Advanced Catalytic Decarboxylation for High-Purity Tyramine Manufacturing and Commercial Scale-Up
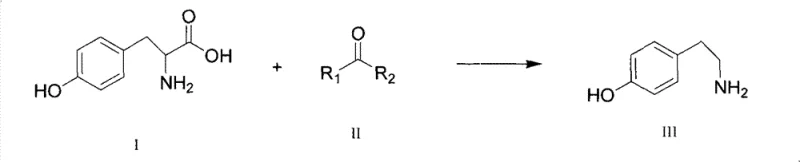
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways for synthesizing critical intermediates, and the production of Tyramine (4-hydroxyphenylethylamine) is no exception. A pivotal advancement in this domain is documented in patent CN101955435B, which discloses a novel method for preparing tyramine through catalytic decarboxylation. This technology represents a significant departure from historical synthesis routes that have long plagued manufacturers with issues regarding yield, purity, and environmental impact. By leveraging a specific catalytic system involving saturated ketones, this method achieves a dramatic reduction in reaction temperature while simultaneously enhancing product quality. For R&D directors and procurement specialists evaluating supply chain resilience, understanding the mechanistic underpinnings and commercial implications of this patent is essential for securing a reliable tyramine supplier. The shift from harsh thermal conditions to a controlled catalytic environment not only optimizes the impurity profile but also aligns with modern green chemistry principles, offering a robust solution for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tyramine has been fraught with significant technical and economic challenges that hinder efficient manufacturing. Early chemical methods, such as those reported by Schmitt and Nass or utilizing phenylacetonitrile and 2-phenylbromoethane, involved lengthy electrophilic substitution reaction routes that resulted in low yields and high production costs. Furthermore, thermal decarboxylation routes using tyrosine as a raw material often required extreme temperatures ranging from 255°C to 260°C, as seen in methods utilizing phenyl ether or diphenylamine solvents. These high-temperature conditions frequently led to product denaturation, deep coloration, and substantial energy consumption, rendering the final product difficult to purify to pharmaceutical grades. Additionally, many traditional processes relied on polluting solvents or expensive reagents, creating significant environmental liabilities and waste disposal burdens that are increasingly unacceptable in modern regulatory landscapes. The cumulative effect of these limitations was a supply chain vulnerable to cost volatility and inconsistent quality, necessitating a fundamental rethinking of the synthetic strategy.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a catalytic decarboxylation reaction mediated by saturated ketones in an organic solvent. This innovation allows the reaction to proceed at significantly milder temperatures, specifically between 140°C and 170°C, which effectively mitigates the risks of thermal degradation and color formation associated with older techniques. The use of saturated ketones, which can also function as the reaction solvent, simplifies the reaction matrix and eliminates the need for exotic or hazardous reagents. By incorporating a water separation device during the reaction, the equilibrium is driven towards the product side, ensuring high conversion rates without the need for excessive reagent loading. This method not only streamlines the operational workflow but also ensures that the post-treatment process is straightforward, typically requiring only simple washing to achieve a white, high-purity product. Such improvements directly address the pain points of cost reduction in pharmaceutical intermediate manufacturing by reducing energy inputs and simplifying downstream processing.
Mechanistic Insights into Ketone-Catalyzed Decarboxylation
The core of this technological breakthrough lies in the specific interaction between the amino acid substrate and the saturated ketone catalyst. In this mechanism, the ketone facilitates the decarboxylation of tyrosine by stabilizing the transition state, thereby lowering the activation energy required for the cleavage of the carbon-carbon bond adjacent to the carboxyl group. Unlike enzymatic systems which are sensitive to pH and temperature fluctuations, or thermal methods which rely solely on brute force energy, this chemical catalysis offers a precise and controllable pathway. The reaction proceeds under nitrogen protection to prevent oxidation of the sensitive phenolic moiety, ensuring that the structural integrity of the tyramine molecule is maintained throughout the process. The choice of saturated ketones, such as diisoamyl ketone or acetophenone, is critical as their steric and electronic properties optimize the catalytic cycle without introducing toxic metal residues. This mechanistic elegance translates directly into a cleaner impurity profile, a key metric for R&D directors focused on the purity and impurity spectrum of API intermediates.
Furthermore, the control of impurities is inherently built into the reaction design through the management of byproducts and solvent interactions. The mild temperature range of 140°C to 170°C prevents the formation of polymeric byproducts and charred residues that are common in high-temperature thermal decarboxylation. The subsequent hydrolysis step, where water is added after the catalytic reaction, effectively quenches any remaining reactive intermediates and facilitates the separation of the organic product from the catalyst system. Because the catalyst is a saturated ketone rather than a heavy metal complex, there is no risk of metal contamination, which is a stringent requirement for pharmaceutical applications. The result is a product that consistently achieves content levels exceeding 98% to 99% after simple washing, demonstrating the robustness of the purification protocol. This level of control over the chemical environment ensures batch-to-batch consistency, a critical factor for maintaining supply chain reliability and meeting the rigorous quality standards of global regulatory bodies.
