ligands or inert atmosphere protection, making it exceptionally practical for industrial application. This simplicity translates directly into reduced operational complexity and enhanced safety profiles for large-scale production environments.
Mechanistic Insights into Pd-Catalyzed Directed C-H Alkenylation
The core of this innovation lies in the synergistic interaction between the palladium catalyst and the heteroatom directing group (DG) present on the arene substrate. The mechanism initiates with the coordination of the palladium species to the directing group, which facilitates the selective cleavage of the ortho C-H bond through a concerted metalation-deprotonation (CMD) or similar pathway. This step forms a stable cyclometallated palladacycle intermediate, ensuring high regioselectivity even in the presence of other reactive sites on the aromatic ring. Subsequently, the vinylsilane coordinates to the palladium center and undergoes migratory insertion into the Pd-C bond. The presence of the oxidant, such as silver acetate or benzoquinone, is critical for regenerating the active Pd(II) species from the reduced Pd(0) state formed after reductive elimination, thus closing the catalytic cycle. This oxidative turnover allows the reaction to proceed with catalytic amounts of palladium, minimizing metal residue in the final product.
From an impurity control perspective, the ligand-free nature of this catalytic system offers distinct advantages. Traditional ligand-based systems often suffer from ligand degradation or dissociation, leading to the formation of homocoupling byproducts or unreactive palladium black. By relying on the substrate's own directing group to stabilize the metal center, this method minimizes side reactions and ensures a cleaner reaction profile. The high stereoselectivity observed, typically favoring the E-isomer, is attributed to the steric constraints imposed during the migratory insertion and beta-hydride elimination steps within the rigid palladacycle framework. This level of control is paramount for pharmaceutical applications where isomeric purity can dictate the efficacy and safety of the final drug substance. The robustness of the catalytic cycle against air and moisture further reduces the risk of oxidation-related impurities that often plague sensitive organometallic transformations.
How to Synthesize Aryl Vinyl Silane Efficiently
The implementation of this synthesis route is designed for operational ease, allowing chemists to produce high-purity aryl vinyl silanes with minimal setup. The process involves simply combining the directed arene, vinylsilane, palladium catalyst, and oxidant in a common organic solvent, followed by heating and stirring. This straightforward protocol removes the barriers associated with handling air-sensitive reagents or synthesizing complex ligands. For detailed procedural specifics regarding stoichiometry, solvent selection, and workup procedures, please refer to the standardized synthesis guide below which outlines the exact experimental parameters derived from the patent examples.
- Combine the heteroatom-directed arene substrate, vinylsilane reagent, palladium salt catalyst precursor, and oxidant in a suitable organic reaction medium.
- Heat the reaction mixture to a temperature between 25°C and 80°C and maintain stirring for a duration of 1 to 24 hours to ensure complete conversion.
- Upon completion, separate the crude product via flash column chromatography to remove impurities, followed by concentration under reduced pressure to isolate the pure aryl vinyl silane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of expensive phosphine ligands and the removal of inert gas requirements fundamentally alter the cost structure of the manufacturing process. By simplifying the reactor setup to standard atmospheric conditions, facilities can utilize existing infrastructure without needing costly upgrades for anaerobic processing. This flexibility enhances supply chain resilience by allowing production to be distributed across a wider range of contract manufacturing organizations (CMOs) that may not possess specialized glovebox capabilities. Furthermore, the use of commodity chemicals like vinylsilanes and simple palladium salts ensures a stable and predictable raw material supply, mitigating risks associated with sourcing niche or proprietary reagents.
- Cost Reduction in Manufacturing: The most significant financial advantage stems from the drastic simplification of the catalyst system. By operating without exogenous ligands, the process avoids the high costs associated with purchasing and disposing of specialized phosphines or N-heterocyclic carbenes. Additionally, the ability to run reactions under air eliminates the continuous consumption of nitrogen or argon gases, which represents a tangible utility saving over long production campaigns. The high atom economy of the direct C-H activation route means less raw material is wasted on leaving groups like halides, further driving down the cost per kilogram of the active pharmaceutical ingredient (API) intermediate. These cumulative efficiencies result in substantial cost savings that improve the overall margin profile of the final product.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on widely available starting materials. The substrates, such as substituted anilines and benzoic acids, are commodity chemicals produced at multi-ton scales globally, ensuring that procurement teams are not held hostage by single-source suppliers. The robustness of the reaction conditions also means that batch-to-batch variability is minimized, reducing the likelihood of failed runs that could disrupt delivery schedules. Since the process does not require stringent moisture control, storage and handling of reagents are less critical, reducing the risk of spoilage during transit or warehousing. This reliability is crucial for maintaining just-in-time inventory levels and meeting tight production deadlines for downstream customers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is inherently safer and more straightforward due to the mild reaction temperatures and absence of pyrophoric reagents. The simplified workup, often involving basic filtration and chromatography, reduces the volume of solvent waste generated compared to multi-step functionalization routes. From an environmental compliance standpoint, the higher atom efficiency aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product). The avoidance of halogenated waste streams from pre-functionalization steps simplifies wastewater treatment and disposal, lowering the environmental burden and associated regulatory compliance costs for the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Pd-catalyzed C-H alkenylation technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the method's versatility and operational parameters. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing process pipelines.
Q: Why is this ligand-free Pd-catalyzed method superior to traditional Heck coupling?
A: Traditional Heck coupling requires pre-functionalized halide substrates, which increases step count and waste. This patented method utilizes direct C-H activation on readily available arenes, significantly improving atom economy and reducing raw material costs by eliminating the need for halogenation steps.
Q: Does this process require expensive inert gas protection?
A: No, one of the distinct advantages of this technology is its operational simplicity. The reaction proceeds efficiently without the need for inert gas protection (such as nitrogen or argon), allowing for standard atmospheric processing which drastically simplifies equipment requirements and lowers operational overhead.
Q: What is the stereoselectivity of the resulting vinyl silanes?
A: The method demonstrates excellent stereoselectivity, predominantly yielding the E-isomer of the aryl vinyl silane compounds. This high selectivity is crucial for downstream applications in pharmaceutical synthesis where geometric purity impacts biological activity and subsequent coupling reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Vinyl Silane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this Pd-catalyzed alkenylation are seamlessly translated into robust manufacturing processes. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of aryl vinyl silane meets the exacting standards required for pharmaceutical applications. We combine deep technical expertise with a commitment to quality, positioning us as the ideal partner for bringing complex organosilicon intermediates to market.
We invite you to collaborate with our technical team to explore how this efficient synthesis route can optimize your supply chain and reduce overall project costs. Please contact our Customized Cost-Saving Analysis department to discuss your specific requirements. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments tailored to your project's unique needs. Reach out to our technical procurement team today to initiate a dialogue on securing a reliable supply of high-quality aryl vinyl silanes for your next breakthrough.
