Industrial Scale Synthesis of Dydrogesterone via Advanced Photochemical Isomerization
Introduction to Next-Generation Dydrogesterone Manufacturing
The global pharmaceutical landscape is witnessing a pivotal shift in the production of critical hormonal APIs, driven by the urgent need for sustainable and scalable synthetic routes. Patent CN110818760A introduces a groundbreaking production technology for the industrialized synthesis of Dydrogesterone (CAS 152-62-5), a vital progestogen widely utilized in gynecological therapies for conditions such as endometriosis and threatened abortion. This innovative methodology transcends traditional limitations by employing a sophisticated sequence of carbonyl protection, selective bromination, and a dual-stage LED-mediated photochemical isomerization, ultimately delivering a process that is not only environmentally benign but also economically superior for large-scale operations. By leveraging readily available Progesterone as the starting feedstock, this route effectively bypasses the supply chain bottlenecks associated with exotic precursors, positioning it as a strategic asset for reliable pharmaceutical intermediates supplier networks seeking to diversify their API portfolios.
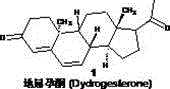
Furthermore, the technical robustness of this synthesis lies in its meticulous control over stereochemistry, particularly during the critical inversion of the C9 and C10 centers, which is achieved through a novel photochemical ring-opening and closing mechanism that minimizes side reactions. The integration of modern LED technology replaces hazardous and energy-intensive mercury lamps, aligning the manufacturing process with stringent global environmental compliance standards while enhancing operator safety. For R&D directors and process chemists, this patent represents a significant leap forward in steroid chemistry, offering a clear pathway to high-purity high-purity API intermediates that meet the rigorous quality specifications demanded by international regulatory bodies. The following analysis dissects the mechanistic advantages and commercial viability of this route, providing actionable insights for stakeholders involved in the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Dydrogesterone has been plagued by significant technical and economic hurdles that have hindered widespread generic production and kept market prices artificially high. Early methodologies, such as those disclosed in British Patent GB929271, relied on photosta-4,7,22-trien-3-one as a starting material, a compound that is notoriously difficult to source and requires complex multi-step preparation, rendering the overall process economically unviable for industrial application. As illustrated in the historical reaction schemes, these legacy routes often suffered from cumulative low yields across multiple steps, necessitating extensive purification protocols that generated substantial chemical waste and increased the carbon footprint of the manufacturing process.
Moreover, alternative approaches like US3198792 utilized trans-progesterone, a non-natural isomer that does not exist in abundant natural products and requires difficult synthetic access, thereby creating a fragile supply chain vulnerable to raw material shortages. The reliance on high-pressure mercury lamps in older photochemical steps further exacerbated operational costs due to high energy consumption and the generation of ozone and heat, which complicated temperature control and often led to the formation of unwanted by-products. These cumulative inefficiencies resulted in a market dominated by a single manufacturer, highlighting the critical need for a disruptive technology that could democratize access to this essential medicine through a more robust and accessible synthetic strategy.
The Novel Approach
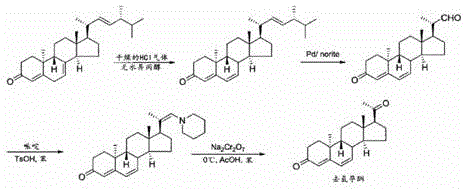
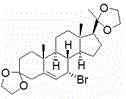
In stark contrast to these archaic methods, the process defined in CN110818760A establishes a new paradigm by utilizing Progesterone, a commodity steroid hormone, as the foundational building block, thereby ensuring immediate access to high-quality starting materials at competitive market rates. The synthetic pathway is elegantly designed to proceed through a stable bis-ketal protected intermediate, which safeguards the sensitive carbonyl functionalities during the subsequent radical bromination and debromination steps, ensuring high fidelity in the construction of the 4,6-diene system. This strategic protection-deprotection sequence allows for harsh reaction conditions to be employed where necessary without compromising the integrity of the steroid backbone, resulting in significantly improved overall yields compared to prior art.
