Advanced Manufacturing of Avibactam Sodium via Micro-Channel Hydrogenation for Global Pharma Supply Chains
Advanced Manufacturing of Avibactam Sodium via Micro-Channel Hydrogenation for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical beta-lactamase inhibitors like Avibactam Sodium, a vital component in combating multi-drug resistant bacterial infections. Patent CN116023379A introduces a groundbreaking preparation method that addresses longstanding challenges in the industrial production of this high-value API intermediate. By leveraging a novel one-pot synthesis strategy combined with continuous micro-channel hydrogenation technology, this innovation delivers a pathway characterized by exceptional safety, high purity exceeding 99.9%, and operational simplicity. This technical advancement is particularly significant for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier status while adhering to stringent GMP standards. The process eliminates the need for highly toxic reagents such as phosgene derivatives, marking a substantial shift towards greener and more sustainable chemical manufacturing practices.
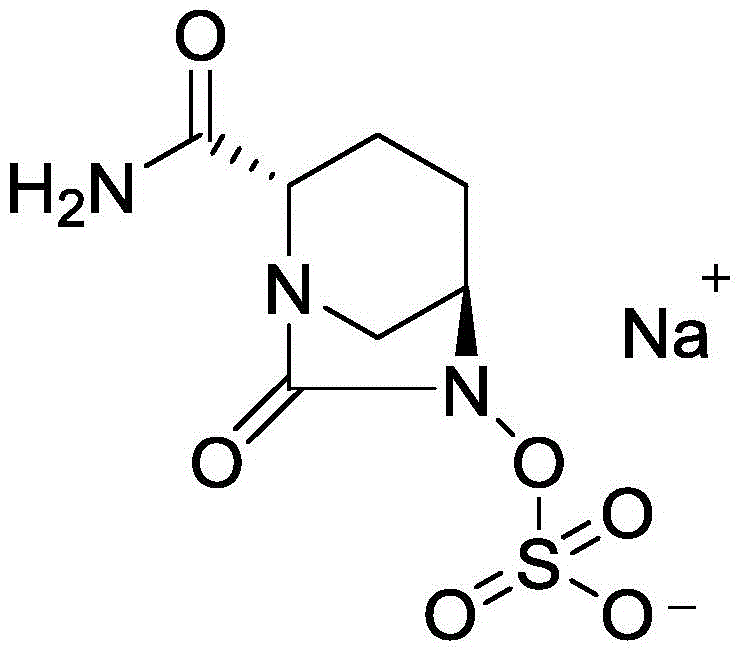
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Avibactam Sodium has been plagued by complex multi-step sequences that rely on hazardous reagents and difficult purification protocols. Prior art, such as the routes disclosed in CN1468242A and CN103328476A, often necessitates the use of diphosgene or triphosgene to construct the critical dicyclic urea structure. These reagents are not only extremely toxic but also require specialized containment infrastructure, significantly driving up capital expenditure and operational risk. Furthermore, conventional strategies frequently employ Fmoc protection groups which require basic conditions for removal, potentially compromising the stability of the sensitive beta-lactam-like core. The hydrolysis processes in these legacy routes are often cumbersome, involving complex workups and recrystallization steps that erode overall yield and extend production lead times. Such inefficiencies create bottlenecks in the supply chain, making it difficult to achieve the consistent quality and volume required for global commercial distribution.
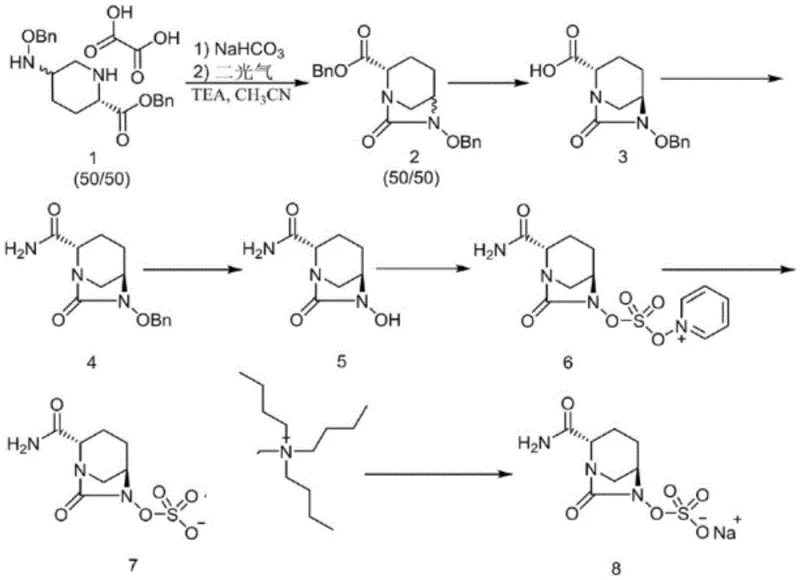
The Novel Approach
In stark contrast, the methodology outlined in CN116023379A revolutionizes the production landscape by implementing a streamlined, low-toxicity workflow. The new route initiates with a highly efficient condensation and ammonolysis reaction using di-tert-butyl dicarbonate, bypassing the need for dangerous carbonylating agents entirely. A key differentiator is the adoption of a micro-channel hydrogenation reactor for the reduction step, which transforms a traditionally hazardous batch operation into a safe, continuous flow process. This approach not only mitigates the risks associated with hydrogen gas handling but also enhances mass and heat transfer, leading to superior reaction control. By integrating these advanced processing techniques with a simplified salt formation step using sodium isooctanoate, the novel method ensures cost reduction in pharmaceutical intermediates manufacturing through reduced waste generation and higher throughput. The result is a commercially viable process that is inherently safer and more adaptable to large-scale automation.
