Advanced Manufacturing of Capecitabine Intermediates via Bicarbonate-Mediated Carbamylation
Advanced Manufacturing of Capecitabine Intermediates via Bicarbonate-Mediated Carbamylation
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective routes for the production of high-value oncology therapeutics like Capecitabine. Patent CN101993463A introduces a transformative methodology for synthesizing key Capecitabine intermediates, specifically focusing on the optimization of the carbamate formation step and the precursor sugar synthesis. This technical breakthrough addresses long-standing inefficiencies in nucleoside analog manufacturing, particularly the reliance on expensive organic bases and labor-intensive purification techniques. By shifting from traditional pyridine-mediated reactions to a bicarbonate-based system, the process achieves superior conversion rates and minimizes hazardous byproduct generation. Furthermore, the innovation extends to the upstream synthesis of the ribose moiety, where a novel halogenation-protection sequence eliminates the need for column chromatography, drastically improving overall throughput. For R&D directors and process chemists, this patent represents a significant leap forward in designing greener, more economical synthetic pathways for complex nucleoside derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N4-alkoxycarbonyl-5'-deoxy-5-fluorocytidine derivatives has been plagued by suboptimal reaction conditions that hinder commercial viability. Prior art methods, such as those described in WO2008/09245A1, typically employ strong organic bases like pyridine or triethylamine to facilitate the reaction between the cytidine derivative and chloroformates. While effective in small-scale laboratory settings, these bases often promote undesirable side reactions, leading to complex impurity profiles that are difficult to separate. Moreover, the conventional synthesis of the critical sugar intermediate, 5-deoxy-tri-O-acetyl-D-ribose, relies heavily on iodination strategies that generate substantial amounts of triphenylphosphine oxide. Removing this stubborn byproduct traditionally requires column chromatography, a technique that is notoriously inefficient, solvent-intensive, and unsuitable for industrial scale-up. Consequently, yields for these key intermediates often stagnate around 35%, creating a bottleneck that inflates production costs and extends lead times for the final API.
The Novel Approach
The methodology disclosed in CN101993463A offers a compelling solution by fundamentally re-engineering the reaction environment and purification strategy. Instead of relying on nucleophilic organic bases, the inventors utilize inorganic bicarbonates, such as sodium bicarbonate, in solvents like 1,2-dichloroethane under reflux conditions. This subtle yet powerful change creates a milder reaction milieu that suppresses side reactions while ensuring complete conversion of the starting materials. The result is a crude product with an HPLC purity exceeding 96%, achievable through simple aqueous workups rather than complex chromatography. Additionally, the patent introduces a streamlined route for the sugar precursor starting from inosine. By integrating halogenation and hydroxyl protection into a cohesive sequence followed by crystallization, the process bypasses the need for silica gel purification entirely. This holistic approach not only boosts the yield of the sugar intermediate to over 78% but also establishes a continuous, scalable workflow that is ideally suited for the demands of modern pharmaceutical manufacturing.
Mechanistic Insights into Bicarbonate-Mediated Carbamylation
The core chemical innovation lies in the mechanistic behavior of the bicarbonate ion during the nucleophilic substitution at the carbonyl center of the chloroformate. In traditional pyridine-catalyzed systems, the base can sometimes act as a nucleophile itself, forming reactive acyl-pyridinium intermediates that may lead to over-acylation or degradation of the sensitive fluoropyrimidine ring. In contrast, the bicarbonate ion functions primarily as a proton scavenger, neutralizing the hydrochloric acid generated during the formation of the carbamate bond without participating in competing nucleophilic attacks. This selectivity is crucial for maintaining the integrity of the 5-fluorocytosine moiety, which is susceptible to hydrolysis or substitution under harsher basic conditions. The use of 1,2-dichloroethane as the solvent further enhances this selectivity by providing a non-polar environment that favors the precipitation of inorganic salts, driving the equilibrium towards product formation. This mechanistic clarity allows process chemists to predict impurity profiles with greater accuracy, ensuring that the final intermediate meets stringent quality specifications required for downstream API synthesis.