The cornerstone of this novel approach is the implementation of a controlled, dual-wavelength LED photochemical system that facilitates the stereospecific inversion of the 9α,10β-isomer to the therapeutically active 9β,10α-configuration with unprecedented precision. By replacing broad-spectrum mercury irradiation with narrow-band LED sources, the process achieves superior selectivity, minimizing the formation of tachysterol and lumisterol by-products that typically plague photochemical steroid transformations. This technological upgrade not only enhances the purity profile of the crude product, reducing the burden on downstream purification, but also drastically lowers energy consumption, making the cost reduction in pharmaceutical intermediates manufacturing a tangible reality rather than a theoretical projection.
Mechanistic Insights into LED-Mediated Photochemical Isomerization
The heart of this innovative synthesis lies in the sophisticated photochemical transformation of the 9α,10β-diene intermediate (Compound 5) into the 9β,10α-isomer (Compound 6), a step that dictates the stereochemical outcome of the entire sequence. The mechanism involves a sequential photo-excitation process where the substrate is first irradiated with light in the 265-300 nanometer range to induce a conrotatory electrocyclic ring-opening reaction, generating a transient triene intermediate. This highly reactive species is then immediately subjected to a second irradiation source in the 305-365 nanometer range, which triggers a disrotatory electrocyclic ring-closing reaction, effectively inverting the stereochemistry at the C9 and C10 positions to yield the desired 9β,10α-configuration. The precision of this dual-wavelength approach is critical, as it allows for the selective population of excited states that favor the desired geometric isomer while suppressing competing pathways.
From an impurity control perspective, the patent specifies the use of a continuous flow or semi-continuous reactor system equipped with precise temperature regulation, maintaining the reaction mixture between -35°C and 35°C to prevent thermal degradation of the photosensitive intermediates. The integration of inert gas protection (Nitrogen or Argon) throughout the photochemical stage is paramount to prevent oxidative degradation of the diene system, which could lead to the formation of peroxides and other oxygenated impurities that are difficult to remove. Furthermore, the choice of solvent plays a pivotal role in the quantum yield of the reaction; the patent highlights the efficacy of alcohols like methanol and ethanol, as well as ethers like THF, in stabilizing the transition states and facilitating the efficient separation of the product from unreacted starting material via crystallization. This deep understanding of the photophysical parameters ensures that the process is not merely a laboratory curiosity but a robust, reproducible method suitable for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Dydrogesterone Efficiently
The execution of this synthesis requires a disciplined adherence to the specified reaction parameters to maximize yield and purity, beginning with the careful preparation of the bis-ketal protected progesterone derivative. The process flows logically through halogenation and elimination steps to set up the diene system before entering the critical photochemical phase, where control of light intensity and wavelength is non-negotiable for success. Following the photo-inversion, the final deprotection and isomerization steps utilize mild acidic conditions to reveal the active ketone functionalities and establish the final conjugated system, yielding the target API with high structural fidelity. For detailed operational procedures, including specific solvent ratios, reagent equivalents, and workup protocols, please refer to the standardized synthesis guide below.
- Protect the carbonyl groups of Progesterone using ethylene glycol and trimethyl orthoformate to form the bis-ketal intermediate.
- Perform radical bromination followed by debromination using organic bases to establish the 4,6-diene system.
- Execute the critical photochemical inversion using sequential LED irradiation at 265-300nm and 305-365nm to convert the 9α,10β-isomer to the 9β,10α-configuration.
- Complete the synthesis via acid-catalyzed deprotection and double bond isomerization to yield high-purity Dydrogesterone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers a compelling value proposition centered around supply security, cost efficiency, and operational flexibility. By anchoring the synthesis on Progesterone, a globally traded commodity with a mature supply chain, manufacturers can mitigate the risks associated with sourcing obscure or proprietary starting materials that often lead to production delays and price volatility. The elimination of expensive and hazardous reagents, such as the heavy metal oxidants or scarce natural precursors used in legacy methods, translates directly into a streamlined bill of materials that is easier to manage and audit, ensuring consistent availability of critical inputs for continuous manufacturing campaigns.