Mechanistic Insights into Micro-Channel Hydrogenation and Cyclization
The core of this technological breakthrough lies in the precise engineering of the reaction mechanism, particularly during the construction of the diazabicyclo [3.2.1] octane skeleton. The synthesis begins with the strategic protection of the amine functionality using a Boc group, which serves a dual purpose: it prevents unwanted side reactions during the subsequent amidation and facilitates easy removal under mild acidic conditions. Following the formation of the amide intermediate, N,N'-carbonyldiimidazole (CDI) is employed to induce cyclization. This reagent is selected for its ability to activate the carboxylic acid without generating the aggressive byproducts associated with acid chlorides or anhydrides. The mechanistic pathway ensures high regioselectivity, effectively suppressing the formation of dimerization impurities that often plague urea-forming reactions. This selectivity is crucial for maintaining the integrity of the chiral centers at the 2S and 5R positions, which are essential for the biological activity of the final drug substance.
Furthermore, the implementation of micro-channel technology for the hydrogenation step represents a paradigm shift in process safety and efficiency. In this stage, the benzyl protecting group is removed, and the molecule is subsequently sulfated. Traditional batch hydrogenation poses significant risks due to the accumulation of hydrogen gas and the exothermic nature of the reaction. The micro-channel reactor confines the reaction mixture to a very small volume, typically in the microliter range, which drastically reduces the potential energy inventory. This physical constraint allows for rapid heat dissipation and precise temperature maintenance between 0°C and 50°C, preventing thermal runaways. The continuous flow nature also ensures uniform exposure to the palladium catalyst (Pd/C and Pd(OH)2), leading to consistent conversion rates and minimizing the formation of over-reduced byproducts. This level of control is instrumental in achieving the reported purity of >99.9%.
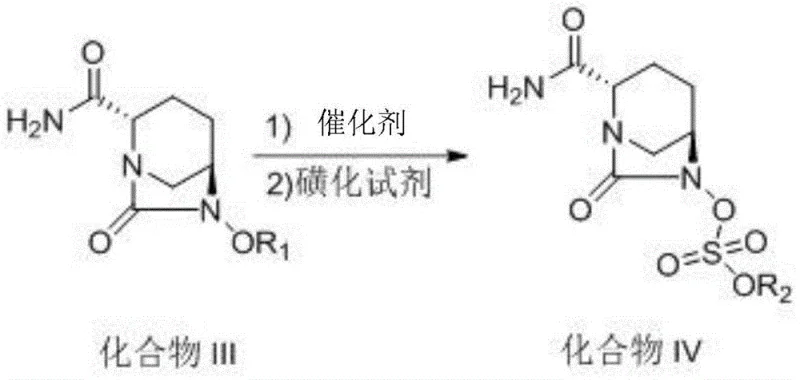
How to Synthesize Avibactam Sodium Efficiently
The synthesis of Avibactam Sodium via this patented route involves a logical sequence of four primary transformations designed for maximum efficiency and safety. The process begins with the protection and amidation of the starting piperidine acid, followed by a cyclization step to close the bicyclic ring system. The critical third step utilizes continuous flow hydrogenation to remove the benzyl group and install the sulfate moiety, followed by a final ion exchange to generate the sodium salt. This workflow is specifically engineered to minimize isolation steps, allowing intermediates to proceed directly to the next stage in solution, thereby reducing solvent consumption and processing time. For detailed operational parameters, including specific solvent ratios, catalyst loadings, and temperature profiles, please refer to the standardized synthesis guide below.