Regarding impurity control, the revised sugar synthesis pathway offers a masterclass in managing phosphorus-containing byproducts. In the prior art, the reaction of inosine with iodine and triphenylphosphine generates stoichiometric amounts of triphenylphosphine oxide, which co-elutes with the product during standard extractions. The new method optimizes the stoichiometry and reaction temperature to favor the formation of the halogenated intermediate, which is then directly acetylated. The subsequent crystallization steps are engineered to exploit the solubility differences between the desired acetylated sugar and the phosphorus residues. By avoiding the column chromatography step, the process not only recovers more product but also prevents the mechanical losses and solvent contamination associated with silica gel beds. This results in a much cleaner intermediate stream entering the glycosylation step, thereby reducing the burden on downstream purification and ensuring a consistent supply of high-quality starting material for the final coupling reactions.
How to Synthesize Capecitabine Intermediate Efficiently
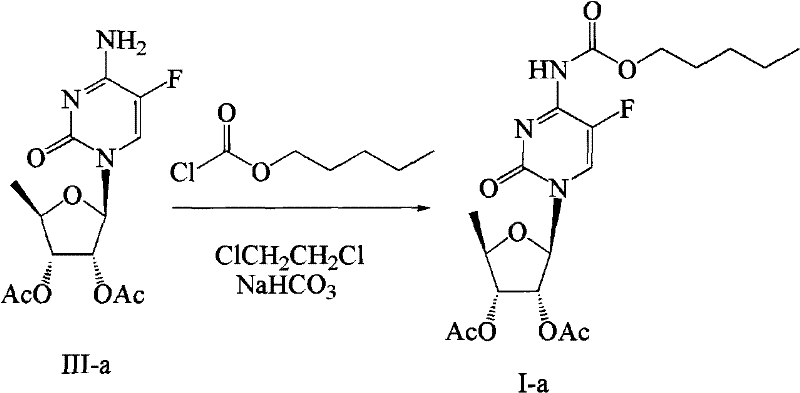
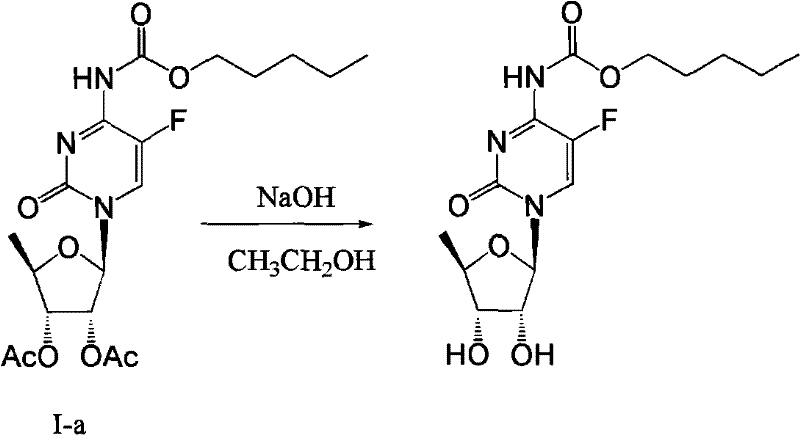
The synthesis of the target Formula I compound involves a three-stage cascade that begins with the modification of inosine and concludes with the carbamylation of the fluorocytidine nucleoside. The process is designed to maximize atom economy and minimize unit operations, making it highly attractive for process development teams looking to transfer technology from the lab to the pilot plant. The initial stage focuses on converting inosine into 5-deoxy-tri-O-acetyl-D-ribose through a sequence of halogenation, protection, and catalytic hydrogenation. This is followed by the condensation of the sugar with 5-fluorocytosine using silylation and Lewis acid catalysis to form the nucleoside backbone. The final and most critical step involves the reaction of this nucleoside with n-amyl chloroformate in the presence of sodium bicarbonate. Detailed standardized operating procedures for each of these stages, including specific temperature ramps, addition rates, and crystallization parameters, are essential for replicating the high yields reported in the patent literature.
- Prepare 5-deoxy-tri-O-acetyl-D-ribose (Formula II) from inosine via halogenation, protection, and reduction steps to avoid column chromatography.
