Advanced One-Pot Synthesis of Spiroisoxazolines for Commercial Pharmaceutical Intermediates
Introduction to Patent CN111253331A
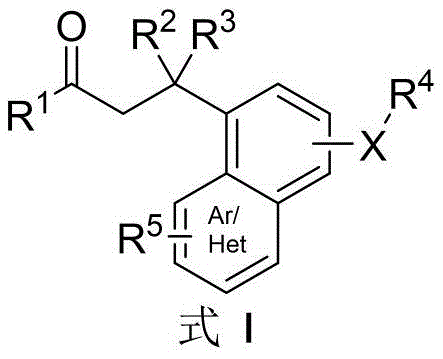
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex heterocyclic scaffolds, particularly those with proven biological activity. Patent CN111253331A introduces a groundbreaking methodology for the synthesis of spiroisoxazolines, a privileged structure found in numerous natural products such as subereamollines and purealidins, which exhibit potent antibacterial, antiviral, and antitumor properties. This innovation represents a significant leap forward in process chemistry by enabling the direct transformation of readily available dihydrochalcone derivatives into valuable spiroisoxazoline cores. Unlike traditional multi-step sequences, this novel approach achieves the construction of the spirocyclic framework through a tandem reaction sequence in a single vessel, drastically reducing processing time and material handling. For R&D directors and procurement specialists, this technology offers a compelling value proposition by merging high chemical efficiency with operational simplicity, positioning it as a critical asset for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spiroisoxazoline scaffolds has been plagued by inefficiencies inherent in multi-step linear syntheses. The conventional state-of-the-art typically necessitates the pre-preparation of phenol oxime compounds as distinct intermediates before the final cyclization can occur. This prerequisite adds substantial burden to the manufacturing process, involving additional reaction vessels, isolation steps, and purification protocols that cumulatively erode the overall yield. Furthermore, these traditional routes often rely on the use of excessive amounts of oxidants to drive the cyclization, which not only increases the raw material costs but also generates significant quantities of chemical waste, posing challenges for environmental compliance and waste treatment. The cumulative effect of these factors is a process that is economically burdensome and operationally complex, making it less attractive for large-scale commercial production where margin compression is a constant concern for supply chain managers.
The Novel Approach
In stark contrast, the methodology disclosed in CN111253331A circumvents these bottlenecks by utilizing dihydrochalcones as the direct starting materials. This innovative route eliminates the need for isolating unstable phenol oxime intermediates, thereby streamlining the entire synthetic sequence into a concise one-pot operation. The reaction employs inexpensive and commercially abundant reagents, specifically sodium nitrite and concentrated hydrochloric acid, in a tetrahydrofuran solvent system. By operating under mild conditions, typically at room temperature and without the stringent requirement for inert gas protection, the process significantly lowers the barrier to entry for manufacturing. As illustrated in the specific transformation below, the conversion is highly efficient, with reported yields reaching up to 97% in optimized examples, demonstrating a robust and scalable pathway for generating these complex molecular architectures.
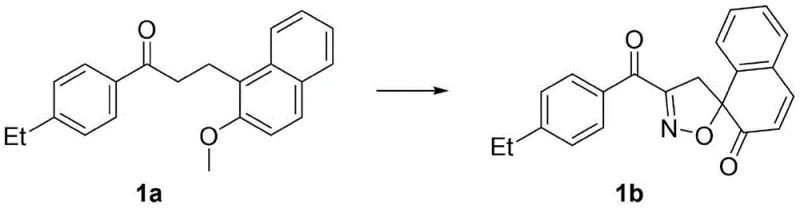
Mechanistic Insights into Tandem Oximation-Dearomatization
The core chemical innovation driving this process is a sophisticated tandem reaction mechanism that seamlessly integrates ketone alpha-oximation with subsequent dearomatization and cyclization. Mechanistically, the reaction initiates with the in situ generation of nitrous acid from sodium nitrite and hydrochloric acid, which then reacts with the alpha-position of the ketone moiety in the dihydrochalcone substrate to form a transient oxime intermediate. Crucially, rather than isolating this intermediate, the reaction conditions promote an immediate intramolecular cyclization involving the aromatic ring. This dearomatization step is key to forming the spirocyclic center, locking the molecule into the rigid spiroisoxazoline geometry. This cascade nature of the reaction ensures that the reactive oxime species is consumed as soon as it is formed, minimizing side reactions and decomposition pathways that often plague stepwise syntheses. For process chemists, understanding this mechanism highlights the elegance of the design, where thermodynamic driving forces are harnessed to push the equilibrium towards the desired spirocyclic product with high fidelity.
From an impurity control perspective, this one-pot strategy offers distinct advantages over sequential methods. In traditional routes, the isolation of the phenol oxime intermediate often leads to the accumulation of isomeric impurities or degradation products that are difficult to remove in later stages. By keeping the reaction mixture homogeneous and driving the transformation to completion in a single phase, the formation of these persistent impurities is suppressed. The patent data supports this, indicating that the crude products obtained after simple concentration often possess purity levels exceeding 95%, as confirmed by HPLC analysis. This high level of intrinsic purity reduces the load on downstream purification units, such as column chromatography or recrystallization, further enhancing the overall process mass intensity (PMI). For quality assurance teams, this translates to a more consistent impurity profile and a lower risk of batch failure due to out-of-specification contaminants.
