Revolutionizing Edoxaban Intermediate Production: A Safer, Scalable Diels-Alder Approach
The pharmaceutical industry is constantly seeking robust, safe, and cost-effective pathways for the synthesis of critical anticoagulant intermediates. Patent CN111606826A introduces a groundbreaking preparation method for an Edoxaban intermediate, specifically targeting the complex cyclohexyl structure essential for Factor Xa inhibition. Unlike conventional routes that rely on hazardous azide chemistry, this innovation leverages a sophisticated Diels-Alder cycloaddition followed by a streamlined catalytic hydrogenation sequence. For R&D directors and procurement specialists, this represents a paradigm shift towards safer, high-yield manufacturing. The patent details a four-step protocol that not only bypasses the use of explosive sodium azide but also integrates multiple transformation steps—desulfurization, double bond hydrogenation, nitro reduction, and Boc protection—into a single operational unit. This consolidation drastically reduces processing time and solvent consumption, offering a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the key Edoxaban intermediate, [(1R,2S,5S)-2-amino-5-[(dimethylamino)carbonyl]cyclohexyl]carbamate tert-butyl ester, has been plagued by significant safety and efficiency bottlenecks. Traditional methodologies typically involve the mesylation of cyclohexylamino alcohols followed by a substitution reaction using sodium azide. This azidation step is notoriously problematic; it often suffers from low reaction yields, frequently hovering around 30%, which severely impacts the overall economic viability of the process. Furthermore, the handling of sodium azide presents extreme safety hazards due to its explosive nature and high toxicity, necessitating specialized equipment and rigorous safety protocols that drive up operational expenditures. Additionally, these legacy routes often exhibit poor diastereomeric selectivity, requiring extensive and costly purification steps to remove unwanted isomers. The cumulative effect of these inefficiencies is a lengthy, multi-step synthesis that is both financially burdensome and operationally risky for large-scale manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN111606826A offers a transformative solution by reconstructing the synthetic logic from the ground up. The new route initiates with a Diels-Alder reaction between a thiophene derivative and acrylic acid, establishing the cyclohexene core with high stereocontrol right from the start. This is followed by a clever one-pot catalytic hydrogenation step using Raney Nickel, which simultaneously accomplishes desulfurization, alkene saturation, nitro group reduction, and Boc protection. By merging these four distinct chemical transformations into a single reactor operation, the process eliminates the need for intermediate isolations and significantly shortens the production timeline. Crucially, this approach completely circumvents the use of sodium azide, thereby removing the associated explosion risks and regulatory hurdles. The result is a streamlined, high-yielding process that delivers the target intermediate with superior purity and safety profiles, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Diels-Alder Cyclization and Catalytic Hydrogenation
The core of this synthetic breakthrough lies in the precise orchestration of pericyclic reactions and heterogeneous catalysis. The initial step involves a thermal Diels-Alder cycloaddition between Compound II (a nitro-thiophene derivative) and acrylic acid. Conducted at elevated temperatures of 130-140°C in diethylene glycol dimethyl ether, this reaction forms the bicyclic skeleton with excellent regioselectivity. The subsequent chiral resolution using R-phenethylamine ensures that only the desired enantiomer proceeds down the synthetic pipeline, establishing the critical stereocenters early in the sequence. This early-stage stereocontrol is vital for minimizing the formation of diastereomeric impurities later in the synthesis, directly addressing the purity concerns of R&D teams.
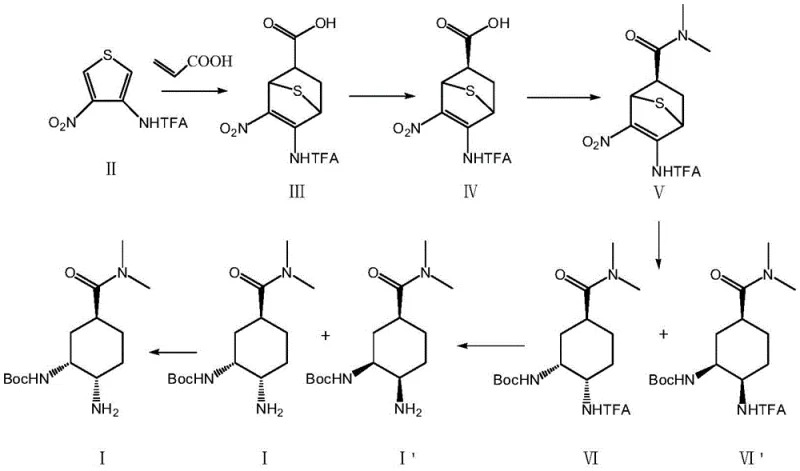
Following the amidation to form Compound V, the process enters its most innovative phase: the Raney Nickel-catalyzed hydrogenation. Under a hydrogen atmosphere at moderate pressures (0.4-0.6 MPa) and temperatures of 45-50°C, the catalyst facilitates a cascade of reductions. The thiophene sulfur atom is removed (desulfurization), the carbon-carbon double bond is saturated, and the nitro group is reduced to an amine. Simultaneously, the newly formed amine reacts with di-tert-butyl dicarbonate (Boc2O) present in the mixture to form the stable carbamate. This tandem reaction mechanism is highly efficient, preventing the accumulation of reactive intermediates and ensuring a clean conversion to Compounds VI and VI'. The final deprotection and resolution steps utilize mild basic conditions and chiral acids like R-mandelic acid to deliver the final API intermediate with chiral purity exceeding 99.5%.
