Advanced One-Pot Synthesis of Spiroisoxazolines for Commercial Pharmaceutical Intermediates
Advanced One-Pot Synthesis of Spiroisoxazolines for Commercial Pharmaceutical Intermediates
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A significant breakthrough in this domain is detailed in patent CN111253331B, which discloses a novel method for synthesizing spiroisoxazoline derivatives using dihydrochalcone as a direct raw material. This technology represents a paradigm shift from traditional multi-step processes, offering a streamlined one-pot cascade reaction that achieves oximino grouping and alpha-position dearomatization simultaneously. For R&D directors and procurement specialists alike, this innovation promises not only higher yields and superior purity profiles exceeding 95 percent but also a drastic reduction in operational complexity. By eliminating the need for pre-derivation into phenol oxime compounds and avoiding the use of excessive oxidants, this method addresses critical pain points in the supply chain of bioactive scaffolds used in antibacterial and antitumor drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spiroisoxazolines, which serve as vital scaffolds for marine natural products like suberanollilines and purealidins, has been fraught with inefficiencies. The conventional wisdom dictated a reliance on pre-prepared phenol oxime compounds as the starting materials. This approach suffers from inherent structural defects in the process design: firstly, the synthesis of the phenol oxime precursor itself is a lengthy endeavor involving multiple steps with often disappointing yields, creating a bottleneck before the main cyclization even begins. Secondly, the subsequent cyclization typically requires an excessive amount of oxidant to drive the reaction, which is not only economically wasteful but also generates a substantial volume of chemical waste, complicating downstream purification and environmental compliance. Finally, even after enduring these cumbersome steps, the final yield of the spiroisoxazoline product remains suboptimal, rendering the overall process unattractive for large-scale commercial manufacturing where margin compression is a constant threat.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111253331B introduces a creative and highly efficient solution by utilizing dihydrochalcone compounds directly as the feedstock. This innovative route bypasses the tedious pre-derivation stage entirely, enabling a direct conversion from ketone to ketoxime and subsequently to the spiroisoxazoline core within a single reaction vessel. The general structure of the versatile dihydrochalcone raw material is depicted below, showcasing the broad substrate tolerance regarding R groups which can range from phenyl and substituted phenyls to various heterocycles.
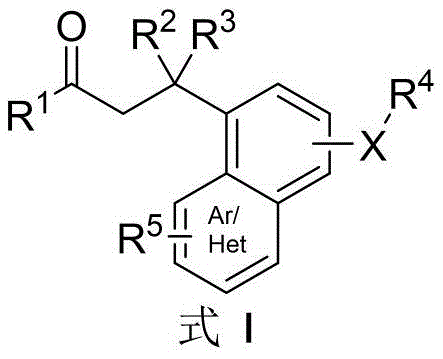
Furthermore, the resulting spiroisoxazoline products, whether forming the ortho-substituted Formula II-A or para-substituted Formula II-B depending on the starting material's substitution pattern, are obtained with remarkable efficiency. This direct transformation is facilitated by a cascade reaction mechanism that merges oximation and dearomatization, fundamentally altering the economic equation of producing these high-value intermediates. The ability to access such complex molecular architectures from simple, commercially available ketones positions this technology as a game-changer for cost reduction in pharmaceutical intermediate manufacturing.
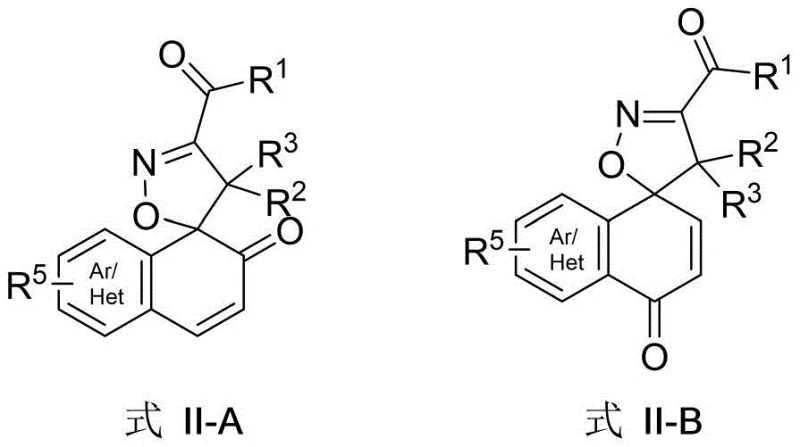
Mechanistic Insights into FeCl3-Compatible Cascade Cyclization
The core of this technological advancement lies in its elegant reaction mechanism, which leverages the in situ generation of reactive species to drive the cascade. The process utilizes sodium nitrite and concentrated hydrochloric acid to generate nitrous acid within the reaction medium, which then reacts with the dihydrochalcone substrate. This initiates the oximino grouping at the ketone position, followed immediately by an intramolecular cyclization that triggers the dearomatization of the adjacent aromatic ring. This tandem sequence is highly atom-economical and avoids the need for external, harsh oxidizing agents that often lead to over-oxidation or decomposition of sensitive functional groups. The reaction proceeds smoothly at room temperature, indicating a favorable activation energy profile that supports mild operating conditions, thereby preserving the integrity of diverse functional groups present on the aromatic rings.