How to Synthesize Spiroisoxazoline Efficiently
Implementing this synthesis protocol in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The general procedure involves charging a reaction vessel with the dihydrochalcone substrate, followed by the addition of sodium nitrite and concentrated hydrochloric acid in tetrahydrofuran. The mixture is then stirred vigorously at ambient temperatures for a duration ranging from 0.5 to 48 hours, depending on the specific electronic nature of the substrate substituents. Following the reaction period, the workup is straightforward, involving the concentration of the reaction liquor under reduced pressure. The resulting residue is then subjected to standard purification techniques, typically silica gel column chromatography using petroleum ether and ethyl acetate mixtures, to afford the target spiroisoxazoline. Detailed standardized synthesis steps see the guide below.
- Charge a reaction vessel with dihydrochalcone substrate, sodium nitrite (3 equivalents), concentrated hydrochloric acid (5 equivalents), and tetrahydrofuran solvent.
- Stir the reaction mixture at room temperature (0 to 75°C range) for 0.5 to 48 hours without the need for inert gas protection.
- Upon completion, concentrate the reaction solution under reduced pressure and purify the crude residue via column chromatography to isolate the high-purity spiroisoxazoline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary value driver is the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures. By collapsing multiple synthetic steps into a single operation, the process eliminates the need for intermediate storage, handling, and quality testing, which are often hidden cost centers in complex API intermediate manufacturing. Furthermore, the reliance on commodity chemicals like sodium nitrite and hydrochloric acid, rather than specialized or proprietary reagents, insulates the supply chain from volatility in raw material pricing and availability. This stability is crucial for maintaining long-term supply continuity for downstream pharmaceutical clients who demand reliability above all else.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the elimination of the pre-derivatization step required in conventional methods. By avoiding the synthesis and isolation of phenol oxime precursors, manufacturers save significantly on labor, solvent consumption, and energy usage associated with running separate reaction batches. Additionally, the absence of expensive transition metal catalysts or exotic oxidants means that the raw material bill of materials (BOM) is kept exceptionally low. The high yields reported, often approaching quantitative levels, ensure that raw material utilization is maximized, minimizing the cost per kilogram of the final active intermediate. This efficiency allows for competitive pricing strategies in the global market for pharmaceutical building blocks.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. Dihydrochalcones are accessible from numerous global suppliers, reducing the risk of single-source dependency. Moreover, the robustness of the reaction conditions—specifically the ability to run the process at room temperature without inert gas protection—means that the manufacturing can be performed in standard glass-lined or stainless steel reactors without the need for specialized nitrogen blanketing systems. This flexibility allows for faster turnaround times and easier scheduling within multi-purpose production facilities, ensuring that delivery commitments to customers can be met consistently even during periods of high demand.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage scale is facilitated by its mild thermal profile and simple exotherm management. The lack of hazardous oxidants and the use of common solvents simplify the waste treatment protocols, aligning with increasingly stringent environmental regulations. The reduction in chemical waste generation not only lowers disposal costs but also enhances the sustainability profile of the manufacturing site. For companies aiming to reduce their carbon footprint, this atom-economical approach represents a greener alternative to traditional oxidative cyclizations, potentially qualifying for green chemistry incentives and improving the corporate social responsibility (CSR) metrics of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this spiroisoxazoline synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing portfolios. The answers reflect the practical realities of scaling this chemistry while maintaining the high standards required for pharmaceutical grade materials.
Q: What are the primary advantages of this new synthesis method over traditional routes?
A: The primary advantage is the elimination of the pre-derivatization step. Conventional methods require synthesizing phenol oxime intermediates first, which is lengthy and low-yielding. This new method converts dihydrochalcones directly to spiroisoxazolines in one pot, significantly simplifying the workflow and improving overall yield.
Q: Does this process require expensive catalysts or inert atmosphere conditions?
A: No, the process is designed for operational simplicity and cost efficiency. It utilizes inexpensive reagents like sodium nitrite and hydrochloric acid. Furthermore, the reaction proceeds effectively at room temperature without the need for inert gas protection, reducing equipment complexity and operational costs.
Q: What level of purity can be expected from the final spiroisoxazoline products?
A: The patent data indicates that the method consistently produces products with purity exceeding 95%. In specific examples provided, HPLC analysis confirmed purity levels as high as 98%, making the output suitable for demanding pharmaceutical applications without extensive recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiroisoxazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this one-pot synthesis technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented methodology from the laboratory bench to full-scale commercial production. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot trials to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify that every batch of spiroisoxazoline intermediate adheres to the highest quality standards, with purity levels consistently maintained above 95% as per the process capabilities.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cost-effective and robust synthetic route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact our technical procurement team today to request specific COA data for our spiroisoxazoline portfolio and to discuss route feasibility assessments for your custom synthesis projects. Together, we can accelerate the development of next-generation therapeutics by securing a reliable and economical supply of these critical chemical building blocks.
