Advanced Catalytic Decarboxylation for High-Purity Tyramine Production and Commercial Scale-Up
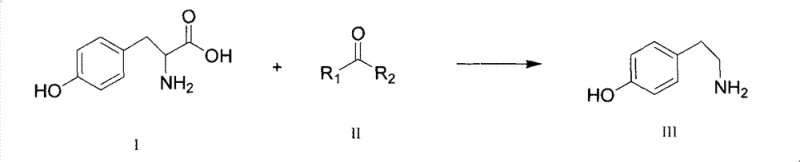
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways for synthesizing critical bioactive intermediates. A significant breakthrough in this domain is documented in Chinese Patent CN101955435B, which details a novel method for preparing tyramine (4-hydroxyphenylethylamine). This compound serves as a vital building block for various therapeutic agents, including drugs for treating migraines and diagnosing pheochromocytoma. The patent introduces a catalytic decarboxylation strategy that fundamentally shifts the production paradigm from harsh thermal conditions or complex enzymatic processes to a more manageable chemical catalysis system. By utilizing saturated ketones as catalysts, this technology enables the conversion of tyrosine to tyramine at significantly reduced temperatures, addressing long-standing issues regarding product stability and color. For a reliable tyramine supplier, understanding and implementing such advanced synthetic routes is essential for maintaining competitiveness in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tyramine has been plagued by significant technical and economic hurdles that hinder efficient manufacturing. Early chemical methods, such as those reported by Sckmitt and Nass, relied on electrophilic substitution reactions using precursors like phenylacetonitrile or 2-phenylbromoethane. These routes are inherently flawed due to their multi-step nature, which inevitably leads to cumulative yield losses and increased operational complexity. Furthermore, alternative thermal decarboxylation methods required extreme temperatures ranging from 255°C to 260°C. Such harsh conditions frequently caused the denaturation of the product, resulting in dark-colored impurities that were difficult to remove without expensive purification steps. Other approaches involving diphenylamine solvents or thiourea additives introduced severe environmental pollution concerns and high raw material costs, making them unsuitable for modern, green manufacturing standards.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN101955435B offers a streamlined and environmentally benign solution. The core innovation lies in the use of saturated ketones, such as diisoamyl ketone or acetophenone, to catalyze the decarboxylation of tyrosine. This catalytic system allows the reaction to proceed efficiently at a much milder temperature range of 140°C to 170°C. This reduction in thermal stress is critical for preserving the integrity of the phenolic structure, ensuring the final product remains a high-quality white powder rather than a tarry residue. The process also incorporates a water separation device to continuously remove water generated during the reaction, driving the equilibrium towards product formation. This method represents a substantial advancement in cost reduction in pharmaceutical intermediates manufacturing by simplifying the workflow and eliminating the need for exotic reagents.
Mechanistic Insights into Ketone-Catalyzed Decarboxylation
The mechanistic elegance of this process centers on the interaction between the amino acid substrate and the ketone catalyst under thermal conditions. While the exact transition states may vary, the saturated ketone likely facilitates the formation of an intermediate Schiff base or acts as a proton shuttle that lowers the activation energy required for the loss of carbon dioxide from the tyrosine molecule. By operating within the 140°C to 170°C window, the system provides sufficient energy to overcome the decarboxylation barrier without triggering the side reactions associated with higher temperatures. The use of nitrogen protection throughout the reaction is crucial to prevent oxidative degradation of the sensitive phenolic moiety, ensuring that the electronic properties of the aromatic ring remain intact. This controlled environment is key to achieving the high purity specifications demanded by regulatory bodies for API intermediates.
Impurity control is another critical aspect where this mechanism excels over traditional thermal routes. In high-temperature pyrolysis, the risk of polymerization and charring is high, leading to complex impurity profiles that are difficult to characterize. However, the ketone-catalyzed pathway promotes a cleaner reaction profile. The subsequent hydrolysis step, where water is added to the cooled reaction mixture, effectively breaks down any transient imine intermediates and precipitates the free amine. The resulting crude solid can be purified through simple washing with solvents like methanol, avoiding the need for column chromatography or recrystallization from hazardous solvents. This simplicity in downstream processing directly translates to reduced waste generation and lower operational expenditures, aligning with the principles of green chemistry.
