Advanced Pd-Catalyzed C-H Alkenylation for Scalable Aryl Vinyl Silane Production
Advanced Pd-Catalyzed C-H Alkenylation for Scalable Aryl Vinyl Silane Production
The chemical industry is constantly seeking more efficient pathways to construct complex organosilicon frameworks, which serve as critical building blocks in modern medicinal chemistry and material science. Patent CN110698507A, published in early 2020, introduces a transformative preparation method for aryl vinyl silane compounds that addresses long-standing challenges in C-H bond functionalization. This technology leverages a palladium-catalyzed oxidative coupling strategy that directly links aromatic hydrocarbons containing heteroatom directing groups with vinylsilanes. Unlike conventional multi-step sequences, this approach operates under remarkably mild conditions, typically between 25°C and 80°C, and completes within a timeframe of 1 to 24 hours. The significance of this innovation lies in its ability to bypass the need for pre-functionalized substrates, thereby enhancing the overall atom economy of the synthesis. For R&D teams focused on rapid library generation and process chemists aiming for robust manufacturing protocols, this patent offers a compelling solution that balances high stereoselectivity with operational simplicity.
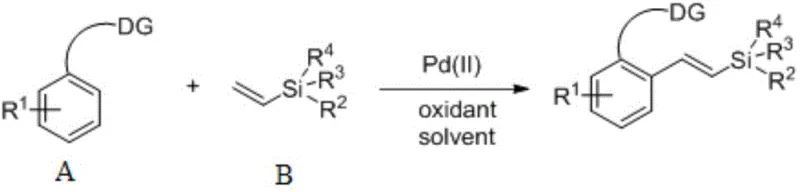
Traditionally, the synthesis of vinylsilane derivatives has relied heavily on classical methodologies such as hydrosilylation of alkynes or Heck coupling reactions involving halogenated aromatics. While these methods are well-established, they suffer from inherent limitations regarding step economy and environmental impact. The requirement for pre-halogenated starting materials introduces additional synthetic steps, each generating stoichiometric amounts of salt waste and consuming valuable reagents. Furthermore, Heck couplings often necessitate the use of expensive phosphine ligands and strict inert atmosphere conditions to prevent catalyst deactivation. In contrast, the novel approach detailed in the patent utilizes a direct C-H alkenylation strategy. By employing vinylsilane directly as the alkenylating agent, the process eliminates the preliminary preparation of functionalized substrates. This shift not only reduces the cumulative cost of raw materials but also simplifies the workflow by removing the need for rigorous exclusion of air and moisture, which is a significant logistical advantage for scale-up operations in standard chemical plants.
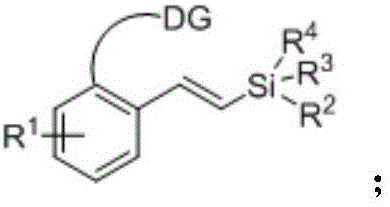
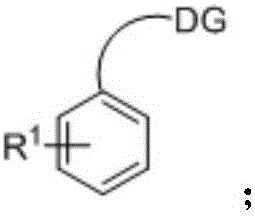
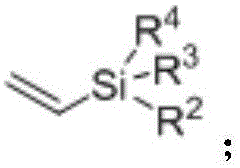
The mechanistic underpinning of this transformation involves a sophisticated yet efficient palladium catalytic cycle driven by coordination-assisted C-H activation. The process initiates with the coordination of the palladium(II) species to the heteroatom directing group (DG) present on the aromatic substrate, such as an amino or carboxyl group. This coordination brings the metal center into close proximity with the ortho-C-H bond, facilitating a concerted metalation-deprotonation (CMD) or electrophilic palladation event that cleaves the carbon-hydrogen bond to form a stable cyclometallated intermediate. Subsequently, the vinylsilane molecule coordinates to the palladium center and undergoes migratory insertion into the Pd-C bond. The unique electronic properties of the vinylsilane, often considered electron-rich and less reactive than activated alkenes, are effectively managed by the specific choice of oxidant and solvent system. The cycle concludes with a beta-hydride elimination step that releases the desired aryl vinyl silane product and generates a palladium-hydride species, which is then re-oxidized by the silver salt oxidant to regenerate the active Pd(II) catalyst. This redox-neutral manifold ensures that the catalyst remains active throughout the reaction duration without the accumulation of inactive palladium black.
Impurity control is a critical aspect of this methodology, particularly given the potential for homocoupling or over-alkenylation side reactions. The patent specifies that the use of specific oxidants like silver acetate or silver carbonate, in conjunction with solvents such as 1,2-dichloroethane or hexafluoroisopropanol, plays a pivotal role in suppressing these undesired pathways. The directing group not only guides the regioselectivity to the ortho-position but also stabilizes the intermediate complexes, preventing non-selective background reactions. Furthermore, the reaction conditions are tuned to favor the formation of the trans-alkene isomer, as evidenced by the large coupling constants observed in the NMR data of the examples. The workup procedure described, involving flash column chromatography followed by concentration under reduced pressure, is highly effective at removing residual palladium species and silver salts, ensuring that the final product meets the stringent purity requirements necessary for downstream applications in pharmaceutical synthesis.
