Advanced Synthesis of Avibactam Intermediates Using Sulfonic Acid Quaternary Ammonium Salts for Commercial Scale
Advanced Synthesis of Avibactam Intermediates Using Sulfonic Acid Quaternary Ammonium Salts for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic pathways for beta-lactamase inhibitors, particularly for next-generation antibiotics like avibactam. A pivotal development in this domain is disclosed in patent CN112679498A, which introduces a novel sulfonic acid quaternary ammonium salt compound and its preparation method. This technology addresses critical bottlenecks in the production of avibactam sodium by establishing a highly efficient intermediate stage that ensures exceptional purity levels exceeding 99.89%. For R&D directors and process chemists, this represents a significant leap forward in impurity control, while supply chain leaders will recognize the potential for streamlined manufacturing protocols that rely on readily available reagents and mild reaction conditions.
The core innovation lies in the strategic use of a quaternary ammonium salt intermediate, specifically defined by Formula (I) in the patent documentation. Unlike traditional methods that may struggle with the isolation of the free acid or unstable sodium salts directly from crude reaction mixtures, this approach leverages the crystalline nature of the quaternary ammonium species. This allows for rigorous purification via crystallization before the final salt exchange to the active pharmaceutical ingredient (API) form. By integrating this intermediate into the workflow, manufacturers can achieve a final avibactam sodium purity of greater than 99.90%, meeting the stringent quality standards required for raw material medicines in global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of avibactam and related diazabicyclo octane derivatives has faced challenges related to yield optimization and impurity profiles during the sulfonation and salt formation stages. Conventional routes often involve direct sulfonation followed by immediate neutralization, which can lead to difficult-to-remove byproducts and variable yields depending on the batch scale. Previous patents, such as CN 103649051A and CN 105283458B, have outlined various synthetic strategies, yet the industry still demands methods that offer superior reproducibility and easier downstream processing. The lack of a stable, crystallizable intermediate in some older protocols necessitates complex chromatographic purifications or multiple recrystallizations, driving up both the cost of goods sold (COGS) and the environmental footprint of the manufacturing process.
The Novel Approach
The methodology presented in CN112679498A circumvents these issues by introducing a discrete quaternary ammonium salt formation step. As illustrated in the structural definitions below, the compound features a specific cationic head group paired with the sulfonated diazabicyclo anion. This ionic pairing enhances the solid-state properties of the intermediate, facilitating high-yield crystallization from solvent systems like ethanol and ethyl acetate. 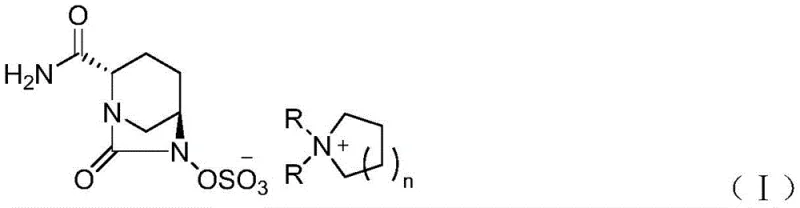 This structural modification acts as a powerful purification tool, effectively locking the molecule into a stable crystal lattice that excludes organic impurities. Consequently, the subsequent conversion to avibactam sodium becomes a straightforward salt exchange reaction, significantly reducing the operational complexity and enhancing the overall reliability of the supply chain for this critical antibiotic intermediate.
This structural modification acts as a powerful purification tool, effectively locking the molecule into a stable crystal lattice that excludes organic impurities. Consequently, the subsequent conversion to avibactam sodium becomes a straightforward salt exchange reaction, significantly reducing the operational complexity and enhancing the overall reliability of the supply chain for this critical antibiotic intermediate.
Mechanistic Insights into Hydrogenolysis and Sulfonation Cascade
The synthetic route begins with a tandem deprotection and sulfonation sequence that is both elegant and efficient. The starting material, (2S,5R)-6-(benzyloxy)-7-oxo-1,6-diazabicyclo[3.2.1]octane-2-carboxamide, undergoes catalytic hydrogenolysis using palladium on carbon (Pd/C) to remove the benzyl protecting group. Simultaneously or sequentially, the exposed hydroxyl group reacts with a sulfur trioxide trimethylamine complex in the presence of a base like triethylamine. This one-pot strategy minimizes the exposure of the sensitive beta-lactam ring to harsh conditions. The reaction is conducted in a mixed solvent system of alcohol and water, preferably isopropanol and water, at mild temperatures ranging from 20-35°C. This thermal window is crucial for maintaining stereochemical integrity and preventing ring-opening degradation pathways that are common in beta-lactam chemistry.
Following the formation of the sulfonic acid anion, the process employs a stoichiometric reaction with a quaternary ammonium bromide, such as 1,1-dipentylcyclohexylmethylammonium bromide. The mechanism here relies on the strong electrostatic attraction between the bulky organic cation and the sulfonate anion, driving the precipitation or crystallization of the product. 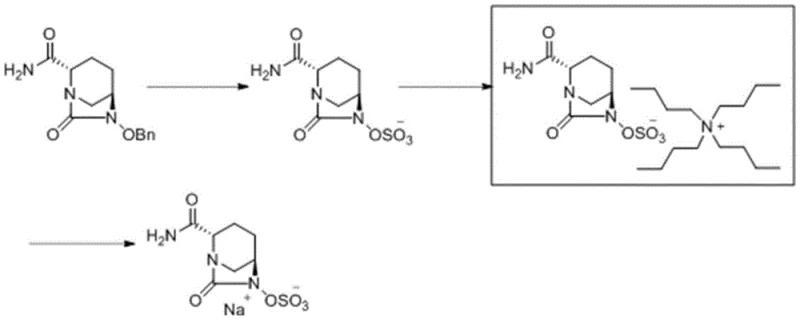 This step is pivotal because the solubility profile of the quaternary salt differs markedly from the starting materials and side products, allowing for effective separation via extraction and crystallization. The final transformation involves dissolving this purified intermediate in ethanol and treating it with sodium isooctanoate, which displaces the quaternary ammonium cation with a sodium ion to yield the final avibactam sodium API with high purity.
