Advanced Aerobic Oxidative Iodination Strategy for Scalable 1-Iodoalkyne Manufacturing
Advanced Aerobic Oxidative Iodination Strategy for Scalable 1-Iodoalkyne Manufacturing
In the rapidly evolving landscape of pharmaceutical intermediate production, the efficient synthesis of functionalized alkynes remains a critical bottleneck for many complex drug candidates. Patent CN112209804B introduces a groundbreaking methodology for the preparation of 1-iodoalkyne compounds that fundamentally shifts the paradigm from traditional, resource-intensive protocols to a streamlined, green chemistry approach. This innovation leverages common inorganic iodide salts as the iodine source and utilizes atmospheric oxygen as the terminal oxidant, effectively eliminating the need for expensive hypervalent iodine reagents or hazardous peroxides. By operating under mild, aerobic conditions at room temperature, this technology offers a robust pathway for generating high-purity alkynyl iodides, which are indispensable building blocks for constructing carbon-carbon and carbon-heteroatom bonds in modern medicinal chemistry. The strategic implementation of this process addresses long-standing challenges regarding cost, safety, and scalability, positioning it as a highly attractive solution for industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct iodination of terminal alkynes to access 1-iodoalkyne derivatives has relied heavily on methodologies that impose significant operational and economic burdens on manufacturing facilities. Traditional routes often necessitate the use of elemental iodine or high-valent iodine compounds, which are not only costly but also generate substantial amounts of chemical waste that require complex disposal procedures. Furthermore, many established protocols demand stringent reaction conditions, such as strictly anhydrous and anaerobic environments, which require specialized equipment like gloveboxes or Schlenk lines, thereby inflating capital expenditure and slowing down throughput. The reliance on heavy metal catalysts in some variations introduces the risk of toxic metal residues in the final product, necessitating additional purification steps that erode overall yield and increase production time. Additionally, the use of aggressive oxidants like peroxides or high-valent metal salts poses safety risks related to thermal runaway and explosion hazards, complicating the scale-up process for commercial production.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN112209804B presents a remarkably simple yet powerful alternative that utilizes benign reagents and ambient conditions to achieve superior results. This novel approach employs inexpensive, water-soluble inorganic iodide salts, such as potassium iodide, in conjunction with sodium phenyl sulfinate derivatives to facilitate the oxidative iodination process efficiently. The reaction proceeds smoothly in low-toxicity alcohol solvents like ethanol, which are environmentally friendly and easy to recover, aligning perfectly with modern green chemistry principles. Crucially, the system harnesses oxygen directly from the air as the oxidant, removing the need for external chemical oxidants and allowing the reaction to be conducted in open vessels at room temperature. This drastic simplification of the reaction setup not only enhances operator safety but also dramatically reduces the complexity of the manufacturing workflow, making it ideal for both laboratory discovery and large-scale commercial synthesis.
Mechanistic Insights into Aerobic Oxidative Iodination
The mechanistic underpinning of this transformation represents a sophisticated interplay between the iodide salt, the sulfinate promoter, and molecular oxygen, avoiding the need for transition metal catalysis. It is hypothesized that the sodium phenyl sulfinate acts as a crucial mediator that facilitates the activation of molecular oxygen, generating reactive oxygen species in situ that subsequently oxidize the iodide anion to an electrophilic iodine species. This activated iodine then reacts selectively with the terminal alkyne triple bond to form the desired carbon-iodine bond with high regioselectivity. The absence of transition metals eliminates the possibility of metal-catalyzed side reactions, such as Glaser coupling or polymerization, which are common pitfalls in alkyne chemistry. This clean reaction profile ensures that the crude reaction mixture contains primarily the target 1-iodoalkyne, significantly simplifying downstream processing and purification efforts.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in maintaining product integrity and minimizing degradation. Because the reaction occurs at room temperature without strong acids or bases, sensitive functional groups on the alkyne substrate, such as esters, halides, or hydroxyl groups, remain intact throughout the process. The use of ethanol as a solvent further contributes to a homogeneous reaction environment that promotes consistent mass transfer and heat dissipation, preventing local hot spots that could lead to byproduct formation. The high selectivity observed across a wide range of substrates, including electron-rich and electron-deficient aryl alkynes as well as heterocycles, suggests a robust catalytic cycle that tolerates diverse electronic environments. This mechanistic stability is essential for ensuring batch-to-batch consistency, a critical requirement for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize 1-Iodoalkyne Efficiently
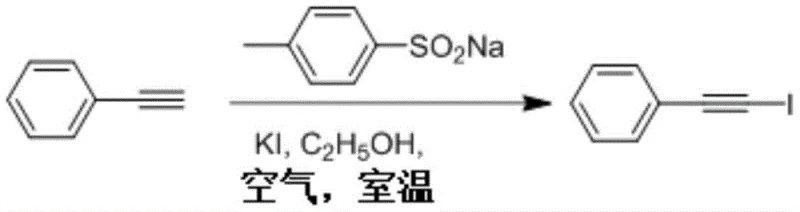
The practical execution of this synthesis protocol is designed to be accessible and straightforward, requiring minimal specialized training or equipment to achieve high yields. The process begins by simply combining the terminal alkyne substrate with the requisite amounts of potassium iodide and sodium p-toluenesulfinate in an ethanol solvent within a standard reaction vessel. The mixture is then stirred openly in the air at room temperature for a defined period, typically around 12 hours, allowing the aerobic oxidation to proceed to completion without the need for inert gas protection. Following the reaction, standard workup procedures involving column chromatography using petroleum ether and ethyl acetate mixtures effectively isolate the pure 1-iodoalkyne product. For a detailed breakdown of the specific molar ratios, solvent volumes, and purification parameters optimized for various substrates, please refer to the standardized synthesis guide below.