How to Synthesize Aryl Vinyl Silane Efficiently
The practical implementation of this synthesis route is designed to be accessible for both laboratory-scale optimization and industrial production. The protocol outlines a straightforward mixing procedure where the aromatic substrate and vinylsilane are combined with the catalytic system in a common organic solvent. The flexibility in choosing palladium precursors, ranging from palladium acetate to various palladium chloride complexes, allows manufacturers to optimize costs based on local availability. Similarly, the oxidant loading can be adjusted between 100% and 300% relative to the substrate to maximize conversion without excessive waste. The reaction temperature window of 25°C to 80°C provides ample room for thermal management, allowing exotherms to be controlled easily in large reactors. Detailed standardized synthesis steps see the guide below.
- Combine the heteroatom-directed aromatic compound (Substrate A) and vinylsilane (Substrate B) in a reaction vessel with a suitable organic solvent such as 1,2-dichloroethane.
- Add the palladium catalytic precursor (e.g., Pd(OAc)2 or PdCl2(MeCN)2) and the oxidant (e.g., silver acetate) to the mixture at room temperature.
- Heat the reaction mixture to 25-80°C and stir for 1-24 hours, then purify the crude product via flash column chromatography to obtain the target aryl vinyl silane.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this C-H activation technology offers substantial strategic benefits that extend beyond mere chemical efficiency. The elimination of pre-functionalized halide substrates represents a significant reduction in the bill of materials, as halogenated aromatics are often more expensive and subject to greater supply volatility than their parent hydrocarbons. Additionally, the removal of expensive ligands from the catalyst system drastically lowers the cost per kilogram of the final product. Since the reaction does not require inert gas protection, facilities can utilize standard glass-lined or stainless steel reactors without the need for specialized nitrogen blanketing systems, thereby reducing capital expenditure and operational overhead. The simplified workup procedure further contributes to cost savings by minimizing solvent consumption and reducing the time required for purification, leading to faster batch turnover rates.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the atom-economic nature of the direct C-H functionalization. By avoiding the synthesis and purification of halogenated intermediates, manufacturers save on reagents, solvents, and labor associated with those extra steps. Furthermore, the use of commodity-grade palladium salts instead of proprietary ligand-catalyst complexes ensures that the catalytic system remains affordable even at large scales. The ability to run the reaction without strict inert atmosphere conditions also reduces utility costs related to nitrogen generation and monitoring, contributing to a leaner manufacturing profile.
- Enhanced Supply Chain Reliability: The reliance on commercially available and structurally simple starting materials enhances the resilience of the supply chain. Aromatic amines and carboxylic acids with directing groups are widely produced commodities, reducing the risk of bottlenecks associated with custom-synthesized intermediates. Vinylsilanes are also readily accessible from major silicon chemical suppliers. This abundance of raw materials ensures consistent production schedules and mitigates the risk of delays caused by the scarcity of specialized reagents. The robustness of the reaction conditions means that production is less susceptible to minor variations in environmental factors, ensuring reliable delivery timelines for customers.
- Scalability and Environmental Compliance: Scaling this process is facilitated by its operational simplicity and the absence of hazardous reagents typically associated with organometallic chemistry, such as pyrophoric bases or sensitive organolithiums. The waste stream is primarily composed of silver salts and organic solvents, which can be managed through established recovery and recycling protocols. The high selectivity of the reaction minimizes the formation of difficult-to-separate byproducts, reducing the load on wastewater treatment facilities. This alignment with green chemistry principles not only simplifies regulatory compliance but also positions the manufacturer favorably in markets increasingly driven by sustainability metrics and environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and reliability for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production lines or research pipelines.
Q: What are the key advantages of this Pd-catalyzed method over traditional Heck coupling?
A: Unlike traditional Heck coupling which requires pre-functionalized halide substrates, this method utilizes direct C-H activation. This eliminates the need for halogenation steps, significantly improving atom economy and reducing waste generation while operating under ligand-free conditions.
Q: Does this process require inert gas protection or specialized equipment?
A: No, one of the distinct operational benefits of this patented protocol is that it proceeds efficiently without the need for inert gas protection (such as nitrogen or argon atmospheres), simplifying the reactor setup and reducing operational complexity for large-scale manufacturing.
Q: What types of directing groups are compatible with this synthesis?
A: The method demonstrates broad substrate tolerance, effectively accommodating various heteroatom directing groups including amino, methylamino, dimethylamino, carboxyl, ethylenediamino, and propylenediamino groups, allowing for diverse functionalization of the aromatic core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Vinyl Silane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating the development of next-generation pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN110698507A can be seamlessly translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of aryl vinyl silane meets the exacting standards required by global drug developers.
We invite you to collaborate with us to leverage this efficient synthesis route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this ligand-free methodology can optimize your budget. Please contact us to request specific COA data for our available inventory or to discuss route feasibility assessments for custom derivatives, ensuring a secure and high-quality supply chain for your critical organosilicon intermediates.