- Couple 5-fluorocytosine with Formula II using HMDS and SnCl4 to generate the nucleoside intermediate (Formula III).
- React Formula III with n-amyl chloroformate in 1,2-dichloroethane using sodium bicarbonate as the base under reflux to yield Formula I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route translates directly into tangible operational efficiencies and risk mitigation. The elimination of column chromatography is perhaps the most significant economic driver, as it removes a major bottleneck that typically limits batch size and throughput in nucleoside manufacturing. Chromatography is not only capital-intensive, requiring large columns and vast quantities of high-purity solvents, but it also introduces variability in batch-to-batch consistency. By replacing this with crystallization-based purification, the process becomes inherently more robust and predictable, allowing for larger batch sizes and reduced cycle times. Furthermore, the switch from expensive organic bases to commodity inorganic bicarbonates reduces raw material costs and simplifies waste disposal protocols, as the aqueous waste streams are easier to treat and neutralize compared to those containing high loads of organic amines.
- Cost Reduction in Manufacturing: The primary cost advantage stems from the drastic simplification of the purification workflow. By avoiding column chromatography for the sugar intermediate, the process eliminates the need for tons of silica gel and hundreds of liters of elution solvents per kilogram of product. This reduction in consumables directly lowers the variable cost of goods sold (COGS). Additionally, the higher yields achieved at each step—particularly the jump from 35% to over 78% in the sugar synthesis—mean that less starting material is required to produce the same amount of final intermediate. This improved material efficiency compounds throughout the synthesis tree, resulting in substantial overall cost savings that can be passed down the supply chain or retained as margin.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical oncology intermediates is paramount, and this process enhances security by utilizing widely available, commodity-grade reagents. Sodium bicarbonate, 1,2-dichloroethane, and inosine are all produced at a global scale, reducing the risk of supply disruptions associated with specialized or custom-synthesized catalysts. The robustness of the crystallization steps also means that the process is less sensitive to minor fluctuations in reaction conditions, leading to higher first-pass yields and fewer failed batches. This consistency ensures a steady flow of material to downstream API manufacturers, minimizing the risk of production delays that could impact the availability of life-saving cancer treatments.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, the new route aligns perfectly with green chemistry principles. The reduction in solvent usage and the elimination of silica waste significantly lower the environmental footprint of the manufacturing process. Easier waste treatment due to the absence of complex organic base residues simplifies compliance with increasingly stringent environmental regulations. Moreover, the scalability of the process is proven by its reliance on standard unit operations like reflux, filtration, and crystallization, which are easily replicated in multi-purpose reactors ranging from 100 liters to 10,000 liters. This ease of scale-up allows manufacturers to respond quickly to market demand surges without requiring significant capital investment in new specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms legacy processes. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement specialists negotiating supply agreements based on these improved specifications.
Q: Why is sodium bicarbonate preferred over pyridine for the carbamate formation step?
A: Sodium bicarbonate significantly reduces side reactions and byproduct formation compared to organic bases like pyridine or triethylamine. It simplifies the post-reaction workup by allowing easy aqueous washing to neutrality, resulting in higher HPLC purity (over 96%) without complex purification steps.
Q: How does this process improve the yield of the sugar intermediate?
A: By utilizing a one-pot halogenation and protection strategy starting from inosine, followed by crystallization rather than column chromatography, the process avoids the significant product loss associated with triphenylphosphine oxide removal. This increases the yield of the key sugar intermediate from approximately 35% in prior art to over 78%.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is highly scalable because it replaces difficult chromatographic purifications with robust crystallization steps. The use of inexpensive inorganic bases and common solvents like 1,2-dichloroethane further enhances its viability for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Capecitabine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires deep technical expertise and rigorous process validation. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this bicarbonate-mediated route are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific solvent systems and crystallization requirements outlined in CN101993463A, guaranteeing that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to continuous improvement, we provide a secure and compliant source for high-purity capecitabine intermediates, enabling our partners to focus on drug development and market expansion with confidence.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic impact of switching to this more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data from our recent pilot runs and to receive comprehensive route feasibility assessments tailored to your volume requirements. Let us help you secure a competitive advantage in the oncology market through superior chemistry and reliable supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
