Advanced Catalytic Strategy for Scalable 1-Iodoalkyne Manufacturing and Commercial Supply
Advanced Catalytic Strategy for Scalable 1-Iodoalkyne Manufacturing and Commercial Supply
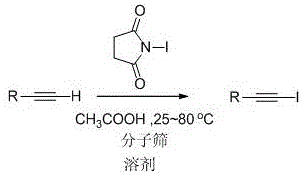
The landscape of organic synthesis for halogenated alkynes is undergoing a significant transformation driven by the need for greener, more cost-effective, and scalable processes. Patent CN111233617A introduces a robust methodology for the synthesis of 1-iodoalkyne compounds that addresses many of the historical bottlenecks associated with traditional halogenation strategies. By utilizing N-iodosuccinimide (NIS) in conjunction with acetic acid as a catalyst and molecular sieves as a desiccant, this technology enables the efficient conversion of terminal alkynes into valuable 1-iodoalkyne intermediates. The process operates under remarkably mild conditions, ranging from 25°C to 80°C, and notably eliminates the stringent requirement for inert gas protection, which is a common constraint in organometallic chemistry. This breakthrough offers a compelling value proposition for industrial manufacturers seeking to optimize their production lines for pharmaceutical and agrochemical intermediates while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-iodoalkynes has relied heavily on methodologies that present substantial challenges for large-scale commercial manufacturing. Traditional approaches often involve the use of strong bases coupled with elemental iodine, or complex systems utilizing hypervalent iodonium salts and expensive transition metal catalysts. These conventional routes frequently suffer from苛刻 reaction conditions that demand strict anhydrous environments and inert atmosphere handling, such as nitrogen or argon blanketing, which significantly increases operational costs and infrastructure requirements. Furthermore, the use of heavy metal catalysts introduces the risk of metal contamination in the final product, necessitating costly and time-consuming purification steps to meet the stringent purity specifications required by the pharmaceutical industry. The environmental footprint of these older methods is also considerable, often generating hazardous waste streams that complicate disposal and regulatory compliance.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a simple yet highly effective catalytic system comprising acetic acid and molecular sieves. This method fundamentally shifts the paradigm by replacing expensive and toxic metal catalysts with inexpensive, commercially available acetic acid. The inclusion of molecular sieves plays a critical role in sequestering water generated during the reaction or present in the solvent, thereby driving the equilibrium towards the desired product and preventing the hydrolysis of sensitive reagents. The operational simplicity is further enhanced by the ability to conduct the reaction in common polar aprotic solvents like acetonitrile, DMF, or DMSO without the need for specialized air-free techniques. This accessibility not only lowers the barrier to entry for production but also enhances the safety profile of the manufacturing process, making it an ideal candidate for continuous flow chemistry or large-batch reactor operations.
Mechanistic Insights into Acetic Acid-Catalyzed Iodination
The mechanistic pathway of this transformation relies on the activation of N-iodosuccinimide (NIS) by the acidic environment provided by acetic acid. In this catalytic cycle, acetic acid likely facilitates the generation of a more electrophilic iodine species from NIS, which then attacks the electron-rich triple bond of the terminal alkyne. The presence of molecular sieves is not merely a passive drying agent but an active component in maintaining the integrity of the electrophilic iodine source, preventing its degradation by moisture which could lead to side reactions or reduced yields. The reaction proceeds through an iodonium intermediate, followed by deprotonation to restore aromaticity or conjugation, ultimately yielding the 1-iodoalkyne. This mechanism is highly selective, minimizing the formation of di-iodinated byproducts or polymerization of the alkyne, which are common issues in less controlled halogenation environments.
From an impurity control perspective, this catalytic system offers distinct advantages. The mild acidity of acetic acid prevents the degradation of acid-sensitive functional groups that might be present on complex alkyne substrates, such as esters or protected amines. Additionally, the byproduct of the reaction, succinimide, is generally water-soluble and can be easily removed during the aqueous workup phase involving sodium thiosulfate quenching and brine washing. This simplifies the downstream processing significantly compared to metal-catalyzed routes where metal scavengers or complex chromatography might be required. The high yields reported across a diverse range of substrates, including those with electron-withdrawing and electron-donating groups, suggest that the electronic nature of the alkyne has a minimal detrimental impact on the reaction efficiency, indicating a robust and forgiving catalytic cycle suitable for varied chemical architectures.