From an impurity control perspective, the simplicity of the reagent system—essentially salt, acid, and solvent—minimizes the introduction of metal contaminants or complex organic byproducts that are difficult to remove. The patent data highlights that the reaction can be performed without inert gas protection, suggesting a robust tolerance to atmospheric conditions that further simplifies the engineering controls required. As demonstrated in specific embodiments, such as the conversion of substrate 1a to product 1b, the reaction cleanly affords the spirocyclic core with high regioselectivity. The structural fidelity of the product is confirmed through rigorous spectroscopic analysis, ensuring that the dearomatization occurs precisely at the intended alpha-position to form the stable isoxazoline ring system fused to the cyclohexadienone moiety.
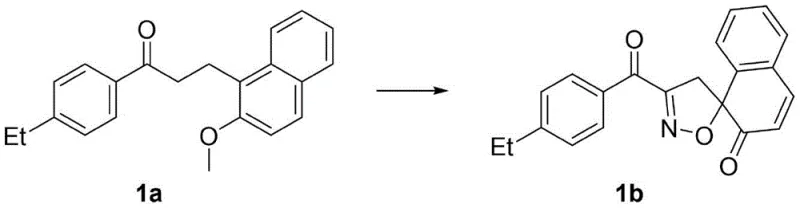
How to Synthesize Spiroisoxazoline Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is straightforward due to the lack of stringent atmospheric requirements and the use of common solvents. The general experimental procedure dictates charging the reactor with the dihydrochalcone starting material, sodium nitrite, concentrated hydrochloric acid, and tetrahydrofuran. The mixture is then stirred at room temperature for a period ranging from 0.5 to 48 hours, depending on the specific substrate reactivity. Following the reaction, the workup involves simple concentration of the reaction liquid followed by standard column chromatography purification. This operational simplicity makes it an ideal candidate for rapid scale-up and process optimization.
- Add dihydrochalcone, sodium nitrite (3 equivalents), concentrated hydrochloric acid (5 equivalents), and tetrahydrofuran solvent into a reaction flask without the need for inert gas protection.
- Stir the reaction mixture vigorously at room temperature (400-1200 rpm) for a duration ranging from 0.5 to 48 hours to allow the cascade oximino grouping and dearomatization to proceed.
- Upon completion, concentrate the reaction liquid under reduced pressure and perform column chromatography separation using petroleum ether and ethyl acetate to isolate the high-purity target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers profound strategic benefits that extend beyond mere chemical yield. The elimination of the pre-synthesis step for phenol oxime precursors translates directly into a shortened production cycle, allowing for faster turnaround times on customer orders. Moreover, the removal of expensive and hazardous oxidants from the bill of materials significantly lowers the raw material costs and reduces the regulatory burden associated with handling and disposing of strong oxidizing agents. The ability to run the reaction without inert gas protection further reduces utility costs and simplifies the reactor setup, making it feasible to utilize existing infrastructure without major capital expenditure on specialized blanketing systems.
- Cost Reduction in Manufacturing: The most significant financial impact comes from the drastic simplification of the synthetic route. By collapsing what was traditionally a multi-step sequence into a single pot, labor costs, energy consumption, and solvent usage are substantially reduced. The replacement of costly, specialized oxidants with commodity chemicals like sodium nitrite and hydrochloric acid ensures that the variable cost per kilogram of the active intermediate is minimized. Additionally, the high purity of the crude product (>95 percent) implies that less resource-intensive purification steps are required, further driving down the cost of goods sold (COGS) and enhancing overall profit margins for the final API or intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and stable starting materials. Dihydrochalcones are accessible from numerous global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions, which tolerate ambient atmosphere and room temperature, means that production is less susceptible to disruptions caused by equipment failure or utility fluctuations. This reliability ensures consistent delivery schedules, a critical factor for pharmaceutical clients who operate on tight timelines and require uninterrupted supply of key building blocks for their drug development pipelines.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is markedly superior. The avoidance of excessive oxidants reduces the generation of hazardous waste streams, simplifying wastewater treatment and lowering disposal costs. The mild reaction conditions reduce the thermal load on the facility, enhancing operational safety. Furthermore, the high atom economy of the cascade reaction aligns with green chemistry principles, making it easier to meet increasingly stringent environmental regulations. This sustainability profile not only mitigates regulatory risk but also enhances the brand value of the manufacturer as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. They are derived from the specific experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical implementation and advantages of this novel route for potential partners and licensees.
Q: What is the primary advantage of this synthesis method over conventional routes?
A: The primary advantage is the elimination of the pre-derivation step. Conventional methods require synthesizing a phenol oxime compound first, which involves long steps and low yields. This novel method converts dihydrochalcone directly to spiroisoxazoline in one pot, significantly simplifying the workflow and improving overall yield.
Q: Does this process require expensive catalysts or inert atmosphere?
A: No, the process is designed for operational simplicity and cost efficiency. It utilizes cheap and readily available reagents like sodium nitrite and concentrated hydrochloric acid. Furthermore, the reaction proceeds effectively at room temperature without the need for inert gas protection, reducing equipment and gas costs.
Q: What purity levels can be achieved with this synthetic route?
A: The method consistently delivers high-purity products. The patent data indicates that the resulting spiroisoxazolines achieve a purity of more than 95 percent, with specific examples demonstrating purity levels as high as 98 percent by HPLC analysis, making it suitable for demanding pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiroisoxazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis method described in patent CN111253331B for the production of high-value spiroisoxazoline intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of this cascade reaction, maintaining stringent purity specifications through our rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our spiroisoxazoline portfolio and to discuss route feasibility assessments for your custom synthesis needs, ensuring a partnership built on transparency, quality, and mutual success.