- Mix terminal alkyne, soluble inorganic iodide salt (such as KI), sodium phenyl sulfinate or its derivatives, and an alcohol solvent in a reaction vessel.
- Maintain the reaction mixture in an aerobic environment (open to air) at room temperature (10-35°C) and stir for approximately 12 hours.
- Monitor reaction completion via TLC, then separate and purify the product using column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aerobic oxidative iodination technology translates into tangible strategic benefits that extend far beyond simple reagent substitution. By shifting away from proprietary or high-cost oxidants to commodity chemicals like potassium iodide and air, the raw material cost structure is fundamentally altered to favor significant economic efficiency. The elimination of complex reaction conditions, such as the need for cryogenic cooling or high-pressure reactors, reduces energy consumption and lowers the barrier for contract manufacturing organizations to adopt the process. Furthermore, the use of non-toxic ethanol as a solvent simplifies waste management protocols and reduces the environmental compliance burden associated with volatile organic compound emissions. These factors collectively contribute to a more resilient and cost-effective supply chain capable of responding rapidly to market demands for key pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive hypervalent iodine reagents and noble metal catalysts with inexpensive inorganic salts results in a drastic reduction in direct material costs per kilogram of product. Since the process does not require specialized anhydrous solvents or inert atmosphere equipment, the capital investment and operational overhead for production facilities are substantially minimized. The high atom economy and reduced waste generation further lower the costs associated with waste treatment and disposal, enhancing the overall profitability of the manufacturing campaign. Additionally, the simplified purification process reduces solvent consumption during workup, contributing to further operational savings without compromising product quality.
- Enhanced Supply Chain Reliability: Utilizing globally available commodity chemicals like KI and ethanol ensures that raw material sourcing is not subject to the volatility often seen with specialized fine chemical reagents. The robustness of the reaction against moisture and oxygen means that production schedules are less likely to be disrupted by environmental control failures or equipment malfunctions related to inert gas systems. This reliability allows for more accurate forecasting and inventory management, ensuring a steady flow of high-purity intermediates to downstream synthesis units. The ability to run reactions in open vessels also increases facility throughput by reducing turnaround times between batches, thereby improving overall supply continuity.
- Scalability and Environmental Compliance: The mild, room-temperature nature of this reaction makes it inherently safer and easier to scale from gram-scale laboratory experiments to multi-ton commercial production without extensive re-optimization. The absence of hazardous peroxides or explosive reagents significantly mitigates process safety risks, facilitating smoother regulatory approvals and insurance assessments for large-scale plants. Moreover, the alignment with green chemistry principles through the use of air as an oxidant and ethanol as a solvent supports corporate sustainability goals and helps meet increasingly stringent environmental regulations. This eco-friendly profile enhances the marketability of the final API by reducing the overall environmental footprint of the synthesis route.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method for 1-iodoalkyne compounds. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity on process capabilities. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing manufacturing workflows for pharmaceutical intermediates.
Q: What is the primary oxidant used in this novel 1-iodoalkyne synthesis method?
A: Unlike conventional methods that require expensive hypervalent iodine compounds or hazardous peroxides, this patented process utilizes oxygen from the air as the sole oxidant, significantly reducing reagent costs and environmental impact.
Q: Does this synthesis method require strict anhydrous or anaerobic conditions?
A: No, one of the key advantages of this technology is that it operates under ambient conditions without the need for rigorous anhydrous or oxygen-free environments, simplifying operational requirements and equipment needs.
Q: What types of substrates are compatible with this aerobic iodination protocol?
A: The method demonstrates broad substrate adaptability, successfully converting various terminal alkynes including aryl, heteroaryl, alkyl, and silyl-substituted alkynes into their corresponding 1-iodoalkyne derivatives with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Iodoalkyne Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this aerobic oxidative iodination technology in streamlining the production of critical pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle the specific requirements of this green chemistry protocol, maintaining stringent purity specifications through our rigorous QC labs to guarantee the highest quality standards for your drug development programs. We are committed to leveraging this innovative synthesis route to deliver cost-effective and reliable supply solutions for complex alkynyl iodide building blocks.
We invite you to engage with our technical procurement team to discuss how this advanced methodology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this greener process for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our capability to support your long-term manufacturing goals with precision and efficiency.