- Cost Reduction in Manufacturing: The transition from high-energy mercury lamp arrays to energy-efficient LED photoreactors represents a significant reduction in utility costs, as LEDs generate less heat and require less cooling infrastructure, thereby lowering the total cost of ownership for the production facility. Additionally, the high selectivity of the LED-mediated step reduces the load on downstream purification units, such as chromatography columns or recrystallization vessels, leading to substantial savings in solvent consumption and waste disposal fees. The ability to recover and recycle solvents like THF and acetonitrile, as detailed in the patent examples, further enhances the economic viability of the process by minimizing raw material loss and reducing the environmental levy associated with volatile organic compound emissions.
- Enhanced Supply Chain Reliability: Utilizing a synthetic route based on abundant natural steroids ensures that production schedules are not held hostage by the availability of niche chemicals, providing a stable foundation for long-term supply agreements with pharmaceutical partners. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without catastrophic failure, means that the process is resilient to minor operational fluctuations, ensuring high batch-to-batch consistency and reliable delivery timelines. This reliability is crucial for maintaining the continuity of supply for life-saving medications, reducing the risk of stockouts that can impact patient care and damage brand reputation in the highly regulated healthcare sector.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with the photochemical step being adaptable to flow chemistry technologies that allow for linear scale-up without the mass transfer and light penetration limitations of traditional batch reactors. This modularity enables manufacturers to incrementally increase capacity in response to market demand without requiring massive capital expenditure on new infrastructure. Furthermore, the greener profile of the synthesis, characterized by reduced energy consumption and the avoidance of toxic heavy metals, aligns perfectly with corporate sustainability goals and increasingly stringent environmental regulations, facilitating smoother regulatory approvals and enhancing the company's ESG (Environmental, Social, and Governance) rating.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Dydrogesterone synthesis technology, drawing directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational feasibility and strategic benefits for potential partners evaluating this manufacturing route for their supply chains.
Q: How does the LED photochemical method improve upon traditional mercury lamp processes?
A: The patented process utilizes specific single-wavelength LED sources (265-300nm and 305-365nm) which offer superior energy efficiency, precise wavelength control, and significantly reduced heat generation compared to broad-spectrum mercury lamps, leading to higher selectivity and lower impurity profiles.
Q: What are the key advantages regarding raw material availability for this synthesis route?
A: Unlike prior art methods that rely on scarce or non-natural precursors like trans-progesterone, this process starts with commercially abundant Progesterone, ensuring a stable supply chain and reducing raw material procurement costs for large-scale manufacturing.
Q: Is this synthesis pathway suitable for multi-ton commercial production?
A: Yes, the process is explicitly designed for industrial scalability, featuring robust solvent recovery systems, mild reaction conditions, and a continuous-flow compatible photochemical reactor design that facilitates safe and efficient scale-up from pilot to commercial volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dydrogesterone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful translation of a patented laboratory process into a commercial reality requires more than just chemical expertise; it demands a partnership built on technical excellence and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate photochemical steps and sensitive steroid transformations described in CN110818760A are executed with precision and reproducibility. We operate state-of-the-art rigorous QC labs equipped with advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch of Dydrogesterone meets the highest international pharmacopoeia standards before it leaves our facility.
We invite forward-thinking pharmaceutical companies and contract manufacturers to collaborate with us to leverage this cutting-edge synthesis technology for their API needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, as well as obtain specific COA data and route feasibility assessments to validate the fit for your production portfolio. Let us help you secure a sustainable, cost-effective, and high-quality supply of Dydrogesterone, empowering you to bring essential therapies to market faster and more efficiently.