- React (2S, 5R)-5-[(benzyloxy)amino]piperidine-2-carboxylic acid with di-tert-butyl dicarbonate and an ammonia source to form the protected amide intermediate.
- Perform cyclization using N,N'-carbonyldiimidazole (CDI) followed by acid-mediated Boc removal to construct the diazabicyclo octane core.
- Execute continuous hydrogenation in a micro-channel reactor using Pd/C and Pd(OH)2, followed by sulfation to introduce the sulfate group.
- Conduct ion exchange with a sodium source (e.g., sodium isooctanoate) to finalize the Avibactam Sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling strategic advantages that extend beyond simple chemistry. The elimination of highly toxic reagents like triphosgene removes the need for expensive scrubbing systems and specialized hazard containment facilities, leading to substantial cost savings in facility operations and regulatory compliance. Moreover, the use of readily available starting materials, such as (2S, 5R)-5-[(benzyloxy)amino]piperidine-2-carboxylic acid, ensures a stable and resilient supply base that is less susceptible to market volatility. The simplified workup procedures, which avoid complex hydrolysis and extensive recrystallization, translate directly into shorter cycle times and increased plant capacity. These factors collectively contribute to a more robust supply chain capable of meeting the rigorous demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by replacing expensive and hazardous reagents with safer, more economical alternatives like CDI and Boc anhydride. The one-pot nature of the initial steps reduces solvent usage and labor costs associated with multiple isolation and purification stages. Additionally, the high yield (>76.0%) and exceptional purity (>99.9%) minimize material loss and the need for reprocessing, directly lowering the cost of goods sold (COGS). The avoidance of transition metal scavengers, often required to meet strict residual metal limits in APIs, further streamlines the downstream processing budget.
- Enhanced Supply Chain Reliability: By utilizing a continuous micro-channel hydrogenation process, the manufacturing timeline is drastically compressed compared to traditional batch methods. This technology enables around-the-clock production with minimal downtime for cleaning and setup, ensuring a consistent output of high-quality intermediates. The reduced reliance on specialized, hard-to-source reagents mitigates the risk of supply disruptions. Furthermore, the inherent safety of the micro-reactor system lowers insurance premiums and regulatory hurdles, facilitating smoother logistics and faster time-to-market for finished drug products.
- Scalability and Environmental Compliance: The modular nature of the micro-channel reactor system allows for seamless scale-up from pilot to commercial production without the typical engineering challenges associated with increasing batch sizes. This "numbering up" approach maintains the same reaction conditions regardless of volume, ensuring product consistency. From an environmental perspective, the process generates significantly less hazardous waste due to the absence of phosgene derivatives and the efficient use of solvents. This aligns perfectly with modern green chemistry principles and helps manufacturers meet increasingly strict environmental, social, and governance (ESG) targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Avibactam Sodium using this advanced methodology. These insights are derived directly from the experimental data and beneficial effects described in patent CN116023379A, providing clarity on how this route compares to existing technologies. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: How does the micro-channel hydrogenation process improve safety compared to traditional batch methods?
A: The micro-channel reactor significantly reduces the liquid volume held during hydrogenation, minimizing the potential energy release in case of an incident. It allows for precise temperature control (0-50°C) and continuous operation, eliminating the amplification effects seen in large batch kettles and essentially changing the safety profile of the reaction.
Q: What are the purity levels achievable with this new synthesis route?
A: The patented process achieves exceptional purity levels, with the final Avibactam Sodium product demonstrating a molar yield of over 76.0% and a purity exceeding 99.9%. This high quality is attributed to the avoidance of toxic phosgene reagents and the efficient one-pot synthesis strategy which minimizes impurity generation.
Q: Why is the Boc protection strategy preferred over Fmoc in this synthesis?
A: The Boc (tert-butoxycarbonyl) group is removed under acidic conditions, which is more beneficial for the stability of the reaction product compared to the basic conditions required for Fmoc removal. Additionally, Boc is easier to remove and simplifies subsequent treatment steps, avoiding the need for recrystallization before the next reaction stage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Avibactam Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the micro-channel hydrogenation technology described in CN116023379A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering Avibactam Sodium with stringent purity specifications, utilizing our rigorous QC labs to ensure every batch meets the highest international standards. Our state-of-the-art facilities are equipped to handle continuous flow chemistry, allowing us to leverage the safety and efficiency benefits of this novel process for our clients.
We invite you to collaborate with us to optimize your supply chain for this vital beta-lactamase inhibitor. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term production goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