How to Synthesize 1-Iodoalkyne Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves dissolving the terminal alkyne substrate, N-iodosuccinimide, powdered molecular sieves, and acetic acid in a suitable solvent such as acetonitrile. The mixture is then heated to a temperature between 25°C and 80°C, with higher temperatures correlating to shorter reaction times, typically completing within 1.5 hours at 80°C. Upon completion, indicated by TLC analysis, the reaction is quenched with saturated sodium thiosulfate solution to reduce any excess iodine species. The product is then extracted into an organic phase, washed, dried, and purified via standard column chromatography. For detailed standardized operating procedures and specific stoichiometric ratios optimized for different substrates, please refer to the technical guide below.
- Combine terminal alkyne, N-iodosuccinimide (NIS), powdered molecular sieves, and acetic acid in a polar aprotic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature range of 25-80°C without the need for inert gas protection until TLC indicates completion.
- Quench with saturated sodium thiosulfate, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents a multitude of strategic benefits that directly impact the bottom line and operational resilience. The primary driver for cost optimization lies in the substitution of high-value reagents with commodity chemicals. Acetic acid is one of the most inexpensive and widely available industrial acids, and N-iodosuccinimide, while a specialized reagent, is far more manageable and cost-effective than many organometallic alternatives. The elimination of noble metal catalysts removes a significant variable cost from the bill of materials and eradicates the need for expensive metal recovery or scavenging processes. Furthermore, the simplified workup procedure reduces solvent consumption and labor hours associated with purification, leading to substantial overall cost savings in the manufacturing of these critical intermediates.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the removal of expensive transition metal catalysts and the avoidance of cryogenic or strictly anhydrous conditions that require specialized infrastructure. By utilizing acetic acid as a catalyst, the process leverages a low-cost commodity chemical that is readily available in bulk quantities globally, ensuring price stability and reducing exposure to volatile raw material markets. The high atom economy and selectivity of the reaction minimize waste generation, which in turn lowers the costs associated with waste treatment and disposal. Additionally, the ability to run the reaction at elevated temperatures without pressure vessels allows for the use of standard glass-lined or stainless steel reactors, avoiding the capital expenditure associated with high-pressure or exotic alloy equipment.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the fact that this method does not require inert gas protection. This means that production is not dependent on the steady supply of high-purity nitrogen or argon, nor is it vulnerable to leaks or failures in air-exclusion systems that can halt production batches. The reagents used, including acetonitrile and molecular sieves, are standard stock items in almost any chemical facility, reducing the risk of supply disruptions. The robustness of the reaction conditions also implies a wider processing window, making the manufacturing process more tolerant to minor fluctuations in temperature or mixing rates, which enhances batch-to-batch consistency and reliability for long-term supply contracts.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or tonne levels is straightforward due to the absence of exothermic hazards typically associated with strong base/iodine combinations. The mild thermal profile allows for safe heat management in large reactors, facilitating a smoother technology transfer from R&D to commercial production. From an environmental standpoint, the process generates succinimide as a benign byproduct, which is easier to treat than heavy metal waste streams. The reduction in hazardous waste and the use of less toxic reagents align with green chemistry principles, helping companies meet increasingly strict environmental regulations and sustainability goals without compromising on production efficiency or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this acetic acid-catalyzed iodination technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing manufacturing workflows or for sourcing teams negotiating specifications with potential suppliers.
Q: What are the primary advantages of this acetic acid-catalyzed method over traditional metal-catalyzed routes?
A: This method eliminates the need for expensive transition metal catalysts and harsh strong bases, significantly reducing raw material costs and simplifying the removal of heavy metal impurities, which is critical for pharmaceutical grade intermediates.
Q: Does this synthesis require specialized equipment like inert gas lines?
A: No, the patent explicitly states that the reaction does not require isolation from air or inert gas protection, allowing for operation in standard open-vessel reactors which drastically lowers capital expenditure and operational complexity.
Q: What is the substrate scope for this iodination reaction?
A: The method demonstrates excellent tolerance for a wide variety of functional groups including esters, halides, ethers, and amides, making it highly versatile for synthesizing complex building blocks for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Iodoalkyne Supplier
The technological advancements described in patent CN111233617A represent a significant leap forward in the efficient production of 1-iodoalkyne intermediates, offering a pathway to higher purity and lower costs. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality materials regardless of volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 1-iodoalkyne meets the exacting standards required for pharmaceutical and fine chemical applications. We are committed to leveraging such innovative synthetic strategies to deliver superior value to our global partners.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic advantages of switching to this method for your supply chain. We encourage you to contact us today to obtain specific COA data for our current inventory and to receive comprehensive route feasibility assessments tailored to your target molecules, ensuring a seamless and profitable partnership.