How to Synthesize Tyramine Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and purity. The process begins with the careful selection of the ketone catalyst and solvent system, which can sometimes be the same substance to further simplify the mixture. Operators must ensure that the water removal system is functioning correctly, as the presence of water can inhibit the forward reaction. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale execution. Detailed standard synthesis steps are provided in the guide below.
- Charge a reaction vessel with L-Tyrosine, a saturated ketone catalyst (such as diisoamyl ketone or acetophenone), and an organic solvent like cyclohexanol.
- Heat the mixture to 140°C–170°C under nitrogen protection while utilizing a water separator to remove generated water continuously.
- Upon completion, cool the reaction, add water for hydrolysis, filter the precipitate, and wash with methanol to obtain white tyramine powder.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic decarboxylation technology offers compelling strategic advantages beyond mere technical feasibility. The elimination of expensive enzymes and toxic solvents like diphenylamine drastically simplifies the raw material sourcing landscape. Suppliers can rely on commodity chemicals such as acetophenone and cyclohexanol, which are readily available in bulk quantities with stable pricing. This shift reduces dependency on niche reagent markets, thereby enhancing supply chain reliability and mitigating the risk of production stoppages due to material shortages. Furthermore, the simplified workup procedure reduces the burden on waste treatment facilities, as the process generates fewer hazardous byproducts compared to older methods involving heavy metals or sulfur-containing compounds.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the significant reduction in energy consumption and reagent costs. By lowering the reaction temperature from over 250°C to approximately 150°C, facilities can utilize standard heating equipment rather than specialized high-temperature reactors, leading to substantial capital expenditure savings. Additionally, the catalyst, being a saturated ketone, is relatively inexpensive and can potentially be recovered and reused, further driving down the variable cost per kilogram. The high purity of the crude product means less solvent and time are spent on purification, directly impacting the bottom line through improved overall process efficiency.
- Enhanced Supply Chain Reliability: The robustness of this chemical route ensures consistent output quality, which is vital for maintaining long-term contracts with downstream pharmaceutical manufacturers. Unlike enzymatic processes that may suffer from batch-to-batch variability due to biological factors, this chemical catalysis offers predictable kinetics and yields. The ability to produce high-purity tyramine with a content exceeding 99% reduces the need for safety stock, as the risk of off-spec batches is minimized. This reliability allows supply chain planners to optimize inventory levels and reduce lead time for high-purity tyramine deliveries to clients.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of complex unit operations. The reaction can be easily adapted to large-scale reactors equipped with standard Dean-Stark traps for water removal. From an environmental perspective, the process avoids the use of chlorinated solvents and heavy metal catalysts, simplifying the permitting process and reducing the cost of effluent treatment. This alignment with environmental regulations ensures long-term operational continuity without the risk of shutdowns due to compliance violations, making it an ideal candidate for commercial scale-up of complex amino acid derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tyramine synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is critical for assessing the feasibility of integrating this method into existing production lines.
Q: How does the ketone-catalyzed method improve product quality compared to thermal decarboxylation?
A: Traditional thermal decarboxylation requires temperatures exceeding 255°C, often leading to product denaturation and dark coloration. The patented ketone-catalyzed route operates at a significantly milder 140°C–170°C, preventing thermal degradation and yielding a white product with purity exceeding 99% after simple washing.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is highly scalable. It eliminates the need for expensive enzymes or toxic solvents like diphenylamine. The use of common saturated ketones as both catalyst and potential solvent simplifies the supply chain, and the straightforward workup (filtration and washing) facilitates easy commercial scale-up of complex amino acid derivatives.
Q: What are the typical yields and purity levels achievable with this method?
A: Experimental data from the patent indicates robust performance, with yields ranging from 64.4% to 72.3% depending on the specific ketone catalyst used. Crucially, the final product consistently achieves high purity levels, often surpassing 99% content, which reduces the need for extensive downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tyramine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes to maintain a competitive edge in the global fine chemical market. Our technical team has extensively analyzed the catalytic decarboxylation methodology described in CN101955435B and possesses the expertise to translate this laboratory-scale success into robust industrial processes. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of tyramine delivered meets the highest international standards for pharmaceutical intermediates.
We invite potential partners to collaborate with us to leverage this advanced technology for their supply chains. By working together, we can conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective, and high-quality supply of tyramine that supports your long-term growth objectives in the pharmaceutical and agrochemical sectors.