How to Synthesize Edoxaban Intermediate Efficiently
The synthesis of this high-value anticoagulant precursor requires strict adherence to the optimized parameters outlined in the patent to ensure maximum yield and safety. The process is divided into four distinct stages, beginning with the thermal cycloaddition and concluding with chiral purification. Each step has been fine-tuned to balance reaction kinetics with selectivity, ensuring that the final product meets the rigorous quality standards demanded by the pharmaceutical industry. For detailed operational protocols, including specific solvent volumes, stirring rates, and workup procedures, please refer to the standardized guide below.
- Perform Diels-Alder reaction between thiophene derivative II and acrylic acid at 130-140°C, followed by chiral resolution to obtain Compound IV.
- Conduct amidation of Compound IV with dimethylamine hydrochloride at 5-15°C to synthesize Compound V.
- Execute a one-pot reaction with Compound V, Boc2O, and Raney Nickel under hydrogen atmosphere for simultaneous desulfurization, hydrogenation, and Boc protection.
- Complete amino deprotection using ammonia water and perform final chiral resolution to isolate the target Edoxaban intermediate Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers substantial strategic benefits beyond mere technical elegance. The elimination of hazardous reagents like sodium azide translates directly into lower insurance premiums, reduced waste disposal costs, and simplified regulatory compliance. By consolidating multiple reaction steps into a single hydrogenation pot, the process significantly reduces solvent usage and energy consumption, leading to a leaner manufacturing footprint. These efficiencies allow for more competitive pricing structures without compromising on quality, making it an attractive option for long-term supply contracts. Furthermore, the robustness of the Diels-Alder starting material and the reliability of the Raney Nickel catalyst ensure a stable supply chain, minimizing the risk of production delays caused by reagent shortages or safety incidents.
- Cost Reduction in Manufacturing: The integration of desulfurization, hydrogenation, reduction, and protection into a single step drastically cuts down on labor, equipment usage, and solvent costs. By avoiding the low-yield azidation step of traditional methods, the overall material throughput is significantly improved, leading to substantial cost savings per kilogram of finished intermediate. The removal of expensive and hazardous reagents further optimizes the bill of materials, enhancing the overall profit margin for manufacturers.
- Enhanced Supply Chain Reliability: The reliance on common, stable starting materials such as acrylic acid and thiophene derivatives ensures a consistent supply of raw materials, unlike specialized azide reagents which can face supply constraints. The simplified process flow reduces the number of potential failure points in the production line, guaranteeing more predictable lead times for customers. This reliability is crucial for maintaining uninterrupted API production schedules for downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process operates at moderate temperatures and pressures, making it inherently safer and easier to scale from pilot plants to multi-ton commercial reactors. The absence of toxic azide waste streams simplifies effluent treatment and aligns with increasingly stringent environmental regulations. This green chemistry approach not only mitigates environmental risk but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this advanced synthesis method. They are derived from the specific improvements and data points highlighted in the patent documentation, providing clarity on safety, purity, and scalability. Understanding these aspects is essential for stakeholders evaluating the feasibility of integrating this intermediate into their supply chains.
Q: How does this new synthesis route improve safety compared to traditional methods?
A: Traditional methods rely on sodium azide, a highly explosive and toxic reagent, for azidation steps. This novel patent eliminates sodium azide entirely by utilizing a Diels-Alder cycloaddition and catalytic hydrogenation strategy, significantly reducing production risks and hazard management costs.
Q: What are the key advantages regarding stereochemical control in this process?
A: The process achieves high diastereomeric selectivity through optimized Diels-Alder conditions (130-140°C) and specific chiral resolution agents like R-phenethylamine and R-mandelic acid. This ensures the final product meets stringent purity specifications (>99.5% chiral purity) required for anticoagulant APIs.
Q: Is this manufacturing process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. Key steps like the one-pot hydrogenation (desulfurization/reduction/protection) simplify operations by combining multiple transformations into a single reactor run, avoiding complex isolation procedures and facilitating efficient scale-up from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Edoxaban Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of safety and efficiency in the production of complex pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative Diels-Alder route described in CN111606826A can be seamlessly transferred to industrial scale. We are committed to delivering high-purity Edoxaban intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to process optimization allows us to offer a stable, high-quality supply that supports your drug development and commercialization goals.
We invite you to collaborate with us to leverage this advanced technology for your anticoagulant projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your supply chain resilience and profitability.