How to Synthesize Tyramine Efficiently
Implementing this synthesis route requires a clear understanding of the operational parameters to maximize efficiency and safety. The process begins with the precise charging of tyrosine and the selected saturated ketone catalyst into a reaction vessel equipped with a water separator and nitrogen inlet. Maintaining an inert atmosphere is crucial to protect the phenolic group from oxidation, while the continuous removal of water drives the decarboxylation equilibrium forward. The detailed standardized synthesis steps involve specific temperature ramps and workup procedures that ensure the highest possible yield and purity. For technical teams looking to adopt this methodology, adhering to the optimized conditions described in the patent literature is essential for replicating the reported success at scale. The following guide outlines the critical phases of this operation, providing a framework for process engineers to integrate this technology into existing manufacturing lines.
- Combine tyrosine with a saturated ketone catalyst and organic solvent in a reaction vessel under nitrogen protection.
- Heat the mixture to 140°C-170°C while utilizing a water separation device to remove generated water and drive the reaction.
- After completion, cool the mixture, add water for hydrolysis, and perform conventional post-treatment to isolate pure tyramine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this catalytic decarboxylation technology offers profound advantages that resonate deeply with procurement managers and supply chain heads focused on cost efficiency and continuity. The elimination of expensive transition metal catalysts and the reduction in energy consumption due to lower operating temperatures contribute to a substantially reduced cost base for manufacturing. By avoiding the use of highly polluting solvents and complex purification steps required to remove heavy metals, the process minimizes waste treatment costs and environmental compliance risks. This streamlined approach enhances the overall economic viability of producing tyramine, allowing for more competitive pricing structures in the global market without compromising on quality. For organizations seeking cost reduction in pharmaceutical intermediate manufacturing, this technology represents a strategic opportunity to optimize their supply chain expenses while securing a high-quality input material.
- Cost Reduction in Manufacturing: The adoption of saturated ketones as catalysts eliminates the financial burden associated with purchasing and recovering precious metal catalysts, leading to significant direct material savings. Additionally, the lower reaction temperature range drastically reduces energy consumption compared to traditional thermal methods, further driving down utility costs per kilogram of product. The simplified post-treatment process, which avoids complex chromatography or extensive recrystallization steps, reduces labor and solvent usage, contributing to a leaner manufacturing cost structure. These cumulative efficiencies allow suppliers to offer more stable pricing models, shielding buyers from the volatility often seen in markets dependent on scarce catalytic resources.
- Enhanced Supply Chain Reliability: The raw materials required for this process, such as tyrosine and common saturated ketones, are readily available in the global chemical market, reducing the risk of supply bottlenecks. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment limitations or strict environmental controls that might halt more polluting processes. This reliability ensures a consistent flow of high-purity tyramine, enabling downstream manufacturers to maintain their own production schedules without fear of raw material shortages. Reducing lead time for high-purity pharmaceutical intermediates becomes achievable when the synthesis route is this resilient and scalable.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, with mild conditions that are easier to manage in large reactors compared to high-temperature thermal decarboxylation. The absence of toxic heavy metals and the use of environmentally friendlier solvents simplify waste management and ensure compliance with increasingly stringent global environmental regulations. This alignment with green chemistry principles future-proofs the supply chain against regulatory changes that could otherwise render older production methods obsolete. Consequently, partners can confidently invest in long-term capacity expansions knowing that the technology meets both economic and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel tyramine synthesis method. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners. Understanding these details is crucial for making informed decisions about integrating this technology into your supply chain. The answers reflect the consensus on the method's capability to deliver high purity and cost efficiency.
Q: What are the primary advantages of the ketone-catalyzed method over traditional thermal decarboxylation?
A: The ketone-catalyzed method significantly lowers the reaction temperature from the traditional 255-260°C down to 140-170°C. This reduction prevents product denaturation and color deepening, resulting in a white, high-purity product with superior yield compared to older thermal methods.
Q: Does this synthesis route require expensive transition metal catalysts?
A: No, this novel approach utilizes saturated ketones as catalysts, which eliminates the need for expensive transition metals. This simplifies the purification process by removing the requirement for costly heavy metal removal steps, thereby reducing overall production costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It features mild operating conditions, simple operation, and easy control, making it highly suitable for mass production without the environmental pollution associated with previous solvent systems like diphenylamine or fluorene.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tyramine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical industry. As a premier CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the ketone-catalyzed decarboxylation of tyrosine are translated into reliable commercial reality. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of tyramine meets the highest international standards. We understand that for R&D directors and procurement leaders, consistency and purity are non-negotiable, and our infrastructure is designed to deliver exactly that.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient synthesis route can optimize your budget. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate the full potential of partnering with a supplier who prioritizes both innovation and reliability. Together, we can build a supply chain that is not only cost-effective but also resilient and sustainable for the long term.