This step is pivotal because the solubility profile of the quaternary salt differs markedly from the starting materials and side products, allowing for effective separation via extraction and crystallization. The final transformation involves dissolving this purified intermediate in ethanol and treating it with sodium isooctanoate, which displaces the quaternary ammonium cation with a sodium ion to yield the final avibactam sodium API with high purity.
How to Synthesize Sulfonic Acid Quaternary Ammonium Salt Efficiently
To implement this synthesis in a pilot or commercial plant, precise control over reagent addition and crystallization parameters is essential. The process starts by dissolving the benzyl-protected precursor in a mixture of isopropanol and water, followed by the addition of the Pd/C catalyst and the sulfonating agent. Hydrogen gas is introduced to drive the deprotection, after which the quaternary ammonium bromide is added in portions to manage the exotherm and optimize crystal growth. The detailed standardized operating procedures for temperature ramps, stirring rates, and filtration protocols are critical for ensuring batch-to-batch consistency. For a comprehensive breakdown of the specific experimental steps and reagent quantities described in the patent examples, please refer to the technical guide below.
- Dissolve the benzyl-protected precursor in alcohol-water, add Pd/C catalyst and sulfur trioxide trimethylamine complex, then introduce hydrogen for deprotection and sulfonation at 20-35°C.
- Filter off the catalyst, wash the filtrate, and react with a specific quaternary ammonium bromide (e.g., 1,1-dipentylcyclohexylmethylammonium bromide) to form the intermediate salt.
- Extract the product, concentrate, crystallize using ethanol-ethyl acetate mixtures, and finally convert to avibactam sodium using sodium isooctanoate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible benefits regarding cost structure and operational resilience. The shift towards a crystallizable quaternary ammonium intermediate fundamentally changes the economics of production by reducing the reliance on expensive preparative chromatography or extensive solvent swaps. By simplifying the purification train, the process inherently lowers the consumption of utilities and solvents, which translates directly into reduced manufacturing costs. Furthermore, the use of commodity chemicals such as palladium on carbon, sulfur trioxide complexes, and common quaternary ammonium salts ensures that the supply chain remains robust against raw material shortages, providing a stable foundation for long-term production planning.
- Cost Reduction in Manufacturing: The elimination of complex purification steps significantly lowers the operational expenditure associated with producing high-purity avibactam intermediates. By leveraging the crystallization of the quaternary salt, manufacturers can avoid the high costs of specialized resin columns or multiple distillation cycles. This streamlined approach not only reduces the direct cost of materials but also decreases the labor hours required for process monitoring and quality control testing, resulting in substantial overall cost savings for the final API production.
- Enhanced Supply Chain Reliability: The reagents utilized in this protocol, including isopropanol, ethyl acetate, and standard quaternary ammonium bromides, are widely available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency, which is a critical consideration for maintaining continuous API manufacturing. Additionally, the mild reaction conditions reduce the wear and tear on reactor vessels and auxiliary equipment, leading to lower maintenance costs and higher equipment availability rates over the lifecycle of the production facility.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvent systems and temperatures that are easily managed in large-scale stainless steel reactors. The ability to perform the reaction in aqueous-alcoholic mixtures reduces the volume of volatile organic compounds (VOCs) emitted compared to processes relying solely on chlorinated or aromatic solvents. This alignment with green chemistry principles facilitates easier regulatory compliance and waste treatment, positioning the manufacturer as a responsible partner in the sustainable production of essential pharmaceutical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. Understanding the nuances of the crystallization behavior and the specific role of the quaternary ammonium counter-ion is vital for process engineers aiming to transfer this technology from the laboratory to the production floor. These insights are derived directly from the experimental data and claims within the patent literature, providing a reliable basis for feasibility assessments and technology transfer discussions.
Q: What is the primary advantage of using the sulfonic acid quaternary ammonium salt intermediate?
A: The primary advantage is the ability to achieve exceptionally high purity (>99.89%) through crystallization of the quaternary ammonium salt, which acts as an effective purification handle before the final conversion to the sodium salt.
Q: What are the critical reaction conditions for the hydrogenolysis step?
A: The reaction requires mild temperatures between 20-35°C and uses a palladium on carbon catalyst (10% Pd) in a mixed solvent system of isopropanol and water to ensure complete deprotection without degrading the sensitive beta-lactam core.
Q: How does this method improve scalability compared to traditional routes?
A: By utilizing common reagents like sulfur trioxide trimethylamine complex and avoiding cryogenic conditions or exotic catalysts, the process simplifies equipment requirements and reduces safety risks associated with large-scale sulfonation reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Avibactam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the global fight against antibiotic resistance. Our team of expert process chemists has extensively evaluated the route described in CN112679498A and confirmed its viability for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of avibactam intermediate we deliver meets the highest international pharmaceutical standards.
We invite you to collaborate with us to optimize your supply chain for beta-lactamase inhibitors. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can accelerate the availability of life-saving antibiotics by ensuring a reliable, cost-effective, and high-quality supply of critical pharmaceutical intermediates.
